1 Electronic Supporting Information 2 Effect of Secondary Organic
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Formaldehyde Induces Bone Marrow Toxicity in Mice by Inhibiting Peroxiredoxin 2 Expression
MOLECULAR MEDICINE REPORTS 10: 1915-1920, 2014 Formaldehyde induces bone marrow toxicity in mice by inhibiting peroxiredoxin 2 expression GUANGYAN YU1, QIANG CHEN1, XIAOMEI LIU1, CAIXIA GUO2, HAIYING DU1 and ZHIWEI SUN1,2 1Department of Preventative Medicine, School of Public Health, Jilin University, Changchun, Jilin 130021; 2 Department of Hygenic Toxicology, School of Public Health, Capital Medical University, Beijing 100069, P.R. China Received November 4, 2013; Accepted June 5, 2014 DOI: 10.3892/mmr.2014.2473 Abstract. Peroxiredoxin 2 (Prx2), a member of the perox- Introduction iredoxin family, regulates numerous cellular processes through intracellular oxidative signal transduction pathways. Formaldehyde (FA) is an environmental agent commonly Formaldehyde (FA)-induced toxic damage involves reactive found in numerous products, including paint, cloth and oxygen species (ROS) that trigger subsequent toxic effects and exhaust gas, as well as other medicinal and industrial products. inflammatory responses. The present study aimed to inves- FA exposure has raised significant concerns due to mounting tigate the role of Prx2 in the development of bone marrow evidence suggesting its carcinogenic potential and severe toxicity caused by FA and the mechanism underlying FA effects on human health (1). Epidemiological and experi- toxicity. According to the results of the preliminary inves- mental data have demonstrated that FA may cause leukemia, tigations, the mice were divided into four groups (n=6 per particularly myeloid leukemia; however, the mechanism group). One group was exposed to ambient air and the other underlying this effect remains unclear (2-4). In the process three groups were exposed to different concentrations of FA of tumor formation, DNA damage may be the initiating (20, 40, 80 mg/m3) for 15 days in the respective inhalation factor (5), while myeloperoxidase (MPO) activity is associ- chambers, for 2 h a day. -

Peroxiredoxins in Neurodegenerative Diseases
antioxidants Review Peroxiredoxins in Neurodegenerative Diseases Monika Szeliga Mossakowski Medical Research Centre, Department of Neurotoxicology, Polish Academy of Sciences, 5 Pawinskiego Street, 02-106 Warsaw, Poland; [email protected]; Tel.: +48-(22)-6086416 Received: 31 October 2020; Accepted: 27 November 2020; Published: 30 November 2020 Abstract: Substantial evidence indicates that oxidative/nitrosative stress contributes to the neurodegenerative diseases. Peroxiredoxins (PRDXs) are one of the enzymatic antioxidant mechanisms neutralizing reactive oxygen/nitrogen species. Since mammalian PRDXs were identified 30 years ago, their significance was long overshadowed by the other well-studied ROS/RNS defense systems. An increasing number of studies suggests that these enzymes may be involved in the neurodegenerative process. This article reviews the current knowledge on the expression and putative roles of PRDXs in neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease and dementia with Lewy bodies, multiple sclerosis, amyotrophic lateral sclerosis and Huntington’s disease. Keywords: peroxiredoxin (PRDX); oxidative stress; nitrosative stress; neurodegenerative disease 1. Introduction Under physiological conditions, reactive oxygen species (ROS, e.g., superoxide anion, O2 -; · hydrogen peroxide, H O ; hydroxyl radical, OH; organic hydroperoxide, ROOH) and reactive nitrogen 2 2 · species (RNS, e.g., nitric oxide, NO ; peroxynitrite, ONOO-) are constantly produced as a result of normal · cellular metabolism and play a crucial role in signal transduction, enzyme activation, gene expression, and regulation of immune response [1]. The cells are endowed with several enzymatic (e.g., glutathione peroxidase (GPx); peroxiredoxin (PRDX); thioredoxin (TRX); catalase (CAT); superoxide dismutase (SOD)), and non-enzymatic (e.g., glutathione (GSH); quinones; flavonoids) antioxidant systems that minimize the levels of ROS and RNS. -

