RSC Advances REVIEW
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
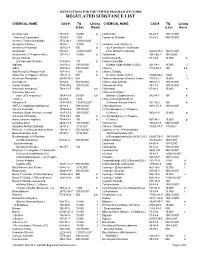
Regulated Substance List
INSTRUCTIONS FOR THE UNIFIED PROGRAM (UP) FORM REGULATED SUBSTANCE LIST CHEMICAL NAME CAS # TQ Listing CHEMICAL NAME CAS # TQ Listing (Lbs) Basis (Lbs) Basis Acetaldehyde 75-07-0 10,000 g Cantharidin 56-25-7 100/10,0001 * Acetone Cyanohydrin 75-86-5 1,000 Carbachol Chloride 51-83-2 500/10,0001 Acetone Thiosemicarbazide 1752-30-3 1,000/10,0001 Acetylene (Ethyne) 74-86-2 10,000 f Carbamic Acid, Methyl-,o- Acrolein (2-Propenal) 107-02-8 500 b (((2,4-Dimethyl-1,3-Dithiolan- Acrylamide 79-06-1 1,000/10,0001 2-YL) Methylene)Amino)- 26419-73-8 100/10,0001 Acrylonitrile (2- Propenenitrile) 107-13-1 10,000 b Carbofuran 1563-66-2 10/10,0001 Acrylyl Chloride Carbon Disulfide 75-15-0 10,000 b (2-Propenoyl Chloride) 814-68-6 100 b Carbon Oxysulfide Aldicarb 116-06-3 100/10,0001 (Carbon Oxide Sulfide (COS)) 463-58-1 10,000 f Aldrin 309-00-2 500/10,0001 Chlorine 7782-50-5 100 a,b Allyl Alcohol (2-Propen-1-ol) 107-18-6 1,000 b Chlorine Dioxide Allylamine (2-Propen-1-Amine) 107-11-9 500 b (Chlorine Oxide (ClO2)) 10049-04-4 1,000 c Aluminum Phosphide 20859-73-8 500 Chlorine Monoxide (Chlorine Oxide) 7791-21-1 10,000 f Aminopterin 54-62-6 500/10,0001 Chlormequat Chloride 999-81-5 100/10,0001 Amiton Oxalate 3734-97-2 100/10,0001 Chloroacetic Acid 79-11-8 100/10,0001 Ammonia, Anhydrous 2 7664-41-7 500 a,b Chloroform 67-66-3 10,000 b Ammonia, Aqueous Chloromethyl Ether (conc 20% or greater) 7664-41-7 20,000 a,b (Methane,Oxybis(chloro-) 542-88-1 100 b * Aniline 62-53-3 1,000 Chloromethyl Methyl Ether Antimycin A 1397-94-0 1,000/10,0001 (Chloromethoxymethane) -
Capsicum Oleoresin and Homocapsaicin
Printed on: Wed Jan 06 2021, 02:44:36 AM Official Status: Currently Official on 06-Jan-2021 DocId: 1_GUID-1560FD9B-BE0F-495E-9994-C5718733DB4C_2_en-US (EST) Printed by: Jinjiang Yang Official Date: Official as of 01-May-2019 Document Type: USP @2021 USPC 1 nordihydrocapsaicin, nonivamide, decanylvanillinamide, Capsicum Oleoresin and homocapsaicin. DEFINITION ASSAY Capsicum Oleoresin is an alcoholic extract of the dried ripe · CONTENT OF TOTAL CAPSAICINOIDS fruits of Capsicum. It contains NLT 6.5% of total Mobile phase: A mixture of acetonitrile and diluted capsaicinoids, calculated as the sum of capsaicin, phosphoric acid (1 in 1000) (2:3) dihydrocapsaicin, nordihydrocapsaicin, nonivamide, Standard solution A: 0.2 mg/mL of USP Capsaicin RS in decanylvanillinamide, and homocapsaicin, all calculated on methanol the anhydrous basis. The nonivamide content is NMT 5% of Standard solution B: 0.1 mg/mL of USP the total capsaicinoids, calculated on the anhydrous basis. Dihydrocapsaicin RS in methanol [CAUTIONÐCapsicum Oleoresin is a powerful irritant, and Sample solution: 5 mg/mL of Capsicum Oleoresin in even in minute quantities produces an intense burning methanol. Pass a portion of this solution through a filter of sensation when it comes in contact with the eyes and 0.2-µm pore size, and use the filtrate as the Sample solution. tender parts of the skin. Care should be taken to protect Chromatographic system the eyes and to prevent contact of the skin with (See Chromatography á621ñ, System Suitability.) Capsicum Oleoresin.] Mode: LC IDENTIFICATION -

Proposed Industrial Powered, Air-Purifying Respirator (PAPR) Standard Draft: November 4, 2005
