Lofentanil Elisa Kit Instructions Product #102419 & 102416 Forensic Use Only
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017
Q UO N T FA R U T A F E BERMUDA PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 BR 111 / 2017 The Minister responsible for health, in exercise of the power conferred by section 48A(1) of the Pharmacy and Poisons Act 1979, makes the following Order: Citation 1 This Order may be cited as the Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017. Repeals and replaces the Third and Fourth Schedule of the Pharmacy and Poisons Act 1979 2 The Third and Fourth Schedules to the Pharmacy and Poisons Act 1979 are repealed and replaced with— “THIRD SCHEDULE (Sections 25(6); 27(1))) DRUGS OBTAINABLE ONLY ON PRESCRIPTION EXCEPT WHERE SPECIFIED IN THE FOURTH SCHEDULE (PART I AND PART II) Note: The following annotations used in this Schedule have the following meanings: md (maximum dose) i.e. the maximum quantity of the substance contained in the amount of a medicinal product which is recommended to be taken or administered at any one time. 1 PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 mdd (maximum daily dose) i.e. the maximum quantity of the substance that is contained in the amount of a medicinal product which is recommended to be taken or administered in any period of 24 hours. mg milligram ms (maximum strength) i.e. either or, if so specified, both of the following: (a) the maximum quantity of the substance by weight or volume that is contained in the dosage unit of a medicinal product; or (b) the maximum percentage of the substance contained in a medicinal product calculated in terms of w/w, w/v, v/w, or v/v, as appropriate. -

Recommended Methods for the Identification and Analysis of Fentanyl and Its Analogues in Biological Specimens
Recommended methods for the Identification and Analysis of Fentanyl and its Analogues in Biological Specimens MANUAL FOR USE BY NATIONAL DRUG ANALYSIS LABORATORIES Laboratory and Scientific Section UNITED NATIONS OFFICE ON DRUGS AND CRIME Vienna Recommended Methods for the Identification and Analysis of Fentanyl and its Analogues in Biological Specimens MANUAL FOR USE BY NATIONAL DRUG ANALYSIS LABORATORIES UNITED NATIONS Vienna, 2017 Note Operating and experimental conditions are reproduced from the original reference materials, including unpublished methods, validated and used in selected national laboratories as per the list of references. A number of alternative conditions and substitution of named commercial products may provide comparable results in many cases. However, any modification has to be validated before it is integrated into laboratory routines. ST/NAR/53 Original language: English © United Nations, November 2017. All rights reserved. The designations employed and the presentation of material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. Mention of names of firms and commercial products does not imply the endorse- ment of the United Nations. This publication has not been formally edited. Publishing production: English, Publishing and Library Section, United Nations Office at Vienna. Acknowledgements The Laboratory and Scientific Section of the UNODC (LSS, headed by Dr. Justice Tettey) wishes to express its appreciation and thanks to Dr. Barry Logan, Center for Forensic Science Research and Education, at the Fredric Rieders Family Founda- tion and NMS Labs, United States; Amanda L.A. -

Understanding and Challenging the Drugs: Chemistry and Toxicology
UNDERSTANDING AND CHALLENGING THE DRUGS: CHEMISTRY AND TOXICOLOGY Presenter: • Dr. Jasmine Drake, Graduate Program Director and Assistant Professor, Administration of Justice Department, Barbara Jordan-Mickey Leland School of Public Affairs, Texas Southern University NACDL Training Defending Drug Overdose Homicides in Pennsylvania Penn State Harrisburg, Middletown, PA November 6th, 2019 11:30- 12:45 p.m. Understanding & Challenging the Drugs: Chemistry & Toxicology Dr. Jasmine Drake, Forensic Science Learning Laboratory, Texas Southern University I. Opioid Drug Classifications A. Types of Opioids B. Classic vs. Synthetic C. Toxicology of Opioids 1) How opioids interact with the body 2) Addiction (psychological vs. physiological II. New Classes of Drugs A. Emerging Threats B. Potency III. National Trends in Opioid Overdose Deaths in the U.S. A. Based on State B. Ethnicity C. Drug-Type (prescription vs. fentanyl vs. heroin) IV. Trends of Opioid Overdose Deaths in Philadelphia A. Based on Ethnicity B. Drug Type (prescription vs. fentanyl vs. heroin) V. Legal Considerations to the Opioid Epidemic A. Punitive Measures vs. Rehabilitative Treatment B. Progressive Jurisdictions Nationwide C. New Legal Measures in Philadelphia VI. Toxicology Reports A. What’s in the report? B. Key Aspects of the Tox Report C. Terminology D. Evaluating and Interpreting the data? E. Questions and considerations. VII. Conclusion and Discussion A. Case Specific Examples B. Sample Toxicology Reports The Opioid Epidemic: What labs have to do with it? Ewa King, Ph.D. Associate Director of Health RIDOH State Health Laboratories Analysis. Answers. Action. www.aphl.org Overview • Overdose trends • Opioids and their effects • Analytical testing approaches • Toxicology laboratories Analysis. Answers. Action. -

