Action on Invasives Report 2020
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Developmental Biology and Population Studies on the Citrus Psylla Trioza Erytreae (Del Guercio) (Hemiptera : Triozidae)
Frets - vol. 47, n°5 . 1992 58 3 Developmental Biology and Population Studies on the Citrus Psylla Trioza erytreae (Del Guercio) (Hemiptera : Triozidae) M.A . VAN DEN BERG and Valerie E . DEACON * Biologie du développement et études des populations du psylle des Developmental Biology and Population Studies on the Citrus Psyll a agtines Trioza erytreae (Del Guercio) (Hemiptera : Triozidae). Trioza erytreae (Del Guercio) (Hemiptera : Triozidae). M .A. VAN DEN BERG and Valerie E. DEACO N M .A . VAN I)EN BERG and Valerie E . DEACO N Fruit, vol . 47, n° 5, p . 583-589 . Fruits, vol . 47, n° 5, p . 583-589. RESUME - A une température journalière moyenne de 20,8 °C, les oeufs ABSTRACT - At a daily mean temperature of 20.8°C, Citrus psylla egg s de Trio_a erytreae, le psylle africain des agrumes, éclosent au bout d e hatched after 7 days and the nymphal stage was completed within 18 t o 7 jours et le déroulement du cycle larvaire s ' effectue en 18 à 23 jours . Les 23 days . Field populations in the Hazyview area were either rising or taux d ' abondance de population au champ dans la région de Hazyvie w declining when the egg/nymph/adult ratios increased above or decline d augmentent ou diminuent quand les rapports de pullulation entre les oeufs, below about 15 :13 :1 respectively . The K-value between the egg and les larves et les adultes augmentent ou diminuent dans des proportions nymph counts was 0.55 and between the nymph and adult counts 0.63 . A 15 :13 :1 . -

Citrus Greening Candidatus Liberibacter Text and Images from FDACS Pest Alert, Susan Halbert, DPI
Citrus Greening Candidatus Liberibacter Text and images from FDACS Pest Alert, Susan Halbert, DPI Asian citrus psyllid adult and nymph Yellow shoots and defoliation caused by citrus greening infection. Citrus greening disease or huanglongbing (yellow dragon disease) may be the most serious citrus disease in the world. It is the major limiting factor for citrus production in parts of Asia and Africa. In areas where the disease is endemic, citrus trees may live for 5-8 years and never produce usable fruit (Roistacher 1996). At the time of this writing, citrus greening disease is widespread in Asia, Africa, and the Saudi Arabian Peninsula. It was reported in July 2004 in São Paulo State, Brazil (Coletta-Filho et al. 2004) and in south Miami-Dade County in Florida in August 2005. Citrus greening has not been found in Australia, or the Mediterranean citrus production regions. Origin PATHOGEN: Citrus greening disease is caused by phloem-limited bacteria in the genus Candidatus Liberibacter. Three species are described, including Candidatus Liberibacter asiaticus, Candidatus Liberibacter africanus, and Candidatus Liberibacter americanus (Texeira et al. 2005). VECTORS: The most common vector species, Diaphorina citri Kuwayama, the Asian citrus psyllid, was found for the first time in the USA in Palm Beach County in June 1998, and has since colonized peninsular Florida. Most long distance spread of the vector occurred as a result of movement of Murraya paniculata (L.) Jack (orange-jasmine), a popular ornamental and a preferred host of D. citri. The other reported vector is the African citrus psyllid, Trioza erytreae (del Guercio). Host Range Citrus greening infects most citrus species, hybrids, cultivars, and some citrus relatives. -

Downloaded from the Same Database
G C A T T A C G G C A T genes Article Genetic Basis of Maize Resistance to Multiple Insect Pests: Integrated Genome-Wide Comparative Mapping and Candidate Gene Prioritization A. Badji 1,* , D. B. Kwemoi 2, L. Machida 3 , D. Okii 1, N. Mwila 1, S. Agbahoungba 4, F. Kumi 5 , A. Ibanda 1 , A. Bararyenya 1, M. Solemanegy 1, T. Odong 1, P. Wasswa 1, M. Otim 2, G. Asea 2, M. Ochwo-Ssemakula 1, H. Talwana 1, S. Kyamanywa 1 and P. Rubaihayo 1 1 Department of Agricultural Production, Makerere Univesity, P.O. Box 7062 Kampala, Uganda; [email protected] (D.O.); [email protected] (N.M.); [email protected] (A.I.); [email protected] (A.B.); [email protected] (M.S.); [email protected] (T.O.); [email protected] (P.W.); [email protected] (M.O.-S.); [email protected] (H.T.); [email protected] (S.K.); [email protected] (P.R.) 2 Cereals Program, National Crop Resource Research Institute, P.O. Box 7084 Kampala, Uganda; [email protected] (D.B.K.); [email protected] (M.O.); [email protected] (G.A.) 3 Alliance Bioversity International-CIAT, P.O. Box 24384 Kampala, Uganda; [email protected] 4 Laboratory of Applied Ecology, University of Abomey-Calavi, 01BP 526 Cotonou, Benin; [email protected] 5 Department of Crop Science, University of Cape Coast, P.O. Box 5007 PMB Cape Coast, Ghana; [email protected] * Correspondence: [email protected] Received: 19 May 2020; Accepted: 1 June 2020; Published: 24 June 2020 Abstract: Several species of herbivores feed on maize in field and storage setups, making the development of multiple insect resistance a critical breeding target. -

