Multiple and Stepwise Interactions Between Coatomer and ADP-Ribosylation Factor-1 (Arf1)-GTP
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Coatomer-Rich Endoplasmic Reticulum LELIO ORCI*, ALAIN PERRELET*, MARIELLA RAVAZZOLA*, Myltne AMHERDT*, JAMES E
Proc. Nati. Acad. Sci. USA Vol. 91, pp. 11924-11928, December 1994 Cell Biology Coatomer-rich endoplasmic reticulum LELIO ORCI*, ALAIN PERRELET*, MARIELLA RAVAZZOLA*, MYLtNE AMHERDT*, JAMES E. ROTHMANt, AND RANDY SCHEKMAN* *Department of Morphology, University of Geneva Medical School, 1211 Geneva 4, Switzerland; tDivision of Biochemistry and Molecular Biology, Howard Hughes Medical Institute, University of California, Berkeley, CA 94720; and tCellular Biochemistry and Biophysics Program, Memorial Sloan-Kettering Cancer Center, 1275 York Avenue, New York, NY 10021 Contributed by Randy Schekman, August 30, 1994 ABSTRACT We identify in normal cells the existence of digestion and incubated at normal (370C) or low (220C, 15'C, two distinct sites of the transitional endoplasmic reticulum 40C) temperatures under continuous shaking and gassing with (ER), one housing the Sec23p protein complex (the classical 95% 02/5% CO2. Monolayer cultures ofpancreatic endocrine transitional element), the other the coatomer protein complex cells (24) were exposed to low temperatures as for isolated (the coatomer-rich ER). Experimental conditions that reduce islets. ATP depletion in both preparations was induced at transport from the ER to the Golgi complex lead to the 370C with 10 ImM antimycin or 1 mM dinitrophenol or by overexpression of this newly defined coatomer-rich ER. gassing the cells with N2 instead of the 02/CO2 mixture. At the end of the incubations, the samples were fixed with Progress in the identification and characterization of the 1% glutaraldehyde in 0.1 M sodium phosphate buffer (pH 7.4) carriers involved in membrane traffic from the endoplasmic for 1 hr at the final temperature of the incubation protocol, reticulum (ER) to and through the Golgi complex was pos- washed with buffer, and processed for Epon embedding sible by combining morphological and biochemical methods, (conventional thin sections) or for cryoultramicrotomy ac- cell-free assay systems, and yeast genetics (for review see refs. -
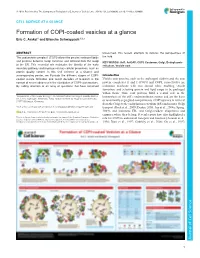
Formation of COPI-Coated Vesicles at a Glance Eric C
© 2018. Published by The Company of Biologists Ltd | Journal of Cell Science (2018) 131, jcs209890. doi:10.1242/jcs.209890 CELL SCIENCE AT A GLANCE Formation of COPI-coated vesicles at a glance Eric C. Arakel1 and Blanche Schwappach1,2,* ABSTRACT unresolved, this review attempts to refocus the perspectives of The coat protein complex I (COPI) allows the precise sorting of lipids the field. and proteins between Golgi cisternae and retrieval from the Golgi KEY WORDS: Arf1, ArfGAP, COPI, Coatomer, Golgi, Endoplasmic to the ER. This essential role maintains the identity of the early reticulum, Vesicle coat secretory pathway and impinges on key cellular processes, such as protein quality control. In this Cell Science at a Glance and accompanying poster, we illustrate the different stages of COPI- Introduction coated vesicle formation and revisit decades of research in the Vesicle coat proteins, such as the archetypal clathrin and the coat context of recent advances in the elucidation of COPI coat structure. protein complexes II and I (COPII and COPI, respectively) are By calling attention to an array of questions that have remained molecular machines with two central roles: enabling vesicle formation, and selecting protein and lipid cargo to be packaged within them. Thus, coat proteins fulfil a central role in the 1Department of Molecular Biology, Universitätsmedizin Göttingen, Humboldtallee homeostasis of the cell’s endomembrane system and are the basis 23, 37073 Göttingen, Germany. 2Max-Planck Institute for Biophysical Chemistry, 37077 Göttingen, Germany. of functionally segregated compartments. COPI operates in retrieval from the Golgi to the endoplasmic reticulum (ER) and in intra-Golgi *Author for correspondence ([email protected]) transport (Beck et al., 2009; Duden, 2003; Lee et al., 2004a; Spang, E.C.A., 0000-0001-7716-7149; B.S., 0000-0003-0225-6432 2009), and maintains ER- and Golgi-resident chaperones and enzymes where they belong. -

