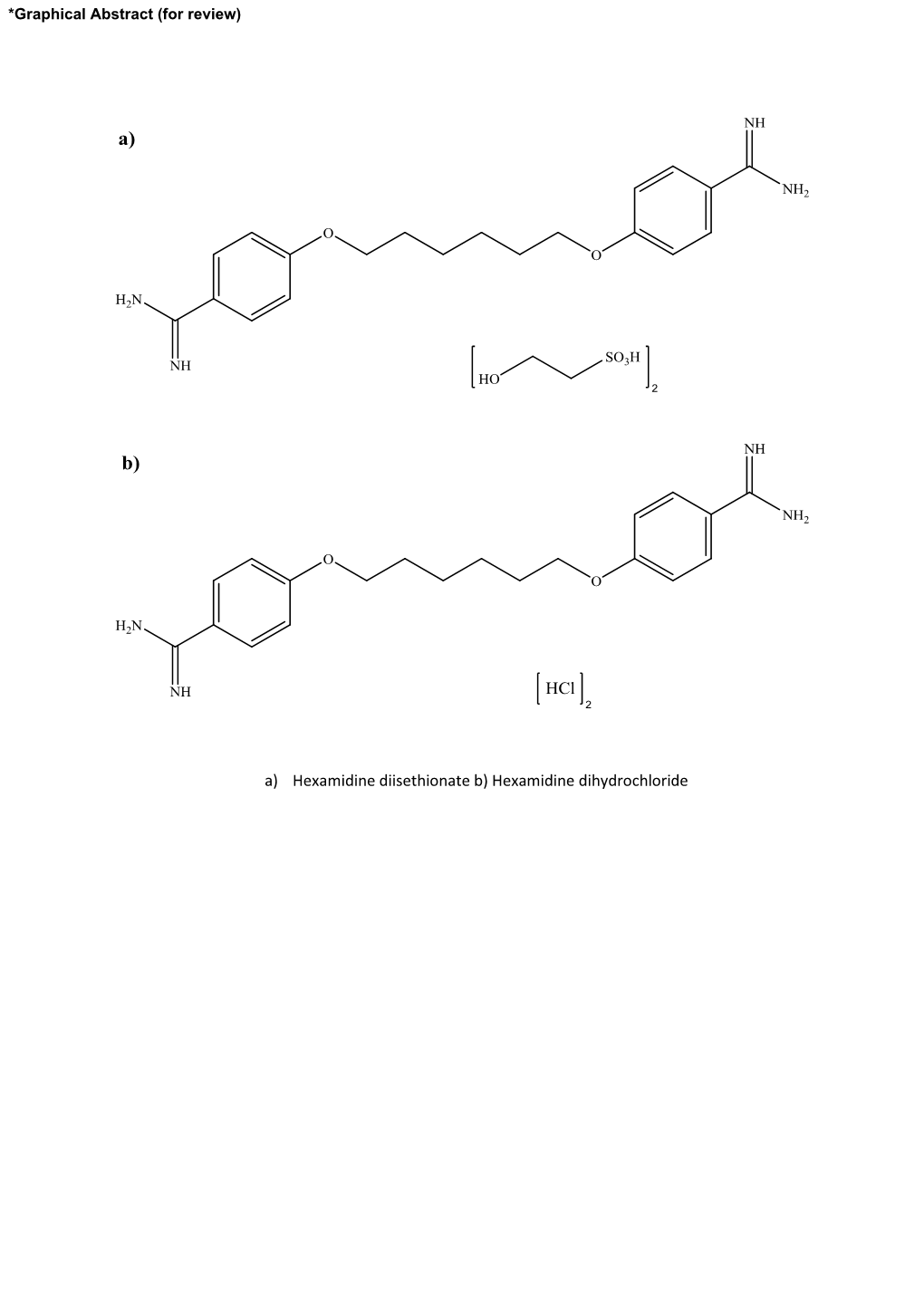
Ethionate B) Hexamidine Dihydrochloride Table(S)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

Genetic Basis of Resistance to Quaternary Ammonium Compounds – the Qac Genes and Their Role: a Review
Veterinarni Medicina, 57, 2012 (6): 275–281 Review Article Genetic basis of resistance to quaternary ammonium compounds – the qac genes and their role: a review Z. Jaglic, D. Cervinkova Veterinary Research Institute, Brno, Czech Republic ABSTRACT: Although the qac genes are named after one of their main substrates (i.e., quaternary ammonium compounds), these genes also code for resistance to a broad spectrum of other cationic compounds such as inter- calating dyes, diamidines and biguanides. The various Qac proteins are involved in relatively low specific efflux- based multidrug pumps and belong to a family of small multidrug resistance proteins. Even though the practical significance of qac-mediated resistance lies mainly in resistance to antiseptics, contradictory findings on this issue are still reported. Therefore, the aim of this review is to summarise the current knowledge on qac-mediated resistance with special emphasis on resistance to antiseptics and its relevance for practice. Keywords: antimicrobial; disinfectant; biocide; benzalkonium; chlorhexidine; cation; susceptibility Contents 3.2. Substrates of qac-mediated resistance 1. Introduction 3.3. Adaptive response to antiseptics 2. The qac genes and their distribution 4. The qac genes and resistance to antibiotics 3. Qac-mediated resistance 5. Conclusions 3.1. Mechanisms of qac-mediated resistance 6. References 1. Introduction first described genetic determinant of resistance to antiseptics was the qacA gene found on pSK1 Resistance to intercalating dyes (i.e. acriflavine and β-lactamase/heavy metal resistance plasmids and ethidium) was associated with particular ge- (Gillespie et al. 1986; Lyon and Skurray 1987). A netic elements, namely staphylococcal β-lactamase range of other qac genes linked with particular plas- plasmids, already more than 40 years ago (Ericson mids (Littlejohn et al. -

ANNEX VI List of Preservatives Allowed for Use in Cosmetic Products
ANNEX VI List of preservatives allowed for use in cosmetic products ANNEX VI LIST OF PRESERVATIVES WHICH COSMETIC PRODUCTS MAY CONTAIN Preamble 1. Preservatives are substances which may be added to cosmetic products for the primary purpose of inhibiting the development of micro-organisms in such products. 2. The substances marked with the symbol (+) may also be added to cosmetic products in concentration other than those laid down in this ANNEX for other purposes apparent from the presentation of the products, e.g. as deodorants in soaps or as anti-dandruff agents in shampoos. 3. Other substances used in the formulation of cosmetic products may also have anti-microbial properties and thus help in the preservation of the products, as, for instance, many essential oils and some alcohols. These substances are not included in the ANNEX. 