Fentanyl and Fentanyl Analogues Dear Judge Pryor
FEDERAL DEFENDER SENTENCING GUIDELINES COMMITTEE Lyric Office Centre 440 Louisiana Street, Suite 1350 Houston, Texas 77002-1634 Chair: Marjorie Meyers Phone: 713.718.4600 November 13, 2017 Honorable William H. Pryor, Jr. Acting Chair United States Sentencing Commission One Columbus Circle, N.E. Suite 2-500, South Lobby Washington, D.C. 20002-8002 Re: Public Comment on Fentanyl and Fentanyl Analogues Dear Judge Pryor: The Commission seeks comment on a number of issues related to fentanyl and fentanyl analogues. Because fentanyl and its analogues account for very few federal drug trafficking offenses,1 and unlike other drugs,2 there has been no reported litigation regarding the appropriate drug equivalency or whether a substance was a fentanyl analogue, most of the information the Commission receives during the comment period will not be derived from federal cases. Defenders have strongly encouraged the Commission to undertake a comprehensive review of the direct harms caused by particular doses of all drugs in the guidelines and amend the guidelines to create proportionate sentences. Such a comprehensive approach is necessary because focusing on a spotlighted drug like fentanyl and its analogues would only exacerbate the disproportionalities in §2D1.1. Much of the disparity created in the drug guidelines is a result of the Commission repeatedly increasing sentences for whatever drug is the current cause cèlebrè with no evidence that increased penalties reduce use or deter distribution.3 1 USSC, Quick Facts: Drug Trafficking Offenses (July 2017) (in FY 2016, crack cocaine, methamphetamine, powder, heroin, oxycodone, and marijuana accounted for 96.3% of drug trafficking offenses). 2 Most of the federal litigation regarding analogues or the appropriate drug quantity has involved synthetic cathinones, cannabinoids, and MDMA even though they account for fewer drug trafficking offenses than the six drugs discussed in the Quick Facts report). -
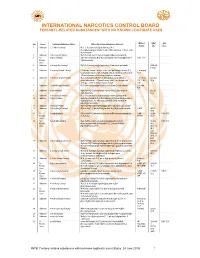
INTERNATIONAL NARCOTICS CONTROL BOARD FENTANYL-RELATED Substancesa with NO KNOWN LEGITIMATE USES
INTERNATIONAL NARCOTICS CONTROL BOARD a FENTANYL-RELATED SUBSTANCES WITH NO KNOWN LEGITIMATE USES Abbrev- CAS Intl No. Uses b Common Substance Name Other/ Alternative Substance Name(s) c iations No.d Ctrl.e 1 Unknown 2,2'-difluorofentanyl N-(1-(2-fluorophenethyl)piperidin-4-yl)-N-(2- fluorophenyl)propionamide; 2'-ortho-difluorofentanyl; 2'-fluoro ortho- fluorofentanyl 2 Unknown 2-fluoro butyrfentanyl N-(2-fluorophenyl)-N-(1-phenethylpiperidin-4-yl) butyramide 3 No 2-fluorofentanyl ortho-fluorofentanyl; N-(2-fluorophenyl)-N-(1-phenethylpiperidin-4- 2-FF; o-FF Known yl)propionamide Uses 4 Unknown 2-furanylethyl fentanyl N-[1-[2-(2-furanyl)ethyl]-4-piperidinyl]-N-phenyl-propanamide 1443-49- 8 (HCl) 5 Unknown 2-isopropylfuranyl fentanyl 2-isopropyl furanyl fentanyl; ortho-isopropyl furanyl fentanyl; N-(2- 2-isopropyl isopropylphenyl)-N-(1-phenethylpiperidin-4-yl)furan-2-carboxamide; Fu-F 2-Furanylfentanyl ortho-2-isopropylphenyl analogue 6 Unknown 2-methoxy furanyl fentanyl N-(2-methoxyphenyl)-N-[1-(2-phenylethyl)-4-piperidinyl]-2- 2-methoxy 101343- furancarboxamide; 2-Furanylfentanyl ortho-2-methoxyphenyl FuF; 2-Meo- 50-4 analogue; ortho-methoxy furanyl fentanyl FuF 7 Unknown 2-methyl furanyl fentanyl N-(1-phenethylpiperidin-4-yl)-N-(o-tolyl)furan-2-carboxamide 2-methyl FuF 8 Unknown 3-allyl fentanyl N-phenyl-N-[1-(2-phenylethyl)-3-(prop-2-en-1-yl)piperidin-4- 82208- yl]propanamide 84-2 9 Unknown 3-fluoro butyrfentanyl N-(3-fluorophenyl)-N-(1-phenethylpiperidin-4-yl) butyramide 10 Unknown 3-fluorofentanyl meta-fluorofentanyl; N-(3-fluorophenyl)-N-(1-phenethylpiperidin-4- -