ADP-Ribosylation Factor, a Small GTP-Binding Protein, Is Required for Binding of the Coatomer Protein Fl-COP to Golgi Membranes JULIE G
Proc. Natl. Acad. Sci. USA Vol. 89, pp. 6408-6412, July 1992 Biochemistry ADP-ribosylation factor, a small GTP-binding protein, is required for binding of the coatomer protein fl-COP to Golgi membranes JULIE G. DONALDSON*, DAN CASSEL*t, RICHARD A. KAHN*, AND RICHARD D. KLAUSNER* *Cell Biology and Metabolism Branch, National Institute of Child Health and Human Development, and tLaboratory of Biological Chemistry, Division of Cancer Treatment, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892 Communicated by Marc Kirschner, April 20, 1992 (receivedfor review February 11, 1992) ABSTRACT The coatomer is a cytosolic protein complex localized to the Golgi complex, although their functions have that reversibly associates with Golgi membranes and is Impli- not been defined. Distinct among these proteins is the ADP- cated in modulating Golgi membrane transport. The associa- ribosylation factor (ARF), originally identified as a cofactor tion of 13-COP, a component of coatomer, with Golgi mem- required for in vitro cholera toxin-catalyzed ADP- branes is enhanced by guanosine 5'-[v-thioltriphosphate ribosylation of the a subunit of the trimeric GTP-binding (GTP[yS]), a nonhydrolyzable analogue of GTP, and by a protein G, (G,.) (19). ARF is an abundant cytosolic protein mixture of aluminum and fluoride ions (Al/F). Here we show that reversibly associates with Golgi membranes (20, 21). that the ADP-ribosylation factor (ARF) is required for the ARF has been shown to be present on Golgi coated vesicles binding of (-COP. Thus, 13-COP contained in a coatomer generated in the presence of GTP[yS], but it is not a com- fraction that has been resolved from ARF does not bind to Golgi ponent of the cytosolic coatomer (22). -

ADP-Ribosylation Factor and Coatomer Couple Fusion to Vesicle Budding Zvulun Elazar,* Lelio Orci,T Joachim Ostermann,* Myl~Nc Amherdt,* Gary Tanigawa,* and James E
ADP-Ribosylation Factor and Coatomer Couple Fusion to Vesicle Budding Zvulun Elazar,* Lelio Orci,t Joachim Ostermann,* Myl~nc Amherdt,* Gary Tanigawa,* and James E. Rothman* * Program in Cellular Biochemistry and Biophysics, Memorial Sloan Kettering Cancer Center, New York 10021; and ¢Institute of Histology and Embryology, University of Geneva Medical School, 1211 Geneva 4, Switzerland Abstract. The coat proteins required for budding pair directly without an intervening vesicle. Coupling COP-coated vesicles from Golgi membranes, coatomer may therefore result from the sequestration of fuso- and ADP-ribosylation factor (ARF) protein, are shown genic membrane proteins into assembling coated vesi- to be required to reconstitute the orderly process of cles that are only exposed when the coat is removed transport between Golgi cisternae in which fusion of after budding is complete. This mechanism of cou- transport vesicles begins only after budding ends. pling explains the phenomenon of "retrograde transport" When either coat protein is omitted, fusion is uncou- triggered by uncouplers such as the drug brefeldin A. pled from budding-donor and acceptor compartments ow is membrane fusion coupled to vesicle budding? Orci et al., 1989). Purification of COP-coated vesicles (Mal- A transport vesicle must fuse with its target only hotra et al., 1989; Serafini et al., 1991a,b) revealed that their H after its budding from the parental membrane is coats consist of a small GTP-binding protein (ADP-ribosyla- completed. Otherwise, the various membrane-bound com- tion factor, ARF) ~ and a complex of seven distinct proteins partments connected by vesicle shuttles would fuse and the termed coatomer (Waters et al., 1992a; Stenbeck et al., topological organization of the endomembrane system in 1993), whose subunits are or, B, B', 3', ~, e, and ~'-COPs. -