4. For the purposes of this list - “Salts” is taken to mean: salts of the cations sodium, potassium, calcium, magnesium, ammonium, and ethanolamines; salts of the anions chloride, bromide, sulphate, acetate. - “Esters” is taken to mean: esters of methyl, ethyl, propyl, isopropyl, butyl, isobutyl, phenyl. 5. All finished products containing formaldehyde or substances in this ANNEX and which release formaldehyde must be labelled with the warning “contains formaldehyde” where the concentration of formaldehyde in the finished product exceeds 0.05%. Revised as per August 2015 ASEAN Cosmetic Documents 1 Annex VI – Part 1 – List of preservatives allowed for use in cosmetic products ANNEX VI – PART 1 LIST OF PRESERVATIVES ALLOWED Reference Substance Maximum authorized Limitations and Conditions of use and Number concentration requirements warnings which must be printed on the label a b c d e 1 Benzoic acid (CAS No. -
![Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set](https://docslib.b-cdn.net/cover/8870/ehealth-dsi-ehdsi-v2-2-2-or-ehealth-dsi-master-value-set-1028870.webp)
Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set
MTC eHealth DSI [eHDSI v2.2.2-OR] eHealth DSI – Master Value Set Catalogue Responsible : eHDSI Solution Provider PublishDate : Wed Nov 08 16:16:10 CET 2017 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 1 of 490 MTC Table of Contents epSOSActiveIngredient 4 epSOSAdministrativeGender 148 epSOSAdverseEventType 149 epSOSAllergenNoDrugs 150 epSOSBloodGroup 155 epSOSBloodPressure 156 epSOSCodeNoMedication 157 epSOSCodeProb 158 epSOSConfidentiality 159 epSOSCountry 160 epSOSDisplayLabel 167 epSOSDocumentCode 170 epSOSDoseForm 171 epSOSHealthcareProfessionalRoles 184 epSOSIllnessesandDisorders 186 epSOSLanguage 448 epSOSMedicalDevices 458 epSOSNullFavor 461 epSOSPackage 462 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 2 of 490 MTC epSOSPersonalRelationship 464 epSOSPregnancyInformation 466 epSOSProcedures 467 epSOSReactionAllergy 470 epSOSResolutionOutcome 472 epSOSRoleClass 473 epSOSRouteofAdministration 474 epSOSSections 477 epSOSSeverity 478 epSOSSocialHistory 479 epSOSStatusCode 480 epSOSSubstitutionCode 481 epSOSTelecomAddress 482 epSOSTimingEvent 483 epSOSUnits 484 epSOSUnknownInformation 487 epSOSVaccine 488 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 3 of 490 MTC epSOSActiveIngredient epSOSActiveIngredient Value Set ID 1.3.6.1.4.1.12559.11.10.1.3.1.42.24 TRANSLATIONS Code System ID Code System Version Concept Code Description (FSN) 2.16.840.1.113883.6.73 2017-01 A ALIMENTARY TRACT AND METABOLISM 2.16.840.1.113883.6.73 2017-01 -

LIST of REGISTRED DRUGS in ARMENIA (Up to 31.12.2017)