D R A F T F O R D I S C U S S I O N Concept: Proposed Industrial Powered, Air-Purifying Respirator (PAPR) Standard Draft: November 4, 2005 84.300 Scope 84.301 Definitions 84.302 Description 84.303 Required components 84.304 General construction 84.305 Breathing tubes 84.306 Body harnesses 84.307 Head harness 84.308 Respiratory inlet coverings 84.309 Eyepieces of respiratory inlet coverings 84.310 Inhalation and exhalation valves 84.311 Exhalation valve leakage 84.312 Breathing resistance 84.313 Air pressure determination for maintaining positive pressure 84.314 Air flow determination for testing cartridges, canisters, and/or filters at a constant flow and for machine breathing gas testing 84.315 PAPR100 and PAPR95 particulate filter efficiency level determination 84.316 Chemical cartridge/canister gas/vapor removing effectiveness 84.317 Laboratory Respiratory Protection Level (LRPL) 84.318 Field of view 84.319 Low temperature fogging 84.320 Noise levels 84.321 Breathing gas: Carbon dioxide (CO2) machine test 84.322 Breathing gas: Oxygen (O2) and Carbon dioxide (CO2) human subject-generated 84.323 Low pressure indicator 84.324 Full and low battery power indicator 84.325 Battery life 84.326 End-of-Service-Life (ESLI) criteria 84.327 Service time limitations 84.328 Shelf life limitations 84.329 Intrinsic safety, optional 84.330 Hydration device (drink tube), optional 84.331 Additional container requirements specific to PAPRs 84.332 Additional labeling requirements specific to PAPRs D R A F T F O R D I S C U S S I O N 1 D R A F T F O R D I S C U S S I O N 84.300 Scope: Powered, air-purifying Respirators (PAPRs) shall be approved under this subpart The purpose of Subpart P is to establish procedures and minimum requirements for issuing approvals and extensions of approval specifically to PAPRs. -

Powered Air Purifying Respirator (PAPR) Standard Subpart P
Proposed Concept: Powered Air-Purifying Respirator (PAPR) Standard Subpart P December 21, 2007 1. Scope 2. Definitions 3. Descriptions 4. Base Requirements – All PAPR shall meet the following base requirements: 4.1 Non-respiratory 4.1.1 Required components 4.1.2 General construction 4.1.3 Breathing tubes 4.1.4 Body harnesses 4.1.5 Head harness 4.1.6 Respiratory inlet coverings 4.1.7 Eyepieces/lens of respiratory inlet coverings 4.1.8 Noise levels 4.1.9 Low pressure indicator 4.1.10 Power 4.1.11 Battery life 4.1.12 End of Service Life (ESLI) criteria 4.1.13 Shelf life limitations 4.1.14 Failure Mode and Effects Analysis (FMEA) 4.2 Respiratory 4.2.1 Inhalation and exhalation valves 4.2.2 Exhalation valve leakage 4.2.3 Breathing resistance 4.2.4 Air flow determination 4.2.5 Breathing gas: Carbon dioxide (CO2) machine tests 4.2.6 Service time limitations 4.2.7 Chemical cartridge/canister gas/vapor removal effectiveness 4.2.8 PAPR P95 and PAPR P100 particulate filter efficiency level determination 4.2.9 Breathing gas concentrations determination: Oxygen (O2) and Carbon dioxide (CO2) human subject generated 4.2.10 Laboratory Respirator Protection Level (LRPL) Draft For Discussion Only 5. Enhanced Chemical, Biological, Radiological, Nuclear (CBRN) Requirements - Performance requirements beyond base requirements that may be desired by applicant for approval 5.1 CBRN requirements 5.2 Lower Level Chemical, Biological, Radiological, Nuclear (LCBRN) requirements 6. Additional Enhanced Requirements 6.1 Flammability and Heat Resistance 6.2 Silent Operation 6.3 Operational Temperature Range 6.4 Hydration device (drink tube) 6.5 Intrinsic Safety 2 Draft For Discussion Only 1. -

TRP Mediation