Surface Phenotype Changes and Increased Response to Oxidative Stress in CD4+Cd25high T Cells
biomedicines Article Surface Phenotype Changes and Increased Response to Oxidative Stress in CD4+CD25high T Cells Yoshiki Yamamoto 1,*, Takaharu Negoro 2, Rui Tada 3 , Michiaki Narushima 4, Akane Hoshi 2, Yoichi Negishi 3,* and Yasuko Nakano 2 1 Department of Paediatrics, Tokyo Metropolitan Ebara Hospital, Tokyo 145-0065, Japan 2 Department of Pharmacogenomics, School of Pharmacy, Showa University, Tokyo 142-8555, Japan; [email protected] (T.N.); [email protected] (A.H.); [email protected] (Y.N.) 3 Department of Drug Delivery and Molecular Biopharmaceutics, School of Pharmacy, Tokyo University of Pharmacy and Life Sciences, Tokyo 192-0392, Japan; [email protected] 4 Department of Internal Medicine, Showa University Northern Yokohama Hospital, Kanagawa 224-8503, Japan; [email protected] * Correspondence: [email protected] (Y.Y.); [email protected] (Y.N.); Tel.: +81-3-5734-8000 (Y.Y.); +81-42-676-3182 (Y.N.) + + + + Abstract: Conversion of CD4 CD25 FOXP3 T regulatory cells (Tregs) from the immature (CD45RA ) to mature (CD45RO+) phenotype has been shown during development and allergic reactions. The relative frequencies of these Treg phenotypes and their responses to oxidative stress during devel- opment and allergic inflammation were analysed in samples from paediatric and adult subjects. The FOXP3lowCD45RA+ population was dominant in early childhood, while the percentage of high + FOXP3 CD45RO cells began increasing in the first year of life. These phenotypic changes were observed in subjects with and without asthma. Further, there was a significant increase in phospho- Citation: Yamamoto, Y.; Negoro, T.; + high rylated ERK1/2 (pERK1/2) protein in hydrogen peroxide (H2O2)-treated CD4 CD25 cells in Tada, R.; Narushima, M.; Hoshi, A.; adults with asthma compared with those without asthma. -

In Thyroid Cancer
Metere et al. Cancer Cell Int (2018) 18:7 https://doi.org/10.1186/s12935-018-0504-4 Cancer Cell International PRIMARY RESEARCH Open Access A possible role for selenoprotein glutathione peroxidase (GPx1) and thioredoxin reductases (TrxR1) in thyroid cancer: our experience in thyroid surgery Alessio Metere1* , Francesca Frezzotti1, Claire Elizabeth Graves2, Massimo Vergine1, Alessandro De Luca1, Donatella Pietraforte3 and Laura Giacomelli1 Abstract Background: Oxidative stress is responsible for some alterations in the chemical structure and, consequently, in the function of proteins, lipids, and DNA. Recent studies have linked oxidative stress to cancers, particularly thyroid cancer, but the mechanisms remain unclear. Here, we further characterize the role of oxidative stress in thyroid cancer by analyzing the expression of two selenium antioxidant molecules, glutathione peroxidase (GPx1) and thioredoxin reductase (TrxR1) in thyroid cancer cells. Methods: Samples of both healthy thyroid tissue and thyroid tumor were taken for analysis after total thyroidectomy. The expression of GPx1 and TrxR1 was revealed by Western blot analysis and quantifed by densitometric analy- ses, while the evaluation of free radicals was performed by Electron Paramagnetic Resonance (EPR)-spin trapping technique. Results: Our results show a decrease in the expression of GPx1 and TrxR1 ( 45.7 and 43.2% respectively, p < 0.01) in the thyroid cancer cells compared to the healthy cells. In addition, the EPR− technique− shows an increase of free radicals in tumor tissue, signifcantly higher than that found in healthy thyroid tissue ( 116.3%, p < 0.01). + Conclusions: Our fndings underscore the relationship between thyroid cancer and oxidative stress, showing the imbalance of the oxidant/antioxidant system in thyroid cancer tissue. -