LIST OF REGISTRED DRUGS IN ARMENIA (Up to 31.12.2017) International nonproprietary Registration Term of Legal status for N Trade name name (generic) or Drug form Dose and packaging Manufacturer Country ATC1 code License holder number registration supply active ingredients name Lek Pharmaceuticals pefloxacin Lek Pharmaceuticals 400mg, 10.03.2015 d.d., Verovskova Str. 1 Abaktal (pefloxacin tablets film-coated d.d., Verovskova Str. Slovenia J01MA03 14308 PoM2 in blister (10/1x10/) 10.03.2020 57, 1526 Ljubljana, mesylate dihydrate) 57, 1526 Ljubljana Slovenia Lilly France S.A.S., Eli Lilly Regional 100IU/ml, Zone Industrielle, 2 17.03.2017 Operations GmbH., 2 Abasaglar insulin glargine solution for injection France A10AE04 16535 PoM 3ml cartridges (5) rue du Colonel Lilly, 17.03.2022 Koelblgasse 8-10, 67640 Fegersheim 1030, Vienna, Austria Help S.A ,10 ambroxol Help S.A. Pedini, Valaoritou str., GR 6mg/ml, 09.03.2016 3 Abrobion (ambroxol syrup Ioanninon, Ioannina, Greece R05CB06 15404 OTC3 144 52, 125ml glass bottle 09.03.2021 hydrochloride) 45500 Metamorphosis, Attika, Greece Salutas Pharma GmbH, Otto-von- Guericke-Alle-1, 100mg/5ml, 39179 Barleben-batch Sandoz 30g powder in 75ml glass powder for oral releaser, Allphamed Pharmaceuticals d.d., bottle and measuring 20.10.2015 4 ACC acetylcysteine solution with orange Pharbil Arzneimittel Germany R05CB01 14947 OTC Verovskova Str. 57, spoon 5ml, 60g powder in 20.10.2020 flavour GmbH Hildebrandstr. 1000 Ljubljana, 150ml glass bottle and 12, 37081 Gottingen, Slovenia measuring spoon 5ml Germany-bulk manufacturer, packager 1 Salutas Pharma GmbH, Otto-von- Guericke-Alle-1, 39179 Barleben - Sandoz batch releaser Pharmaceuticals d.d., 100mg, 21.11.2014 5 ACC 100 acetylcysteine tablets effervescent (Hermes Pharma Germany R05CB01 13967 OTC Verovskova Str. -

CVS Store Brand Restricted Substances List
CVS Store Brand Restricted Substances List The following ingredients are restricted from select CVS Store Brand Baby & Child, Beauty, Personal Care, Foods, Suncare, Household, and Bottled Spring Water Products. This list is updated annually, in May. Baby & Child Products (non-medicated products, e.g., shampoos, lotions, wipes) California Proposition 65 Banned Chemicals (e.g., Toluene, Multiple Styrene, BPA) ii Formaldehyde 50-00-0 Dimethyl-dimethyl (DMDM) Hydantoin 6440-58-0 Imidazolidinyl Urea 39236-46-9 Diazolidinyl Urea 78491-02-8 Sodium Hydroxymethylglycinate 70161-44-3 Formaldehyde 2-bromo-2-nitropropane-1,3-diol (Bronopol) 52-51-7 and donors Quaternium-15 4080-31-3 Glyoxal 107-22-2 Polyoxymethylene Urea 68611-64-3 5-Bromo-5-Nitro-1,3 Dioxane (Bronidox) 30007-47-7 Methylene glycol (methanediol) 463-57-0 Methenamine 100-97-0 Ethylparaben 120-47-8 Butylparaben 94-26-8 Methylparaben 99-76-3 Propylparaben 94-13-3 Heptylparaben 1085-12-7 Isobutylparaben 4247-02-3 Isopropylparaben 4191-73-5 Parabens Benzylparaben 94-18-8 Octylparaben 1219-38-1 Pentylparaben 6521-29-5 Phenylparaben 17696-62-7 Isodecylparaben 2664-60-0 Calcium Paraben 69959-44-0 Potassium Paraben 16782-08-4 5026-62-0 35285-69-9 Sodium Parabens 35285-68-8 36457-20-2 Hexamidine Paraben Not Found Hexamidine Diparaben 93841-83-9 Undecylenoyl PEG 5 Paraben Not Found Phenoxyethylparaben 55468-88-7 4-Hydroxybenzoic acid 99-96-7 Di-2-ethylhexyl phthalate (DEHP) 117-81-7 Benzyl butyl phthalate (BBP) 85-68-7 Di-n-butyl phthalate (DBP) 84-74-2 Diisodecyl phthalate (DIDP) 26761-40-0 -