The Structure of the COPI Coat Determined Within the Cell
RESEARCH ADVANCE The structure of the COPI coat determined within the cell Yury S Bykov1,2, Miroslava Schaffer3, Svetlana O Dodonova1†, Sahradha Albert3, Ju¨ rgen M Plitzko3, Wolfgang Baumeister3*, Benjamin D Engel3*, John AG Briggs1,2* 1Structural and Computational Biology Unit, European Molecular Biology Laboratory, Heidelberg, Germany; 2Structural Studies Division, MRC Laboratory of Molecular Biology, Cambridge, United Kingdom; 3Department of Molecular Structural Biology, Max Planck Institute of Biochemistry, Martinsried, Germany Abstract COPI-coated vesicles mediate trafficking within the Golgi apparatus and from the Golgi to the endoplasmic reticulum. The structures of membrane protein coats, including COPI, have been extensively studied with in vitro reconstitution systems using purified components. Previously we have determined a complete structural model of the in vitro reconstituted COPI coat (Dodonova et al., 2017). Here, we applied cryo-focused ion beam milling, cryo-electron tomography and subtomogram averaging to determine the native structure of the COPI coat within vitrified Chlamydomonas reinhardtii cells. The native algal structure resembles the in vitro mammalian structure, but additionally reveals cargo bound beneath b’–COP. We find that all coat components *For correspondence: disassemble simultaneously and relatively rapidly after budding. Structural analysis in situ, [email protected] (WB); maintaining Golgi topology, shows that vesicles change their size, membrane thickness, and cargo [email protected] (BDE); content as they progress from cis to trans, but the structure of the coat machinery remains [email protected] constant. (JAGB) DOI: https://doi.org/10.7554/eLife.32493.001 Present address: †Department of Molecular Biology, Max Planck Institute for Biophysical Chemistry, Go¨ ttingen, Germany Introduction Vesicular transport between cellular compartments is a fundamental mechanism of eukaryotic cells. -

Cryo-Tomography of Coat Complexes ISSN 2059-7983
research papers Visualizing membrane trafficking through the electron microscope: cryo-tomography of coat complexes ISSN 2059-7983 Evgenia A. Markova and Giulia Zanetti* Institute of Structural and Molecular Biology, Birkbeck College, Malet Street, London WC1E 7HX, England. Received 8 March 2019 *Correspondence e-mail: [email protected] Accepted 12 April 2019 Coat proteins mediate vesicular transport between intracellular compartments, Keywords: cryo-electron tomography; which is essential for the distribution of molecules within the eukaryotic cell. three-dimensional reconstruction; coat proteins; The global arrangement of coat proteins on the membrane is key to their vesicular transport; COPII; subtomogram function, and cryo-electron tomography and subtomogram averaging have been averaging. used to study membrane-bound coat proteins, providing crucial structural insight. This review outlines a workflow for the structural elucidation of coat proteins, incorporating recent developments in the collection and processing of cryo-electron tomography data. Recent work on coat protein I, coat protein II and retromer performed on in vitro reconstitutions or in situ is summarized. These studies have answered long-standing questions regarding the mechanisms of membrane binding, polymerization and assembly regulation of coat proteins. 1. Introduction Recent advances in cryo-electron microscopy (cryo-EM) have enabled numerous high-resolution studies which have addressed long-standing structural biology questions. The large body of cryo-EM work carried out for protein structure characterization has utilized single-particle electron micro- scopy (Cheng, 2018). Recently, developments in hardware, data collection and data processing have placed cryo-electron tomography (cryo-ET) and subtomogram averaging (STA) at the forefront of structural studies of repetitive assemblies, reaching resolutions comparable to those of single-particle EM (Schur et al., 2016; Wan et al., 2017; Hutchings et al., 2018; Dodonova et al., 2017; Himes & Zhang, 2018). -

Mechanisms of Membrane Fusion: Disparate Players and Common Principles
REVIEWS Mechanisms of membrane fusion: disparate players and common principles Sascha Martens and Harvey T. McMahon Abstract | Membrane fusion can occur between cells, between different intracellular compartments, between intracellular compartments and the plasma membrane and between lipid-bound structures such as viral particles and cellular membranes. In order for membranes to fuse they must first be brought together. The more highly curved a membrane is, the more fusogenic it becomes. We discuss how proteins, including SNAREs, synaptotagmins and viral fusion proteins, might mediate close membrane apposition and induction of membrane curvature to drive diverse fusion processes. We also highlight common principles that can be derived from the analysis of the role of these proteins. Syncytium Membrane fusion is the process whereby two separate membrane–membrane proximity, by bending and A cell that contains multiple lipid bilayers merge to become one. It is essential for remodelling membranes, or by acting upstream to regu- nuclei and that is formed either communication between membrane-delineated comp- late the lipid or protein composition of the respective by cell–cell fusion or by artments in all eukaryotic cells (FIG. 1). The best-studied lipid bilayers. In the hemifusion model the fusion pore is incomplete cell division. process involving membrane fusion is exocytosis, lipidic, but according to an alternative hypothesis — the Hemifusion whereby vesicles fuse with the limiting membrane of protein-pore model — the initial fusion pore is generated An intermediate stage during a cell in order to release their contents (for example, and lined by transmembrane proteins rather than lipids2. membrane fusion that is hormones or neurotransmitters) into the extracellular There is good evidence that transmembrane domains characterized by the merger of SNARE only the contacting monolayers milieu, or to deposit receptors, transporters, channels of proteins are essential for efficient -dependent and not the two distal or adhesion molecules into the limiting membrane. -