Pharmaceutical Appendix to the Tariff Schedule 2
Harmonized Tariff Schedule of the United States (2007) (Rev. 2) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2007) (Rev. 2) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. ABACAVIR 136470-78-5 ACIDUM LIDADRONICUM 63132-38-7 ABAFUNGIN 129639-79-8 ACIDUM SALCAPROZICUM 183990-46-7 ABAMECTIN 65195-55-3 ACIDUM SALCLOBUZICUM 387825-03-8 ABANOQUIL 90402-40-7 ACIFRAN 72420-38-3 ABAPERIDONUM 183849-43-6 ACIPIMOX 51037-30-0 ABARELIX 183552-38-7 ACITAZANOLAST 114607-46-4 ABATACEPTUM 332348-12-6 ACITEMATE 101197-99-3 ABCIXIMAB 143653-53-6 ACITRETIN 55079-83-9 ABECARNIL 111841-85-1 ACIVICIN 42228-92-2 ABETIMUSUM 167362-48-3 ACLANTATE 39633-62-0 ABIRATERONE 154229-19-3 ACLARUBICIN 57576-44-0 ABITESARTAN 137882-98-5 ACLATONIUM NAPADISILATE 55077-30-0 ABLUKAST 96566-25-5 ACODAZOLE 79152-85-5 ABRINEURINUM 178535-93-8 ACOLBIFENUM 182167-02-8 ABUNIDAZOLE 91017-58-2 ACONIAZIDE 13410-86-1 ACADESINE 2627-69-2 ACOTIAMIDUM 185106-16-5 ACAMPROSATE 77337-76-9 -

Appendix-2Final.Pdf 663.7 KB
North West ‘Through the Gate Substance Misuse Services’ Drug Testing Project Appendix 2 – Analytical methodologies Overview Urine samples were analysed using three methodologies. The first methodology (General Screen) was designed to cover a wide range of analytes (drugs) and was used for all analytes other than the synthetic cannabinoid receptor agonists (SCRAs). The analyte coverage included a broad range of commonly prescribed drugs including over the counter medications, commonly misused drugs and metabolites of many of the compounds too. This approach provided a very powerful drug screening tool to investigate drug use/misuse before and whilst in prison. The second methodology (SCRA Screen) was specifically designed for SCRAs and targets only those compounds. This was a very sensitive methodology with a method capability of sub 100pg/ml for over 600 SCRAs and their metabolites. Both methodologies utilised full scan high resolution accurate mass LCMS technologies that allowed a non-targeted approach to data acquisition and the ability to retrospectively review data. The non-targeted approach to data acquisition effectively means that the analyte coverage of the data acquisition was unlimited. The only limiting factors were related to the chemical nature of the analyte being looked for. The analyte must extract in the sample preparation process; it must chromatograph and it must ionise under the conditions used by the mass spectrometer interface. The final limiting factor was presence in the data processing database. The subsequent study of negative MDT samples across the North West and London and the South East used a GCMS methodology for anabolic steroids in addition to the General and SCRA screens. -