Vr Meds Ex01 3B 0825S Coding Manual Supplement Page 1
vr_meds_ex01_3b_0825s Coding Manual Supplement MEDNAME OTHER_CODE ATC_CODE SYSTEM THER_GP PHRM_GP CHEM_GP SODIUM FLUORIDE A12CD01 A01AA01 A A01 A01A A01AA SODIUM MONOFLUOROPHOSPHATE A12CD02 A01AA02 A A01 A01A A01AA HYDROGEN PEROXIDE D08AX01 A01AB02 A A01 A01A A01AB HYDROGEN PEROXIDE S02AA06 A01AB02 A A01 A01A A01AB CHLORHEXIDINE B05CA02 A01AB03 A A01 A01A A01AB CHLORHEXIDINE D08AC02 A01AB03 A A01 A01A A01AB CHLORHEXIDINE D09AA12 A01AB03 A A01 A01A A01AB CHLORHEXIDINE R02AA05 A01AB03 A A01 A01A A01AB CHLORHEXIDINE S01AX09 A01AB03 A A01 A01A A01AB CHLORHEXIDINE S02AA09 A01AB03 A A01 A01A A01AB CHLORHEXIDINE S03AA04 A01AB03 A A01 A01A A01AB AMPHOTERICIN B A07AA07 A01AB04 A A01 A01A A01AB AMPHOTERICIN B G01AA03 A01AB04 A A01 A01A A01AB AMPHOTERICIN B J02AA01 A01AB04 A A01 A01A A01AB POLYNOXYLIN D01AE05 A01AB05 A A01 A01A A01AB OXYQUINOLINE D08AH03 A01AB07 A A01 A01A A01AB OXYQUINOLINE G01AC30 A01AB07 A A01 A01A A01AB OXYQUINOLINE R02AA14 A01AB07 A A01 A01A A01AB NEOMYCIN A07AA01 A01AB08 A A01 A01A A01AB NEOMYCIN B05CA09 A01AB08 A A01 A01A A01AB NEOMYCIN D06AX04 A01AB08 A A01 A01A A01AB NEOMYCIN J01GB05 A01AB08 A A01 A01A A01AB NEOMYCIN R02AB01 A01AB08 A A01 A01A A01AB NEOMYCIN S01AA03 A01AB08 A A01 A01A A01AB NEOMYCIN S02AA07 A01AB08 A A01 A01A A01AB NEOMYCIN S03AA01 A01AB08 A A01 A01A A01AB MICONAZOLE A07AC01 A01AB09 A A01 A01A A01AB MICONAZOLE D01AC02 A01AB09 A A01 A01A A01AB MICONAZOLE G01AF04 A01AB09 A A01 A01A A01AB MICONAZOLE J02AB01 A01AB09 A A01 A01A A01AB MICONAZOLE S02AA13 A01AB09 A A01 A01A A01AB NATAMYCIN A07AA03 A01AB10 A A01 -

Analyse Des Consommations 2008 De Médicaments
Observatoire des Médicaments, des Dispositifs médicaux et des Innovations Thérapeutiques Bilan de la collecte 2008 AAnnaallyyssee ddeess ccoonnssoommmmaattiioonnss ddee mmééddiiccaammeennttss ddaannss lleess ééttaabblliisssseemmeennttss ppuubblliiccss eett pprriivvééss MMCCOO** ddee llaa rrééggiioonn Centre * hors HAD et Centres de dialyse Coordination de l’OMéDIT de la région Centre A. Rouleau - MC. Lanoue SOMMAIRE page INTRODUCTION 1 ANALYSE GLOBALE DES CONSOMMATIONS (hors rétrocessions) 5 Consommations en valeur 5 Consommations en unités délivrées 6 Consommations par type d’établissement 7 ANALYSE DES CONSOMMATIONS PAR CLASSE THÉRAPEUTIQUE 9 Classe A Voies digestives et métabolisme 14 Classe B Sang et organes hématopoïétiques 16 B01 Anti thrombotiques 18 B05 Substituts du sang et solutés de perfusion 19 Classe C Système cardiovasculaire 20 Classe D Médicaments dermatologiques 21 Classe G