Natural Colonization of Australian Ladybird Beetle, Cryptolaemus Montrouzieri Mulsant in Papaya Plantation Infested with Paracoc
Journal of Biological Control, 26 (4): 389–390, 2012 Research Note Natural colonization of Australian ladybird beetle, Cryptolaemus montrouzieri Mulsant in papaya plantation infested with Paracoccus marginatus Williams and Granara de Willink in Tamil Nadu M. KALAYANASUNDARAM, M. MANI* and C. SHIVARAJU* Department of Agricultural Entomology, Tamil Nadu Agricultural University, Coimbatore 641 003 Division of Entomology and Nematology, Indian Institute of Horticultural Research, Bangalore 560 089 Corresponding author E-mail: [email protected] ABSTRACT: The role of Cryptolaemus montrouzieri as an effective predator of Paracoccus marginatus Williams and Granara de Willink was often doubtful. Natural colonisation of C. montrouzieri was observed on papaya at Sathyamangalam (Tamil Nadu). The number of larvae were 18 to 30 per leaf. The massive colonisation of C. montrouzieri, will help in its effective utilisation against P. marginatus. KEY WORDS: Paracoccus marginatus, Cryptlolaemus montrouzieri (Article chronicle: Received: 16-08-2012; Revised: 24-11-2012; Accepted: 06-12-2012) The papaya mealybug, Paracoccus marginatus field visits in Coimbatore district. During the visit in Williams and Granara de Willink (Hemiptera: Pseudo- July 2012, large number of Cryptolaemus montrouzieri coccidae), native of Mexico, was reported in Coimbatore Mulsant were found feeding on P. marginatus on papaya (Tamil Nadu) in July 2008. P. marginatus sucks the plantation located at Sathyamangalam. All stages of sap from the leaf resulting in leaf distortion. Fruits C. montrouzieri were found amongst the mealybug covered with mealybugs and sooty mould lose market colonies indicating colonization on papaya mealybug. value (Suresh et al., 2010; Tanwar et al., 2010). Chemicals Number of larvae ranged from 18 to 30 per papaya were used desperately when there was outbreak of leaf. -

Response of Chilo Partellus (Lepidoptera: Crambidae) to Bt Maize in South Africa
Response of Chilo partellus (Lepidoptera: Crambidae) to Bt maize in South Africa J Vorster orcid.org/0000-0001-8126-6860 Dissertation submitted in fulfilment of the requirements for the Masters degree in Environmental Science at the North-West University Supervisor: Prof J van den Berg Co-supervisor: Prof MJ du Plessis Assistant supervisor: Dr A Erasmus Graduation May 2018 23441674 ACKNOWLEDGEMENTS This dissertation would not have been possible without the help of so many people. I am blessed and very grateful to have them in my life. I would like to start with our God Almighty and our Saviour who bestowed upon me the strength, wisdom and peace of mind to finish this project and who also have sent me these blessed people in my life. I would like to thank Prof. Johnnie van den Berg and Dr. Annemie Erasmus for all the guidance and support they have given me. You taught me that small things can make a big difference. Statistics can be difficult sometimes and I thank Prof. Hannalene du Plessis and Prof. Suria Elis for the help with the statistics. Thank you to all the staff at the ARC-GCI that assisted me with the trials in the lab and the planting. Elrine Strydom, Mabel du Toit, Heidi Meyer and Ursula du Plessis, thank you for the countless after hours we had to spend and for the warm hearted kindness you have given me. I would also like to thank my parents whom I dearly love for all the encouragement and motivation to do my best. You taught me that hard work does not come easily, but the fruit that you pick from it is what motivates us. -