Bond Elut Certify Methods Manual
AGILENT BOND ELUT CERTIFY AND CERTIFY II METHODS MANUAL TABLE OF CONTENTS INTRODUCTION AND OVERVIEW OF THE MANUAL .................................3 M2724 Meperidine (Pethidine) in Urine by GC or GC/MS ........................... 49 SUMMARY OF BOND ELUT CERTIFY AND CERTIFY II M2725 Methadone in Urine by GC or GC/MS ............................................. 50 MIXED MODE EXTRACTION ............................................................................5 M2726 Methaqualone in Urine by GC or GC/MS ........................................ 51 METHOD OPTIMIZATION .................................................................................6 M2727A 6-Monoacetyl Morphine in Urine by GC or GC/MS ........................ 52 PART NUMBERS................................................................................................8 M2727B 6-Monoacetyl Morphine in Urine by LC or LC/MS ......................... 53 SOLVENTS, SOLVENT MIXTURES, REAGENTS, AND SOLUTIONS .............................................................................................11 M2728 Nicotine in Urine by GC or GC/MS ................................................... 54 EQUIPMENT AND ACCESSORIES ................................................................17 M2729 Opiates (Free/Unbound) in Serum, Plasma, or Whole Blood by GC or GC/MS....................................... 55 BOND ELUT CERTIFY EXTRACTION METHODS ........................................ 29 M2730A Opiates in Urine by GC or GC/MS .................................................... 56 M2707A -

Advisories on Fentanyl and Other Synthetic Opioids
FIN-2019-A006 August 21, 2019 Advisory to Financial Institutions on Illicit Financial Schemes and Methods Related to the Trafficking of Fentanyl and Other Synthetic Opioids Transnational criminal organizations (TCOs), foreign fentanyl suppliers, and Internet purchasers located in the United States engage in the trafficking of fentanyl, fentanyl analogues, and other synthetic opioids and the subsequent laundering of the proceeds from such illegal sales. Introduction The Financial Crimes Enforcement Network (FinCEN) is This Advisory should be issuing this advisory1 to alert financial institutions to illicit shared with: financial schemes and mechanisms related to the trafficking • Chief Executive Officers of fentanyl, fentanyl analogues, and other synthetic • Chief Operations Officers opioids,2 and to assist them in detecting and reporting • Chief Risk Officers related activity. • Legal Departments The United States is in the midst of an unparalleled • Chief Compliance/BSA Officers epidemic of addiction and death, fueled by the illicit • AML Officials trafficking, sale, distribution, and misuse of fentanyl and other synthetic opioids. The statistics are sobering; • Sanctions Compliance Officials between 2013 and 2017, deaths in the United States from • Cybersecurity Units synthetic opioids, other than methadone, increased over 800 percent.3 Every day in the United States, more than 130 people die from an opioid-related overdose.4 These numbers alone cannot fully capture the 1. This advisory is also part of a larger, United States government Fentanyl advisory covering the movement, manufacturing, marketing, and monetary aspects of the trafficking of fentanyl and other synthetic opioids. A summary of the 21st Century Drug Trafficking: Advisories on Fentanyl and Other Synthetic Opioids, which includes links to each advisory, can be found here: https://www.whitehouse.gov/wp-content/uploads/2019/08/White-House- Fentanyl-Advisories-Summary.pdf. -

Fentanyl Family at the Mu-Opioid Receptor: Uniform Assessment of Binding and Computational Analysis
molecules Article Fentanyl Family at the Mu-Opioid Receptor: Uniform Assessment of Binding and Computational Analysis Piotr F. J. Lipi ´nski 1,* , Piotr Kosson 2, Joanna Matali ´nska 1, Piotr Roszkowski 3, Zbigniew Czarnocki 3, Małgorzata Jaro ´nczyk 4 , Aleksandra Misicka 1,3 , Jan Cz. Dobrowolski 4 and Joanna Sadlej 4,5,* 1 Department of Neuropeptides, Mossakowski Medical Research Centre, Polish Academy of Sciences, 02-106 Warsaw, Poland; [email protected] (J.M.); [email protected] (A.M.) 2 Toxicology Research Laboratory, Mossakowski Medical Research Centre, Polish Academy of Sciences, 02-106 Warsaw, Poland; [email protected] 3 Faculty of Chemistry, University of Warsaw, 02-093 Warsaw, Poland; [email protected] (P.R.); [email protected] (Z.C.); [email protected] (A.M.) 4 National Medicines Institute, 00-725 Warsaw, Poland; [email protected] (M.J.); [email protected] (J.Cz.D.) 5 Faculty of Mathematics and Natural Sciences, University of Cardinal Stefan Wyszy´nski,1/3 Wóycickiego-Str., 01-938 Warsaw, Poland * Correspondence: [email protected] (P.F.J.L.); [email protected] (J.S.) Received: 30 January 2019; Accepted: 15 February 2019; Published: 19 February 2019 Abstract: Interactions of 21 fentanyl derivatives with µ-opioid receptor (µOR) were studied using experimental and theoretical methods. Their binding to µOR was assessed with radioligand competitive binding assay. A uniform set of binding affinity data contains values for two novel and one previously uncharacterized derivative. The data confirms trends known so far and thanks to their uniformity, they facilitate further comparisons. -