Système génito-urinaire et hormones sexuelles 22 Classe H Hormones systémiques - hormones sexuelles exclues 23 Classe J Anti-infectieux généraux et immunomodulateurs 24 J01 Antibactériens à usage systémique 24 J02 Antimycosiques à usage systémique 27 J06 Immunsérums et immunoglobulines 36 J05 Antiviraux à usage systémique 39 J07 Vaccins 39 Classe L Antinéoplasiques et immunomodulateurs 40 L01 Antinéoplasiques 40 L04 Immunosuppresseurs 46 Classe M Muscle et squelette 48 Classe N Système nerveux 50 Classe P Antiparasitaires et insecticides 53 Classe R Système respiratoire 54 Classe S Organes sensoriels 55 Classe V Divers 56 LES RÉTROCESSIONS 59 UTILISATION DES DONNÉES 64 IINNTTRROODDUUCCTTIIOONN La nécessité d’une extraction automatisée des données de consommations à partir des logiciels de gestion s'est imposée dès 2002 comme source d’information, d’évaluation et de pilotage pour les actions menées par l’OMéDIT de la région Centre. -

LIST of REGISTRED OTC DRUGS in ARMENIA (Up to 31.03.2017)
LIST OF REGISTRED OTC DRUGS IN ARMENIA (Up to 31.03.2017) International nonproprietary Registration Term of Legal status for N Trade name name (generic) or Drug form Dose and packaging Manufacturer Country ATC1 code License holder number registration supply active ingredients name Help S.A ,10 ambroxol Help S.A. Pedini, Valaoritou str., GR 6mg/ml, 09.03.2016 1 Abrobion (ambroxol syrup Ioanninon, Ioannina, Greece R05CB06 15404 OTC 144 52, 125ml glass bottle 09.03.2021 hydrochloride) 45500 Metamorphosis, Attika, Greece Salutas Pharma GmbH, Otto-von- Guericke-Alle-1, 100mg/5ml, 39179 Barleben-batch Sandoz 30g powder in 75ml glass powder for oral releaser, Allphamed Pharmaceuticals d.d., bottle and measuring 20.10.2015 2 ACC acetylcysteine solution with orange Pharbil Arzneimittel Germany R05CB01 14947 OTC Verovskova Str. 57, spoon 5ml, 60g powder in 20.10.2020 flavour GmbH Hildebrandstr. 1000 Ljubljana, 150ml glass bottle and 12, 37081 Gottingen, Slovenia measuring spoon 5ml Germany-bulk manufacturer, packager Salutas Pharma GmbH, Otto-von- Guericke-Alle-1, 39179 Barleben - Sandoz batch releaser Pharmaceuticals d.d., 100mg, 21.11.2014 3 ACC 100 acetylcysteine tablets effervescent (Hermes Pharma Germany R05CB01 13967 OTC Verovskova Str. 57, in plastic container (20) 21.11.2019 G.m.b.H., Allgau 36, 1526 Ljubljana, A-9400 Wolsberg, Slovenia Germany - bulk manufacturer, packager) 1 Salutas Pharma GmbH, Otto-von- Guericke-Alle-1, Sandoz 39179 Barleben-batch Pharmaceuticals d.d., powder for oral 100mg, releaser, Lindopharm 14.04.2015 4 ACC 100 acetylcysteine Germany R05CB01 14430 OTC Verovskova Str. 57, solution 3g sachets (20) GmbH, Neustrasse 82, 14.04.2020 1000 Ljubljana, D-40721 Hilden, Slovenia Germany- bulk manufacturer, packager Salutas Pharma GmbH, Otto-von- Guericke-Alle-1, 39179 Barleben - Sandoz batch releaser Pharmaceuticals d.d., 200mg, 21.11.2014 5 ACC 200 acetylcysteine tablets effervescent (Hermes Pharma Germany R05CB01 13968 OTC Verovskova Str. -