Australia and Huanglongbing
AUSTRALIA AND HUANGLONGBING GAC Beattie1, P Holford1, DJ Mabberley1,2, AM Haigh1 and P Broadbent3 1Centre for Plant and Food Science, University of Western Sydney, Locked Bag 1797, Penrith South DC, New South Wales 1797, Australia; 2 Royal Botanic Gardens, Kew, Richmond, Surrey, TW9 3AB, United Kingdom; 3 PO Box 46 Mulgoa, NSW 2745, Australia ABSTRACT Preparations are now underway for potential incursions of huanglongbing and its two known vectors, Diaphorina citri Kuwayama and Trioza erytreae del Guercio, into Australia. These preparations, particularly the development of an incursion management plan (IMP), involve extensive reviews of literature related to the origins of Citrus and huanglongbing, and of host records for the disease and its vectors. This paper briefly discusses issues and aspects of the IMP, including pre- and post-incursion management plans. Key words: Incursion Management Plan (IMP), huanglongbing, Australia INTRODUCTION prepared for the Australian citrus industry (Beattie and Barkley 2009). Preparation of The pathogens that cause huanglongbing the plan has involved a thorough review of (HLB) are not known to occur in Australia host records for the disease and its vectors, neither are the two known vectors of the assessment of likely entry pathways, the disease, the Asiatic citrus psyllid Diaphorina biology of the disease and the vectors, and citri Kuwayama [Hemiptera: Sternorrhyncha: methods to limit the impact of the disease Psylloidea: Psyllidae] and the African citrus should one or both vectors be introduced psyllid -
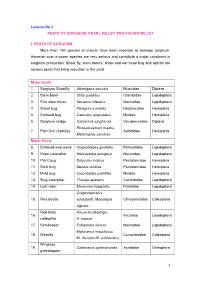
Lecture No 3 PESTS of SORGHUM, PEARL MILLET and FINGER MILLET
Lecture No 3 PESTS OF SORGHUM, PEARL MILLET AND FINGER MILLET I. PESTS OF SORGHUM More than 150 species of insects have been reported to damage sorghum. However over a dozen species are very serious and constitute a major constraint in sorghum production. Shoot fly, stem borers, shoot and ear head bug and aphids are serious pests that bring reduction in the yield. Major pests 1. Sorghum Shootfly Atherigona soccata Muscidae Diptera 2. Stem borer Chilo partellus Crambidae Lepidoptera 3. Pink stem borer Sesamia inferens Noctuidae Lepidoptera 4 Shoot bug Peregrinus maidis Delphacidae Hemiptera 5. Earhead bug Calocoris angustatus Miridae Hemiptera 6. Sorghum midge Contarinia sorghicola Cecidomyiidae Diptera Rhopalosiphum maidis, 7. Plant lice (Aphids) Aphididae Hemiptera Melanaphis sacchari Minor Pests 8. Earhead web worm Cryptoblabes gnidiella Pyraustidae Lepidoptera 9. Gram caterpillar Helicoverpa armigera Noctuidae Lepidoptera 10. Plant bug Dolycoris indicus Pentatomidae Hemiptera 11. Stink bug Nezara viridula Pentatomidae Hemiptera 12. Mirid bug Creontiades pallidifer Miridae Hemiptera 13. Slug caterpillar Thosea apierens Cochlididae Lepidoptera 14. Leaf roller Marasmia trapezalis Pyralidae Lepidoptera Cryptocephalus 15. Flea beetle schestedii, Monolepta Chrysomelidae Coleoptera signata Red hairy Amsacta albistriga, 16. Arctiidae Lepidoptera caterpillar A. moorei 17. Semilooper Eublemma silicula Noctuidae Lepidoptera Myllocerus maculosus 18. Weevils Curculionidae Coleoptera M. discolor,M. subfaciatus Wingless 19. Colemania sphenaroides Acrididae Orthoptera grasshopper MAJOR PESTS 1.Sorghum Shootfly: Atherigona soccata (Muscidae: Diptera) Distribution and status Maharashtra, Andhra Pradesh, Tamil Nadu and Karnataka Host range: Maize, ragi, bajra, rice, wheat and grasses Damage symptoms The maggot on hatching migrates to the upper surface of leaf and enters between the leaf sheath and stem. -