Oxidative Protein Biogenesis and Redox Regulation in the Mitochondrial Intermembrane Space
Cell Tissue Res (2017) 367:43–57 DOI 10.1007/s00441-016-2488-5 REVIEW Oxidative protein biogenesis and redox regulation in the mitochondrial intermembrane space Phanee Manganas1 & Lisa MacPherson1 & Kostas Tokatlidis1 Received: 2 June 2016 /Accepted: 5 August 2016 /Published online: 8 September 2016 # The Author(s) 2016. This article is published with open access at Springerlink.com Abstract Mitochondria are organelles that play a central role membrane (IM). The presence of these two membranes allows in cellular metabolism, as they are responsible for processes the formation of two aqueous subcompartments within the such as iron/sulfur cluster biogenesis, respiration and apopto- mitochondria, which are the intermembrane space (IMS) and sis. Here, we describe briefly the various protein import path- the matrix. Each one of these compartments is characterised ways for sorting of mitochondrial proteins into the different by a specific set of proteins that carry out specialised func- subcompartments, with an emphasis on the targeting to the tions. The majority of these proteins are encoded in the nucle- intermembrane space. The discovery of a dedicated redox- ar genome and synthesised in the cytosol, making it necessary controlled pathway in the intermembrane space that links pro- for the mitochondria to possess mechanisms through which to tein import to oxidative protein folding raises important ques- import all the proteins required for the correct function of the tions on the redox regulation of this process. We discuss the organelle (Neupert 1997). During the import process, the in- salient features of redox regulation in the intermembrane coming proteins are targeted to their correct location within space and how such mechanisms may be linked to the more the organelle by utilising a series of different import pathways. -

PRDX4 (Human) ELISA Kit 1
PRDX4 (Human) ELISA Kit 1. The Association of Peroxiredoxin 4 with the Initiation and Progression of Hepatocellular Carcinoma. Guo X, Catalog Number: KA2121 Noguchi H, Ishii N, Homma T, Hamada T, Hiraki T, Zhang J, Matsuo K, Yokoyama S, Ishibashi H, Regulatory Status: For research use only (RUO) Fukushige T, Kanekura T, Fujii J, Uramoto H, Tanimoto A, Yamada S. Antioxid Redox Signal. 2018 Apr 24. Product Description: PRDX4 (Human) ELISA Kit is a [Epub ahead of print] sandwich enzyme immunoassay for the quantitative 2. Galectin-3 downregulates antioxidant peroxiredoxin-4 measurement of human PRDX4. in human cardiac fibroblasts: a new pathway to induce cardiac damage? Ibarrola J, Arrieta V, Sadaba R, Suitable Sample: Buffered solution Martinez-Martinez E, Garcia-Pena A, Alvarez V, Fernandez-Celis A, Gainza A, Santamaria E, Sample Volume: 100 uL Fernandez-Irigoyen J, Cachofeiro V, Zalba G, Fay R, Label: HRP-conjugated Rossignol P, Lopez-Andres N. Clin Sci (Lond). 2018 Apr 19. pii: CS20171389. [Epub ahead of print] Detection Method: Colorimetric 3. Overexpression of Peroxiredoxin 4 Affects Intestinal Function in a Dietary Mouse Model of Nonalcoholic Fatty Calibration Range: 78.13 to 5000 pg/mL Liver Disease. Nawata A, Noguchi H, Mazaki Y, Kurahashi T, Izumi H, Wang KY, Guo X, Uramoto H, Limit of Detection: 6.77 pg/mL Kohno K, Taniguchi H, Tanaka Y, Fujii J, Sasaguri Y, Tanimoto A, Nakayama T, Yamada S. PLoS One. 2016 Reactivity: Human Apr 1;11(4):e0152549. Applications: Quant (See our web site product page for detailed applications information) Protocols: See our web site at http://www.abnova.com/support/protocols.asp or product page for detailed protocols Storage Instruction: Store the kit at 4°C. -