molecules Review Remedia Sternutatoria over the Centuries: TRP Mediation Lujain Aloum 1 , Eman Alefishat 1,2,3 , Janah Shaya 4 and Georg A. Petroianu 1,* 1 Department of Pharmacology, College of Medicine and Health Sciences, Khalifa University of Science and Technology, Abu Dhabi 127788, United Arab Emirates; [email protected] (L.A.); Eman.alefi[email protected] (E.A.) 2 Center for Biotechnology, Khalifa University of Science and Technology, Abu Dhabi 127788, United Arab Emirates 3 Department of Biopharmaceutics and Clinical Pharmacy, Faculty of Pharmacy, The University of Jordan, Amman 11941, Jordan 4 Pre-Medicine Bridge Program, College of Medicine and Health Sciences, Khalifa University of Science and Technology, Abu Dhabi 127788, United Arab Emirates; [email protected] * Correspondence: [email protected]; Tel.: +971-50-413-4525 Abstract: Sneezing (sternutatio) is a poorly understood polysynaptic physiologic reflex phenomenon. Sneezing has exerted a strange fascination on humans throughout history, and induced sneezing was widely used by physicians for therapeutic purposes, on the assumption that sneezing eliminates noxious factors from the body, mainly from the head. The present contribution examines the various mixtures used for inducing sneezes (remedia sternutatoria) over the centuries. The majority of the constituents of the sneeze-inducing remedies are modulators of transient receptor potential (TRP) channels. The TRP channel superfamily consists of large heterogeneous groups of channels that play numerous physiological roles such as thermosensation, chemosensation, osmosensation and mechanosensation. Sneezing is associated with the activation of the wasabi receptor, (TRPA1), typical ligand is allyl isothiocyanate and the hot chili pepper receptor, (TRPV1), typical agonist is capsaicin, in the vagal sensory nerve terminals, activated by noxious stimulants. -

Downloads/DL Praevention/Fachwissen/Gefahrstoffe/TOXIKOLOGI SCHE BEWERTUNGEN/Bewertungen/Toxbew072-L.Pdf
Distribution Agreement In presenting this thesis or dissertation as a partial fulfillment of the requirements for an advanced degree from Emory University, I hereby grant to Emory University and its agents the non-exclusive license to archive, make accessible, and display my thesis or dissertation in whole or in part in all forms of media, now or hereafter known, including display on the world wide web. I understand that I may select some access restrictions as part of the online submission of this thesis or dissertation. I retain all ownership rights to the copyright of the thesis or dissertation. I also retain the right to use in future works (such as articles or books) all or part of this thesis or dissertation. Signature: _____________________________ ______________ Jedidiah Samuel Snyder Date Statistical analysis of concentration-time extrapolation factors for acute inhalation exposures to hazardous substances By Jedidiah S. Snyder Master of Public Health Global Environmental Health _________________________________________ P. Barry Ryan, Ph.D. Committee Chair _________________________________________ Eugene Demchuk, Ph.D. Committee Member _________________________________________ Paige Tolbert, Ph.D. Committee Member Statistical analysis of concentration-time extrapolation factors for acute inhalation exposures to hazardous substances By Jedidiah S. Snyder Bachelor of Science in Engineering, B.S.E. The University of Iowa 2010 Thesis Committee Chair: P. Barry Ryan, Ph.D. An abstract of A thesis submitted to the Faculty of the Rollins School of Public Health of Emory University in partial fulfillment of the requirements for the degree of Master of Public Health in Global Environmental Health 2015 Abstract Statistical analysis of concentration-time extrapolation factors for acute inhalation exposures to hazardous substances By Jedidiah S. -

Pharmacokinetics of Daikenchuto, a Traditional Japanese Medicine (Kampo) After Single Oral Administration to Healthy Japanese Volunteers