Crambidae Biosecurity Occurrence Background Subfamilies Short Description Diagnosis
Diaphania nitidalis Chilo infuscatellus Crambidae Webworms, Grass Moths, Shoot Borers Biosecurity BIOSECURITY ALERT This Family is of Biosecurity Concern Occurrence This family occurs in Australia. Background The Crambidae is a large, diverse and ubiquitous family of moths that currently comprises 11,500 species globally, with at least half that number again undescribed. The Crambidae and the Pyralidae constitute the superfamily Pyraloidea. Crambid larvae are concealed feeders with a great diversity in feeding habits, shelter building and hosts, such as: leaf rollers, shoot borers, grass borers, leaf webbers, moss feeders, root feeders that shelter in soil tunnels, and solely aquatic life habits. Many species are economically important pests in crops and stored food products. Subfamilies Until recently, the Crambidae was treated as a subfamily under the Pyralidae (snout moths or grass moths). Now they form the superfamily Pyraloidea with the Pyralidae. The Crambidae currently consists of the following 14 subfamilies: Acentropinae Crambinae Cybalomiinae Glaphyriinae Heliothelinae Lathrotelinae Linostinae Midilinae Musotiminae Odontiinae Pyraustinae Schoenobiinae Scopariinae Spilomelinae Short Description Crambid caterpillars are generally cylindrical, with a semiprognathous head and only primary setae (Fig 1). They are often plainly coloured (Fig. 16, Fig. 19), but can be patterned with longitudinal stripes and pinacula that may give them a spotted appearance (Fig. 10, Fig. 11, Fig. 14, Fig. 22). Prolegs may be reduced in borers (Fig. 16). More detailed descriptions are provided below. This factsheet presents, firstly, diagnostic features for the Pyraloidea (Pyralidae and Crambidae) and then the Crambidae. Information and diagnostic features are then provided for crambids listed as priority biosecurity threats for northern Australia. -

Biology of Papaya Mealybug, Paracoccus Marginatus on Laboratory Hosts, Sprouted Potato and Long Bottle Gourd
International Journal of Entomology Research International Journal of Entomology Research ISSN: 2455-4758; Impact Factor: RJIF 5.24 Received: 11-11-2019; Accepted: 12-12-2019 www.entomologyjournals.com Volume 5; Issue 1; January 2020; Page No. 52-56 Biology of papaya mealybug, Paracoccus marginatus on laboratory hosts, sprouted potato and long bottle gourd Iqra1*, Seema Tahir2, Muhammad Samiullah Channa3, Tahir Anwar4 1, 2 Department of Zoology, University of Karachi, Karachi, Sindh, Pakistan 3, 4 Pest Management Research Institute (PMRI), PARC-SARC, Pakistan Agricultural Research Council (PARC), Karachi, Sindh, Pakistan Abstract In this study, biological parameters of the papaya mealybug (PMB), Paracoccus marginatus Williams and Granara de Willink, on two hosts, sprouted potato (Solanum tuberosum) and long bottle gourd (Lagenaria siceraria) vegetables, were investigated under semi-controlled laboratory conditions; to determine the host suitability for rearing of this insect that to be used for augmentation of its natural enemies and for experimental animal in scientific research. Papaya mealybugs have established and completed their lifecycle on both of the hosts; however, there were differences in the biological parameters. The total lifespan of Paracoccus marginatus on sprouted potato was 38.8±4.4 days for female and 25±2.3 days for male while on long bottle gourd, it was 30.8±5.8 days for female and 22.4±2.6 days for male, respectively. The observed nymphal period of the PMB female varies from 15 to 20 (17.8±2.28) days on sprouted potato and 13 to 17 days (15.0±2.0) on long bottle gourd. The nymphal period exhibited by the PMB male on sprouted potato and long bottle gourd was 22.2±2.0 and 20.8±2.3 days, correspondingly. -

Unravelling the Pathogenesis and Molecular Interactions of Liberibacter Phytopathogens with Their Psyllid Vectors
agronomy Review Unravelling the Pathogenesis and Molecular Interactions of Liberibacter Phytopathogens with Their Psyllid Vectors Poulami Sarkar and Murad Ghanim * Department of Entomology, Volcani Center, Rishon LeZion 7505101, Israel; [email protected] * Correspondence: [email protected] Received: 30 June 2020; Accepted: 1 August 2020; Published: 4 August 2020 Abstract: Insect-borne bacterial pathogens pose a global economic threat to many agricultural crops. Candidatus liberibacter species, vectored by psyllids (Hemiptera: psylloidea), are an example of devastating pathogens related to important known diseases such as Huanglongbing or the citrus greening disease, Zebra chip disease, and carrot yellowing, along with vegetative disorders in umbellifers. Studies on liberibacter–plant interactions have gained more focus in disease control over the last few decades. However, successful and sustainable disease management depends on the early disruption of insect–pathogen interactions, thereby blocking transmission. Recent knowledge on the liberibacter genomes and various omics approaches have helped us understand this host–pathogen relationship, despite the complexity associated with the inability to culture these bacteria. Here, we discuss the cellular and molecular processes involved in the response of insect-host immunity, and the liberibacter-associated pathogenesis mechanisms that involve virulence traits and effectors released to manipulate the insect–host defense mechanism for successful transmission. Understanding such mechanisms is an important milestone for developing sustainable means for preventing liberibacter transmission by psyllids. Keywords: liberibacter; psyllids; vector-borne bacteria; pathogenicity; insect defense; phloem-limited bacteria 1. Introduction Candidatus liberibacter species and the diseases they cause have gained recent importance due to their rapid proliferation, leading to global economic losses [1–3]. -