Oxidative Stress Modulates the Expression Pattern of Peroxiredoxin-6 in Peripheral Blood Mononuclear Cells of Asthmatic Patients and Bronchial Epithelial Cells
Allergy Asthma Immunol Res. 2020 May;12(3):523-536 https://doi.org/10.4168/aair.2020.12.3.523 pISSN 2092-7355·eISSN 2092-7363 Original Article Oxidative Stress Modulates the Expression Pattern of Peroxiredoxin-6 in Peripheral Blood Mononuclear Cells of Asthmatic Patients and Bronchial Epithelial Cells Hyun Jae Shim ,1 So-Young Park ,2 Hyouk-Soo Kwon ,1 Woo-Jung Song ,1 Tae-Bum Kim ,1 Keun-Ai Moon ,1 Jun-Pyo Choi ,1 Sin-Jeong Kim ,1 You Sook Cho 1* 1Division of Allergy and Clinical Immunology, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea 2Division of Pulmonary, Allergy and Critical Care Medicine, Department of Internal Medicine, Konkuk University Medical Center, Seoul, Korea Received: Jul 8, 2019 ABSTRACT Revised: Nov 29, 2019 Accepted: Dec 23, 2019 Purpose: Reduction-oxidation reaction homeostasis is vital for regulating inflammatory Correspondence to conditions and its dysregulation may affect the pathogenesis of chronic airway inflammatory You Sook Cho, MD, PhD diseases such as asthma. Peroxiredoxin-6, an important intracellular anti-oxidant molecule, Division of Allergy and Clinical Immunology, Department of Internal Medicine, Asan is reported to be highly expressed in the airways and lungs. The aim of this study was to Medical Center, University of Ulsan College of analyze the expression pattern of peroxiredoxin-6 in the peripheral blood mononuclear cells Medicine, 88 Olympic-ro 43-gil, Songpa-gu, (PBMCs) of asthmatic patients and in bronchial epithelial cells (BECs). Seoul 05505, Korea. Methods: The expression levels and modifications of peroxiredoxin-6 were evaluated in Tel: +82-2-3010-3285 PBMCs from 22 asthmatic patients. -

A Machine Learning Tool for Predicting Protein Function Anna
The Woolf Classifier Building Pipeline: A Machine Learning Tool for Predicting Protein Function Anna Farrell-Sherman Submitted in Partial Fulfillment of the Prerequisite for Honors in Computer Science under the advisement of Eni Mustafaraj and Vanja Klepac-Ceraj May 2019 © 2019 Anna Farrell-Sherman Abstract Proteins are the machinery that allow cells to grow, reproduce, communicate, and create multicellular organisms. For all of their importance, scientists still have a hard time understanding the function of a protein based on its sequence alone. For my honors thesis in computer science, I created a machine learning tool that can predict the function of a protein based solely on its amino acid sequence. My tool gives scientists a structure in which to build a � Nearest Neighbor or random forest classifier to distinguish between proteins that can and cannot perform a given function. Using default Min-Max scaling, and the Matthews Correlation Coefficient for accuracy assessment, the Woolf Pipeline is built with simplified choices to guide users to success. i Acknowledgments There are so many people who made this thesis possible. First, thank you to my wonderful advisors, Eni and Vanja, who never gave up on me, and always pushed me to try my hardest. Thank you also to the other members of my committee, Sohie Lee, Shikha Singh, and Rosanna Hertz for supporting me through this process. To Kevin, Sophie R, Sophie E, and my sister Phoebe, thank you for reading my drafts, and advising me when the going got tough. To all my wonderful friends, who were always there with encouraging words, warm hugs, and many congratulations, I cannot thank you enough. -
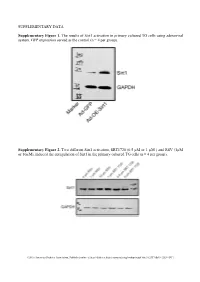
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

The Prognostic Values of the Peroxiredoxins Family in Ovarian Cancer
Bioscience Reports (2018) 38 BSR20180667 https://doi.org/10.1042/BSR20180667 Research Article The prognostic values of the peroxiredoxins family in ovarian cancer Saisai Li, Xiaoli Hu, Miaomiao Ye and Xueqiong Zhu Department of Obstetrics and Gynecology, the Second Affiliated Hospital of Wenzhou Medical University, Wenzhou 325027, Zhejiang, China Correspondence: Xueqiong Zhu ([email protected]) Purpose: Peroxiredoxins (PRDXs) are a family of antioxidant enzymes with six identified mammalian isoforms (PRDX1–6). PRDX expression is up-regulated in various types of solid tumors; however, individual PRDX expression, and its impact on prognostic value in ovarian cancer patients, remains unclear. Methods: PRDXs family protein expression profiles in normal ovarian tissues and ovarian cancer tissues were examined using the Human Protein Atlas database. Then, the prog- nostic roles of PRDX family members in several sets of clinical data (histology, pathological grades, clinical stages, and applied chemotherapy) in ovarian cancer patients were investi- gated using the Kaplan–Meier plotter. Results: PRDXs family protein expression in ovarian cancer tissues was elevated com- pared with normal ovarian tissues. Meanwhile, elevated expression of PRDX3, PRDX5, and PRDX6 mRNAs showed poorer overall survival (OS); PRDX5 and PRDX6 also predicted poor progression-free survival (PFS) for ovarian cancer patients. Furthermore, PRDX3 played sig- nificant prognostic roles, particularly in poor differentiation and late-stage serous ovarian cancer patients. Additionally, PRDX5 predicted a lower PFS in all ovarian cancer patients treated with Platin, Taxol, and Taxol+Platin chemotherapy. PRDX3 and PRDX6 also showed poor PFS in patients treated with Platin chemotherapy. Furthermore, PRDX3 and PRDX5 indicated lower OS in patients treated with these three chemotherapeutic agents. -