A Lc-Ms/Ms-Based Method for the Multiplex Detection of 24
A LC-MS/MS-BASED METHOD FOR THE MULTIPLEX DETECTION OF 24 FENTANYL ANALOGS AND METABOLITES IN WHOLE BLOOD AT SUB NG ML-1 CONCENTRATIONS A thesis submitted in partial fulfillment of the requirements for the degree of Master of Science By KRAIG EDWARD STRAYER B.S., Wright State University, 2015 2018 Wright State University Copyright Permission for Use of ACS Omega Article The proceeding thesis is reproduced from a recently published ACS Omega manuscript authored by Kraig E. Strayer. The article, “LC/MS-MS-Based Method for the Multiplex Detection for 24 Fentanyl Analogues and Metabolites in Whole Blood at Sub ng mL-1 Concentrations” was accepted on January 3rd, 2018 (DOI: 10.1021/acsomega.7b01536, https://pubs.acs.org/doi/10.1021/acsomega.7b01536). Permission to use this work in its entirety for this thesis/dissertation has been granted by the ACS Omega editorial staff. Further permissions related to the material excerpted should be directed to the ACS. The full citation of the early access article is the following: Strayer, K. E.; Antonides, H. M.; Juhascik, M. P.; Daniulaityte, R.; Sizemore, I. E. LC- MS/MS-Based Method for the Multiplex Detection of 24 Fentanyl Analogues and Metabolites in Whole Blood at Sub ng mL-1 Concentrations. ACS Omega 2018, 3, 514- 523. WRIGHT STATE UNIVERSITY GRADUATE SCHOOL February 23, 2018 I HEREBY RECOMMEND THAT THE THESIS PREPARED UNDER MY SUPERVISION BY Kraig Edward Strayer ENTITLED A LC-MS/MS-Based Method for the Multiplex Detection of 24 Fentanyl Analogs and Metabolites in Whole Blood at Sub ng mL-1 Concentrations BE ACCEPTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF Master of Science. -

Words Underlined Are Additions. Hb0477-01-C1 FLORIDA HOUSE of REPRESENTATIVE S
FLORIDA HOUSE OF REPRESENTATIVE S CS/HB 477 2017 1 A bill to be entitled 2 An act relating to controlled substances; amending s. 3 381.887, F.S.; providing that certain emergency 4 responders and crime laboratory personnel may possess, 5 store, and administer emergency opioid antagonists; 6 amending s. 782.04, F.S.; providing that unlawful 7 distribution of specified controlled substances and 8 analogs or mixtures thereof by an adult which 9 proximately cause a death is murder; providing 10 criminal penalties; creating s. 893.015, F.S.; 11 specifying purpose relating to drug abuse prevention 12 and control; providing that a reference to ch. 893, 13 F.S., or to any section or portion thereof, includes 14 all subsequent amendments; amending s. 893.03, F.S.; 15 adding certain synthetic opioid substitute compounds 16 to the list of Schedule I controlled substances; 17 amending s. 893.13, F.S.; prohibiting possession of 18 more than 10 grams of specified substances; providing 19 criminal penalties; amending s. 893.135, F.S.; 20 revising the substances that constitute the offenses 21 of trafficking and capital trafficking in, and capital 22 importation of, hydrocodone and oxycodone; creating 23 the offense of trafficking in fentanyl; providing 24 penalties and specifying minimum terms of imprisonment 25 and fines based on the quantity involved in the Page 1 of 167 CODING: Words stricken are deletions; words underlined are additions. hb0477-01-c1 FLORIDA HOUSE OF REPRESENTATIVE S CS/HB 477 2017 26 offense; revising the substances that constitute