First Report of the Papaya Mealybug, Paracoccus Marginatus (Hemiptera
First report of the papaya mealybug, Paracoccus marginatus (Hemiptera: Pseudococcidae), in China and genetic record for its recent invasion in Asia and Africa Muhammad Z. Ahmed1,5, Ri-Rong He2, Mu-Tao Wu2, Yu-Juan Gu2, Jing-Mei Ren3, Fan Liang2, Hai-Lin Li4, Xue-Nan Hu2, Bao-Li Qiu3, Catharine M. Mannion1, and Jun Ma2,5,* Abstract The papaya mealybug, Paracoccus marginatus Williams and Granara de Willink (Hemiptera: Pseudococcidae), is a polyphagous pest that damages many tropical crops. It is a native of Central America and spread to the Caribbean region and South America in the 1990s; since then it has accidentally been introduced to some islands in the Pacific region and some countries in Africa and Asia. We recorded its presence in China for the first time in 2013 from Guangdong Province and Yunnan Province in southern and southwestern China, respectively. Our genetic analysis revealed that only 1 haplotype of P. marginatus has been recorded in all of Asia, including China, reflecting the very recent invasion of P. marginatus in Asia. This study also includes a summary of the global geographical distribution of P. marginatus. Key Words: genetic identity; haplotype; global distribution; insect pest Resumen La cochinilla de la papaya, Paracoccus marginatus Williams y Granara de Willink (Hemiptera: Pseudococcidae), es una plaga polífaga que daña muchos cultivos tropicales. Es originario de América Central y se extendió a la región del Caribe y América del Sur en la década de 1990; desde entonces ha sido introducida accidentalmente en algunas islas de la región del Pacífico y algunos países de África y Asia. -

Assessment of Grain Yield Losses in Pearl Millet Due to the Millet Stemborer, Coniesta Ignefusalis (Hampson)
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Faculty Publications: Department of Entomology Entomology, Department of 2003 ASSESSMENT OF GRAIN YIELD LOSSES IN PEARL MILLET DUE TO THE MILLET STEMBORER, CONIESTA IGNEFUSALIS (HAMPSON) Aissetou Drame-Yaye Université Abdou Moumouni Ousmane Youm University of Nebraska-Lincoln, [email protected] Jonathan N. Ayertey University of Ghana Follow this and additional works at: https://digitalcommons.unl.edu/entomologyfacpub Part of the Entomology Commons Drame-Yaye, Aissetou; Youm, Ousmane; and Ayertey, Jonathan N., "ASSESSMENT OF GRAIN YIELD LOSSES IN PEARL MILLET DUE TO THE MILLET STEMBORER, CONIESTA IGNEFUSALIS (HAMPSON)" (2003). Faculty Publications: Department of Entomology. 328. https://digitalcommons.unl.edu/entomologyfacpub/328 This Article is brought to you for free and open access by the Entomology, Department of at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Faculty Publications: Department of Entomology by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Insect Sci. Applic. Vol. 23, No. 3,Coniesta pp. 259–265, ignefusalis 2003 larval establishment and damage 0191-9040/03 $3.00 + 0.00 259 Printed in Kenya. All rights reserved © 2003 ICIPE ASSESSMENT OF GRAIN YIELD LOSSES IN PEARL MILLET DUE TO THE MILLET STEMBORER, CONIESTA IGNEFUSALIS (HAMPSON) AISSETOU DRAME-YAYE1, OUSMANE YOUM2 AND JONATHAN N. AYERTEY3 1Faculté d’Agronomie, Université Abdou Moumouni , BP 12040 Niamey, Niger; 2 ICRISAT Sahelian Centre, BP 12404, Niamey, Niger; 3 Department of Crop Science, University of Ghana, P. O. Box LG 44, Legon Accra, Ghana (Accepted 4 June 2003) Abstract—Studies were conducted at the ICRISAT Sahelian Centre, Niger, to assess damage and yield loss by the millet stemborer, Coniesta ignefusalis (Lepidoptera: Pyralidae) on Pennisetum glaucum (L.) R.