Differential Localization of Coatomer Complex Isoforms Within the Golgi Apparatus
Differential localization of coatomer complex isoforms within the Golgi apparatus Jo¨ rg Moelleken*, Jo¨ rg Malsam*, Matthew J. Betts†, Ali Movafeghi‡, Ingeborg Reckmann*, Ingrid Meissner*, Andrea Hellwig§, Robert B. Russell†, Thomas So¨ llner*, Britta Bru¨ gger*, and Felix T. Wieland*¶ *Biochemistry Center and §Department of Neurobiology, University of Heidelberg, 69120 Heidelberg, Germany; †Structural and Computational Biology Unit, European Molecular Biology Laboratory, Meyerhofstrasse 1, 69117 Heidelberg, Germany; and ‡Department of Plant Biology, Faculty of Natural Sciences, Tabriz University, Tabriz, Iran Communicated by James E. Rothman, Columbia University Medical Center, New York, NY, December 20, 2006 (received for review December 8, 2006) Coatomer, the coat protein of coat protein complex (COP)I-vesicles, Genome analyses identified a second isoform for each of the is a soluble protein complex made up of seven subunits, ␣-, -, -, two coatomer subunits, ␥- and -COP (14), the paralogs ␥2- and ␥-, ␦-, -, and -COP. Higher eukaryotes have two paralogous 2-COP. Previously, the corresponding cDNAs were expressed versions of the ␥- and - subunits, termed ␥1- and ␥2-COP and 1- as tagged constructs (15), and the endogenous gene products and 2-COP. Different combinations of these subunits are known to were shown to be incorporated into coatomer complexes in a exist within coatomer complexes, and ␥1/1-, ␥1/2-, and ␥2/1- stoichiometric manner, which resulted in three major isoforms of COP represent the major coatomer populations in mammals. The the complex with the compositions ␥11-, ␥12-, and ␥21-COP role of COPI vesicles in the early secretory pathway is the subject (16). To extend this work, we have prepared polyclonal antibod- ␥ ␥ of considerable debate. -

University of London Thesis
REFERENCE ONLY UNIVERSITY OF LONDON THESIS Degree fVvO Year""^© *" Name of Author iA vAa ^ V COPYRIGHT This is a thesis accepted for a Higher Degree of the University of London. It is an unpublished typescript and the copyright is held by the author. All persons consulting the thesis must read and abide by the Copyright Declaration below. COPYRIGHT DECLARATION I recognise that the copyright of the above-described thesis rests with the author and that no quotation from it or information derived from it may be published without the prior written consent of the author. LOANS Theses may not be lent to individuals, but the Senate House Library may lend a copy to approved libraries within the United Kingdom, for consultation solely on the premises of those libraries. Application should be made to: Inter-Library Loans, Senate House Library, Senate House, Malet Street, London WC1E 7HU. REPRODUCTION University of London theses may not be reproduced without explicit written permission from the Senate House Library. Enquiries should be addressed to the Theses Section of the Library. Regulations concerning reproduction vary according to the date of acceptance of the thesis and are listed below as guidelines. A. Before 1962. Permission granted only upon the prior written consent of the author. (The Senate House Library will provide addresses where possible). B. 1962- 1974. In many cases the author has agreed to permit copying upon completion of a Copyright Declaration. C. 1975 - 1988. Most theses may be copied upon completion of a Copyright Declaration. D. 1989 onwards. Most theses may be copied. This thesis comes within category D. -