DMD Fast Forward. Published on July 1, 2011 as DOI: 10.1124/dmd.111.040097 DMDThis Fast article Forward. has not been Published copyedited and on formatted. July 1, The2011 final as version doi:10.1124/dmd.111.040097 may differ from this version. DMD #040097 Pharmacokinetics of daikenchuto, a traditional Japanese medicine (Kampo) after single oral administration to healthy Japanese volunteers Masaya Munekage, Hiroyuki Kitagawa, Kengo Ichikawa, Junko Watanabe, Katsuyuki Aoki, Toru Kono, Kazuhiro Hanazaki Department of Surgery, Kochi Medical School, Nankoku, Kochi, Japan (M.M., H.K., K.I., K.H); Downloaded from Tsumura Laboratories, TSUMURA & CO., Ami, Ibaraki, Japan (J.W.); Pharmaceutical & Quality Research Department, TSUMURA & CO., Ami, Ibaraki , Japan (K.A.); Division of dmd.aspetjournals.org Gastroenterologic and General Surgery, Department of Surgery, Asahikawa Medical University, Hokkaido, Japan (T.K.). at ASPET Journals on September 26, 2021 1 Copyright 2011 by the American Society for Pharmacology and Experimental Therapeutics. DMD Fast Forward. Published on July 1, 2011 as DOI: 10.1124/dmd.111.040097 This article has not been copyedited and formatted. The final version may differ from this version. DMD #040097 Running title: Pharmacokinetics study of daikenchuto Address correspondence to: Kazuhiro Hanazaki, M.D., Ph.D. Department of Surgery, Kochi Medical School, Oko-cho kohasu, Nankoku-shi, Kochi 783-8505, Japan. E-mail: [email protected] , Phone: 81-88-880-2370, Fax: 81-88-880-2371 Number of text pages: 17 Downloaded from Number of Tables: 1 Number of Figures: 2 dmd.aspetjournals.org Number of References: 17 Number of Words: Abstract: 199 at ASPET Journals on September 26, 2021 Introduction: 377 Results and Discussion: 855 ABBREVIATIONS: TJ-100, daikenchuto; HAS, hydroxy-α-sanshool; HBS, hydroxy-β-sanshool; 6S, [6]-shogaol; 10S, [10]-shogaol; GRB1, ginsenoside Rb1; GRG1, ginsenoside Rg1; HPLC, high-performance liquid chromatography; LC, liquid chromatography; MS, mass spectrometry; MS/MS, tandem mass spectrometry 2 DMD Fast Forward. -

Warfare Agents for Modeling Airborne Dispersion in and Around Buildings
LBNL-45475 ERNEST ORLANDO LAWRENCE BERKELEY NATIn NAL LABORATORY Databaseof Physical,Chemicaland ToxicologicalPropertiesof Chemical and Biological(CB)WarfitreAgentsfor ModelingAirborneDispersionIn and AroundBuildings TracyThatcher,RichSextro,andDonErmak Environmental Energy Technologies Division DISCLAIMER This document was prepared as an account of work sponsored by the United States Government. While this document is believed to contain correct information, neither the United States Government nor any agency thereof, nor The Regents of the University of Catifomia, nor any of their employees, makes any warranty, express or implied, or assumes any legal responsibility for the accuracy, completeness, or usefulness of anY information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process, or service by its trade name, trademark, manufacturer, or otherwise, does not necessarily constitute or imply its endorsement, recommend at i on, or favoring by the United States Government or any agency thereof, or The Regents of the University of California. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof, or The Regents of the University of California. Ernest Orlando Lawrence Berkeley National Laboratory is an equal opportunity employer. DISCLAIMER Portions of this document may be illegible in electronic image products. Images are produced -

Product Nutritional Analysis