Role of Peroxiredoxin 6 in Human Melanoma
Role of Peroxiredoxin 6 in human melanoma Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen Julius-Maximilians-Universität Würzburg vorgelegt von Alexandra Schmitt aus Würzburg Würzburg 2015 Eingereicht am:__________________________ Mitglieder der Promotionskommission: Vorsitzender:____________________________ Gutachter:______________________________ Gutachter:______________________________ Tag des Promotionskolloquiums:___________________ Doktorurkunde ausgehändigt am:___________________ Eidesstattliche Erklärung Gemäß §4, Abs. 3, Ziff. 3, 5 und 8 der Promotionsordnung der Fakultät für Biologie der Bayerischen Julius-Maximilians-Universität Würzburg Hiermit erkläre ich ehrenwörtlich, dass ich die vorliegende Dissertation selbständig angefertigt und keine anderen als die angegebenen Quellen und Hilfsmittel verwendet habe. Ich erkläre weiterhin, dass die vorliegende Dissertation weder in gleicher, noch in ähnlicher Form bereits in einem anderen Prüfungsverfahren vorgelegen hat. Weiterhin erkläre ich, dass ich außer den mit dem Zulassungsantrag urkundlich vorgelegten Graden keine weiteren akademischen Grade erworben oder zu erwerben versucht habe. Würzburg, Januar 2015 ______________________________________ Alexandra Schmitt Table of contents 1. Abstract ................................................................................................................. 1 2. Zusammenfassung ............................................................................................... 3 3. Introduction .......................................................................................................... -

Thiol Peroxidases Mediate Specific Genome-Wide Regulation of Gene Expression in Response to Hydrogen Peroxide
Thiol peroxidases mediate specific genome-wide regulation of gene expression in response to hydrogen peroxide Dmitri E. Fomenkoa,1,2, Ahmet Koca,1, Natalia Agishevaa, Michael Jacobsena,b, Alaattin Kayaa,c, Mikalai Malinouskia,c, Julian C. Rutherfordd, Kam-Leung Siue, Dong-Yan Jine, Dennis R. Winged, and Vadim N. Gladysheva,c,2 aDepartment of Biochemistry, University of Nebraska, Lincoln, NE 68588-0664; bDepartment of Life Sciences, Wayne State College, Wayne, NE 68787; dDepartment of Medicine, University of Utah Health Sciences Center, Salt Lake City, UT 84132; eDepartment of Biochemistry, University of Hong Kong, Hong Kong, China; and cDivision of Genetics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA 02115 Edited by Joan Selverstone Valentine, University of California, Los Angeles, CA, and approved December 22, 2010 (received for review July 21, 2010) Hydrogen peroxide is thought to regulate cellular processes by and could withstand significant oxidative stress. It responded to direct oxidation of numerous cellular proteins, whereas antioxi- several redox stimuli by robust transcriptional reprogramming. dants, most notably thiol peroxidases, are thought to reduce However, it was unable to transcriptionally respond to hydrogen peroxides and inhibit H2O2 response. However, thiol peroxidases peroxide. The data suggested that thiol peroxidases transfer have also been implicated in activation of transcription factors oxidative signals from peroxides to target proteins, thus activating and signaling. It remains unclear if these enzymes stimulate or various transcriptional programs. This study revealed a previously inhibit redox regulation and whether this regulation is widespread undescribed function of these proteins, in addition to their roles or limited to a few cellular components.