Coatomer, Arf1p, and Nucleotide Are Required to Bud Coat Protein Complex I-Coated Vesicles from Large Synthetic Liposomes
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 11199–11204, September 1998 Cell Biology This contribution is part of the special series of Inaugural Articles by members of the National Academy of Sciences elected on April 28, 1998. Coatomer, Arf1p, and nucleotide are required to bud coat protein complex I-coated vesicles from large synthetic liposomes ANNE SPANG*, KEN MATSUOKA*, SUSAN HAMAMOTO*, RANDY SCHEKMAN*, AND LELIO ORCI† *Department of Molecular and Cell Biology and Howard Hughes Medical Institute, University of California, Berkeley, CA 94720; and †Department of Morphology, University of Geneva, CH-1211 Geneva, Switzerland Contributed by Lelio Orci, July 30, 1998 ABSTRACT Synthetic coat protein complex I (COPI)- favorable binding environment for coatomer (14). According coated vesicles form spontaneously from large ('300 nm in to this view, Arf1p-GTP may act indirectly to promote COPI- diameter), chemically defined liposomes incubated with coated vesicle formation. coatomer, Arf1p, and guanosine 5*-[g-thio]triphosphate. Coatomer recruitment to membranes also may be driven by Coated vesicles are 40–70 nm in diameter, approximately the the recognition of protein cargo molecules. The C-terminal size of COPI vesicles formed from native membranes. The KKXX-motif of membrane proteins is known to interact with formation of COPI-coated buds and vesicles and the binding coatomer and to engage COPI vesicles in retrograde transport of Arf1p to donor liposomes depends on guanosine 5*-[g- from the Golgi to the ER. However, although subunits of the thio]triphosphate. In contrast to the behavior of the COPII anterograde carrier, COPII, also interact directly with protein coat, coatomer binds to liposomes containing a variety of cargo leaving the ER membrane (15), the COPII coat is charged or neutral phospholipids. -

June N&V Final
NEWS AND VIEWS studies is not fully functional, although this is disturbed, whereas binding of the calcium- our knowledge of the various steps in the not proven. An alternative explanation is that activated sytI domain would be sufficient to fusion process and their regulatory mecha- at synapses and in cells, full-length sytI (an promote SNARE zippering and fusion (Fig. 2). nisms. This might ultimately result in the integral membrane protein) is needed not only In conclusion, the results from Tucker et identification of other factors that regulate the for exocytic fusion, but also to initiate vesicle al.1 are an important milestone in the recon- system, in particular those allowing mem- docking to the plasma membrane through stitution of presynaptic events that result in brane fusion to operate in the sub-millisecond binding to SNAP25. In this case, inhibition by neurotransmitter release. On the basis of these timescale as it does in synaptic physiology. the cytosolic domain could be explained by an results, the need for proteins and lipids other 1. Tucker, W. C., Weber, T. & Chapman, E. R. Science unproductive interaction with the t-SNARE than sytI in the calcium regulation of synaptic 304, 435–438 (2004). 5,6 that prevents binding of full-length sytI (refs 9, vesicle fusion can finally be excluded. 2. Weber, T. et al. Cell 92, 759–72 (1998). 10). The reconstituted bilayers, however, could Exocytic fusion, however, is a complex process 3. Geppert, M. et al. Cell 79, 717–727 (1994). 4. Ubach, J. et al. EMBO J. 17, 3921–3930 (1998). -

Vesicle Coats: Structure, Function, And
Review Vesicle coats: structure, function, and general principles of assembly 1 2 2 1,3 Marco Faini , Rainer Beck , Felix T. Wieland , and John A.G. Briggs 1 Structural and Computational Biology Unit, European Molecular Biology Laboratory, Meyerhofstrasse 1, 69117 Heidelberg, Germany 2 Heidelberg University Biochemistry Center, Heidelberg University, Im Neuenheimer Feld 328, 69120 Heidelberg, Germany 3 Cell Biology and Biophysics Unit, European Molecular Biology Laboratory, Meyerhofstrasse 1, 69117 Heidelberg, Germany The transport of proteins and lipids between distinct The three best-characterized types of vesicular carrier cellular compartments is conducted by coated vesicles. involved in intracellular trafficking are distinguished by These vesicles are formed by the self-assembly of coat their different coat proteins and their different trafficking proteins on a membrane, leading to collection of the routes. Clathrin-coated vesicles (CCVs) act in the late vesicle cargo and membrane bending to form a bud. secretory pathway and in the endocytic pathway, Scission at the bud neck releases the vesicle. X-ray COPII-coated vesicles export proteins from the endoplas- crystallography and electron microscopy (EM) have re- mic reticulum (ER), and COPI-coated vesicles shuttle cently generated models of isolated coat components within the Golgi organelle and from the Golgi back to and assembled coats. Here, we review these data to the ER. Despite having different compartment specifici- present a structural overview of the three main coats: ties and different structural components, the mechanisms clathrin, COPII, and COPI. The three coats have similar of their formation follow similar rules. The time and place function, common ancestry, and structural similarities, at which vesicle formation occurs are most often regulated but exhibit fundamental differences in structure and by small GTP-binding proteins.