Product Nutritional Analysis CA Nutri Chemical Analysis Analysis Unit Price (ex VAT) SANAS Accredited? Lead Time Energy by Calculation kJ or kcal No charge if part of Full Nutri Carbohydrate by Calculation g/100g No charge if part of Full Nutri Moisture g/100g R 193 2 Ash g/100g R 193 3 Protein g/100g R 355 3 Glycaemic Carbohydrates g/100g R 1 649 5 Total Sugars g/100g R 1 088 5 (Glucose, Fructose, Sucrose, Lactose and Maltose) Total Fat by AOAC 996.06 g/100g R 950 7 Full Nutritional Fatty acid Composition by AOAC 996.06 g/100g R 1 087 Yes Label of which Saturated g/100g of which Monounsaturated g/100g of which Polyunsaturated g/100g Included as part of Fatty Acid 7 of which Trans Fatty Acids g/100g Composition Omega 3 mg/100g Omega 6 mg/100g Cholesterol mg/100g R 842 7 Total Dietary Fibre by AOAC 985.29 g/100g R 1 636 8 to 12 Sodium mg/100g R 529 5 Salt calculated from Sodium results g/100g No charge if Sodium requested Yes Salt Salt by chloride titration g/100g R 662 Yes 5 Acid Insoluble Ash g/100g R 390 5 to 7 Crude Fibre g/100g R 1 374 Yes 5 to 7 Water Activity - R 527 7 Other pH - R 150 2 Density g/ml R 133 Yes 3 Caffeine mg/100g R 705 Yes 5 Total Capsaicinoids mg/kg R 665 5 (Capsaicin, dihydrocapsaicin, nordihydrocapsaicin and Scoville Heat Value) Antimony (Sb) mg/kg R 813 Arsenic (As) mg/kg R 813 Subc to SGS 10 Calcium (Ca) mg/100g R 529 Chromium (Cr) mg/kg R 813 Copper (Cu) mg/kg R 529 Yes 5 to 7 Iron (Fe) mg/kg R 529 Yes 5 Inorganic Food Testing Potassium (K) mg/100g R 529 Yes 5 Phosphorus (P) mg/kg R 813 Subc 10 Selenium (Se) -

Medicine with Deeper, More Biochemical Conception Of
Edinburgh Medical Journal May 1951 APPLICATIONS of chemical defence research IN MEDICINET? By Professor R. A. PETERS to be invited It is a great responsibility, as well as a great honour, to Cameron lecturers give this lecture ; and I expect that with other I this ancient seat of share a sense of awe upon standing here in is learning. For me it is an awe tinged with much feeling (it possible that this is partly because some forebears of my mother came from near to recent of the this ; it is in part due the perusal city) perhaps " address here Froude in on the Times of John Knox, given by 1865 " " entitled Influence of the Reformation upon the Scottish Character ; this address drives home to the Sassenach the sterner stuff of which all of us in the the Scots are made ; but I really think it is more that as south, even if we come as I do from a Medical School as ancient the Hospital of St Bartholomew, know full well that Edinburgh stands high indeed in the world of Medical Schools as Scotland itself stands in the world of intellect. Elsewhere I have discussed fully the historical background of our for me back researches upon Chemical Defence substances, which go to World War I.23 Again in my Dixon lecture,24 I have indicated some of the interest for pharmacology. The outlook for medicine has not the been reviewed so far. Therefore I propose to-day to discuss of British influence upon the future of therapeutics of the discovery anti-lewisite and of some of the other researches upon war gas but substances ; and to consider not only the leads towards therapy, the possible newer trends given to medicine itself. -

Determination of Capsaicinoid Profile of Some Peppers Sold in Nigerian Markets