Antiseptics and Disinfectants: Activity, Action, and Resistance
CLINICAL MICROBIOLOGY REVIEWS, Jan. 1999, p. 147–179 Vol. 12, No. 1 0893-8512/99/$04.0010 Copyright © 1999, American Society for Microbiology. All Rights Reserved. Antiseptics and Disinfectants: Activity, Action, and Resistance 1 2 GERALD MCDONNELL * AND A. DENVER RUSSELL STERIS Corporation, St. Louis Operations, St. Louis, Missouri 63166,1 and Welsh School of Pharmacy, Cardiff University, Cardiff CF1 3XF, United Kingdom2 INTRODUCTION .......................................................................................................................................................148 DEFINITIONS ............................................................................................................................................................148 MECHANISMS OF ACTION ...................................................................................................................................148 Introduction.............................................................................................................................................................148 General Methodology .............................................................................................................................................148 Alcohols ....................................................................................................................................................................151 Aldehydes .................................................................................................................................................................151 -

Consultation Paper: International Harmonisation of Ingredient Names
International Harmonisation of Ingredient Names (IHIN) Consultation paper Version 1.0, May 2013 Historical consultation document Therapeutic Goods Administration About the Therapeutic Goods Administration (TGA) · The Therapeutic Goods Administration (TGA) is part of the Australian Government Department of Health and Ageing, and is responsible for regulating medicines and medical devices. · The TGA administers the Therapeutic Goods Act 1989 (the Act), applying a risk management approach designed to ensure therapeutic goods supplied in Australia meet acceptable standards of quality, safety and efficacy (performance), when necessary. · The work of the TGA is based on applying scientific and clinical expertise to decision- making, to ensure that the benefits to consumers outweigh any risks associated with the use of medicines and medical devices. · The TGA relies on the public, healthcare professionals and industry to report problems with medicines or medical devices. TGA investigates reports received by it to determine any necessary regulatory action. · To report a problem with a medicine or medical device, please