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2014, 6(4):648-654 ISSN : 0975-7384 Research Article CODEN(USA) : JCPRC5 Determination of capsaicinoid profile of some peppers sold in Nigerian markets 1N. C. Nwokem *, 2C. O. Nwokem, 2Y. O. Usman, 1O. J. Ocholi, 2M. L. Batari and 3A. A. Osunlaja 1Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria 2National Research Institute for Chemical Technology, Zaria, Nigeria 3Umar Suleiman College of Education, Gashua, Nigeria ____________________________________________________________________________________________ ABSTRACT The capsaicinoid profile of six different peppers sold in Nigerian markets was determined by Gas Chromatography- Mass Spectrometry. The capsaicinoids were extracted from the peppers using methanol as extractant and analyzed without need for derivatization. A total of eight (8) capsaicinoids were identified and quantitated: Capsaicin, Dihydrocapsaicin Dihydrocapsaicin 1, Dihydrocapsaicin 2, Norcapsaicin, Nordihydrocapsaicin 1, Nordihydrocapsaicin 2 and Nornordihydrocapsaicin though, not fully present in all the varieties. Dihydrocapsaicin 1, Dihydrocapsaicin 2, Nordihydrocapsaicin 1, Nordihydrocapsaicin 2 and Nornordihydrocapsaicin are isomers. Seven were identified in the Cameroun pepper variety, six in “Zaria atarugu” and Miango, and five in each of the remaining varieties. In all the peppers analyzed, capsaicin had the highest relative concentration, which ranged from 27.3% in the Cameroun variety to 49.38% in the “Zaria atarugu” variety. The sum of the relative concentrations of capsaicin and dihydrocapsaicin ranged from 47.03% in the “Miango” variety to 87.3% in the “Zaria atarugu” variety. Keywords: Capsaicinoids, Gas Chromatography-Mass Spectrometry, Methanol, Pepper ____________________________________________________________________________________________ INTRODUCTION Peppers are widely used in many parts of the world as a result of their valued sensory attributes; colour, purgency and aroma. -

Determination of Capsaicinoids in Peppers by Microwave-Assisted Extraction–High-Performance Liquid Chromatography with fluorescence Detection Gerardo F
Analytica Chimica Acta 578 (2006) 227–233 Determination of capsaicinoids in peppers by microwave-assisted extraction–high-performance liquid chromatography with fluorescence detection Gerardo F. Barbero, Miguel Palma ∗, Carmelo G. Barroso Grupo de Investigaci´on Qu´ımico Anal´ıtica del Vino y Productos Agroalimentarios, Departamento de Qu´ımica Analitica, Facultad de Ciencias, Universidad de C´adiz, Apartado 40, 11510 Puerto Real, C´adiz, Spain Received 30 March 2006; received in revised form 27 June 2006; accepted 28 June 2006 Available online 4 July 2006 Abstract A new method has been developed for the extraction of capsaicinoids (nordihydrocapsaicin, capsaicin, dihydrocapsaicin, homocapsaicin and homodihydrocapsaicin) in peppers employing microwave-assisted extraction. The parameters studied are: extraction solvent (methanol, ethanol, acetone, ethyl acetate and water), temperature (50–200 ◦C), sample quantity (0.1–1 g), volume of solvent (15–50 mL) and the extraction time (5–20 min). The results found for the optimum conditions are: 125 ◦C as extraction temperature, 25 mL of solvent, 0.5 g of freshly triturated peppers and extraction for 5 min, employing 100% ethanol as solvent. The capsaicinoids obtained were stable under the optimised extraction conditions. The resulting method presents a high degree of reproducibility (R.S.D. < 6%). © 2006 Elsevier B.V. All rights reserved. Keywords: Microwave-assisted extraction; Capsaicinoids; Peppers 1. Introduction Many techniques for the extraction of capsaicinoids from peppers have been studied, such as maceration [9], magnetic stir- Hot or spicy peppers are savoury food additives that are ring [10], Soxhlet [11,12], ultrasound-assisted extraction [13], widely utilised in many parts of the world, and are highly valued extraction by means of supercritical fluids [14], extraction by for their attributes of colour, pungency, and aroma.