see the information on the TGA website <www.tga.gov.au>. Copyright © Commonwealth of Australia 2013 This work is copyright. You may reproduce the whole or part of this work in unaltered form for your own personal use or, if you are part of an organisation, for internal use within your organisation, but only if you or your organisation do not use the reproduction for any commercial purpose and retain this copyright notice and all disclaimer notices as part of that reproduction. Apart from rights to use as permitted by the Copyright Act 1968 or allowed by this copyright notice, all other rights are reserved and you are not allowed to reproduce the whole or any part of this work in any way (electronic or otherwise) without first being given specific written permission from the Commonwealth to do so. -

A New Ophthalmic Formulation Containing Antiseptics and Dexpanthenol: in Vitro Antimicrobial Activity and Effects on Corneal and Conjunctival Epithelial Cells
Experimental Eye Research 201 (2020) 108269 Contents lists available at ScienceDirect Experimental Eye Research journal homepage: www.elsevier.com/locate/yexer Research article A new ophthalmic formulation containing antiseptics and dexpanthenol: In vitro antimicrobial activity and effects on corneal and conjunctival epithelial cells Rita Mencucci a,*,1, Eleonora Favuzza a,1, Paolo Bottino b, Costanza Mazzantini c, Elisa Zanotto b, Domenico E. Pellegrini-Giampietro c, Elisa Landucci c a Eye Clinic, Department of Neurosciences, Psychology, Pharmacology and Child Health (NEUROFARBA), University of Florence, Largo Brambilla 3, 50134, Florence, Italy b AOU Citta` della Salute e della Scienza di Torino, Microbiology and Virology Unit, Turin, Italy c Department of Health Sciences, Section of Clinical Pharmacology and Oncology, University of Florence, Viale Pieraccini 6, 50139, Florence, Italy ARTICLE INFO ABSTRACT Keywords: Antibiotic resistance is increasing even in ocular pathogens, therefore the interest towards antiseptics in Antiseptics Ophthalmology is growing. The aim of this study was to analyze the in vitro antimicrobial efficacy and the in Hexamidine vitro effects of an ophthalmic formulation containing hexamidine diisethionate 0.05%, polyhexamethylene Dexpanthenol biguanide (PHMB) 0.0001% disodium edetate (EDTA) 0.01%, dexpanthenol 5% and polyvinyl alcohol 1.25% Polyhexamethylene biguanide (Keratosept, Bruschettini, Genova, Italy) on cultured human corneal and conjunctival cells. The in vitro anti PHMB EDTA microbial activity was tested on Staphylococcus aureus, Methicillin-Resistant Staphylococcus aureus (MRSA), Corneal epithelial cells Pseudomonas aeruginosa, Streptococcus pneumoniae, Streptococcus pyogenes and Streptococcus mitis. For each mi ◦ Conjunctival epithelial cells crobial strain 10 μL of a 0.5 MacFarland standardized bacterial inoculum were incubated at 25 C with 100 μL of ophthalmic solution for up to 6 h.