Vesicle Coats: Structure, Function, And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Large-Scale Conformational Change Couples Membrane Recruitment to Cargo Binding in the AP2 Clathrin Adaptor Complex
A Large-Scale Conformational Change Couples Membrane Recruitment to Cargo Binding in the AP2 Clathrin Adaptor Complex Lauren P. Jackson,1,5 Bernard T. Kelly,1,5 Airlie J. McCoy,1 Thomas Gaffry,2 Leo C. James,3 Brett M. Collins,4 Stefan Ho¨ ning,2 Philip R. Evans,3,* and David J. Owen1,* 1Cambridge Institute for Medical Research, Department of Clinical Biochemistry, University of Cambridge, Hills Road, Cambridge CB2 0XY, UK 2Institute of Biochemistry I and Center for Molecular Medicine Cologne, University of Cologne, Joseph-Stelzmann-Str. 52 50931 Cologne, Germany 3Medical Research Council Laboratory of Molecular Biology, Hills Road, Cambridge CB2 0QH, UK 4Institute for Molecular Bioscience, The University of Queensland, Brisbane QLD 4072, Australia 5These authors contributed equally to this work *Correspondence: [email protected] (P.R.E.), [email protected] (D.J.O.) DOI 10.1016/j.cell.2010.05.006 SUMMARY by clathrin adaptors to an outer polymeric clathrin scaffold (Cheng et al., 2007; Fotin et al., 2004). Clathrin adaptors contain The AP2 adaptor complex (a, b2, s2, and m2 sub- a folded membrane-proximal domain, which binds to phospha- units) crosslinks the endocytic clathrin scaffold to tidyl inositol polyphosphate (PIP) headgroups and/or Arf PtdIns4,5P2-containing membranes and transmem- GTPases in their membrane-attached, GTP-bound forms, and brane protein cargo. In the ‘‘locked’’ cytosolic form, at least one natively unstructured region, which harbors a cla- AP2’s binding sites for the two endocytic motifs, thrin-binding motif (Owen et al., 2004). Transmembrane proteins YxxF on the C-terminal domain of m2 (C-m2) and are generally selected as cargo for incorporation into a CCV through the direct interaction of either widely used, short, linear [ED]xxxL[LI] on s2, are blocked by parts of b2. -

Mea6 Controls VLDL Transport Through the Coordinated Regulation of COPII Assembly
Cell Research (2016) 26:787-804. npg © 2016 IBCB, SIBS, CAS All rights reserved 1001-0602/16 $ 32.00 ORIGINAL ARTICLE www.nature.com/cr Mea6 controls VLDL transport through the coordinated regulation of COPII assembly Yaqing Wang1, *, Liang Liu1, 2, *, Hongsheng Zhang1, 2, Junwan Fan1, 2, Feng Zhang1, 2, Mei Yu3, Lei Shi1, Lin Yang1, Sin Man Lam1, Huimin Wang4, Xiaowei Chen4, Yingchun Wang1, Fei Gao5, Guanghou Shui1, Zhiheng Xu1, 6 1State Key Laboratory of Molecular Developmental Biology, Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, Beijing 100101, China; 2University of Chinese Academy of Sciences, Beijing 100101, China; 3School of Life Science, Shandong University, Jinan 250100, China; 4Institute of Molecular Medicine, Peking University, Beijing 100871, China; 5State Key Laboratory of Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences, Beijing 100101, China; 6Translation- al Medical Center for Stem Cell Therapy, Shanghai East Hospital, Tongji University School of Medicine, Shanghai 200120, China Lipid accumulation, which may be caused by the disturbance in very low density lipoprotein (VLDL) secretion in the liver, can lead to fatty liver disease. VLDL is synthesized in endoplasmic reticulum (ER) and transported to Golgi apparatus for secretion into plasma. However, the underlying molecular mechanism for VLDL transport is still poor- ly understood. Here we show that hepatocyte-specific deletion of meningioma-expressed antigen 6 (Mea6)/cutaneous T cell lymphoma-associated antigen 5C (cTAGE5C) leads to severe fatty liver and hypolipemia in mice. Quantitative lip- idomic and proteomic analyses indicate that Mea6/cTAGE5 deletion impairs the secretion of different types of lipids and proteins, including VLDL, from the liver. -

Protein Sorting and Vesicular Traffic in the Golgi Apparatus
The Golgi Apparatus 63 E.G. Berger & J. Roth (eds) © 1997 Birkhauser Verlag Basel/Switzerland Protein sorting and vesicular traffic in the Golgi apparatus M.G. Farquhar1 and H.-P. Hauri2 JDivision ofCellular and Molecular Medicine and Department ofPathology, University ofCalifornia, San Diego, CA 92093-0651, USA 2Department ofPharmacology, Biozelltrum, University ofBasel, CH-4056 Basel, Switzerland Summary 65 Introduction 65 Major routes of protein and membrane traffic to and through the GA 66 Overview oftraffic along the exocytic pathway 66 Models ofGolgi organization 68 Mechanisms of sorting, targeting and transport from the ER 71 Exitfrom the ER occurs at specific export sites 71 Selective transportfrom the ER vs bulk-jlow models 72 Transit through the ER-Golgi intermediate compartment (ERGIC) 74 Other proximal pre-Golgi, pre-ERGIC, smooth ER compartments 76 Mechanisms for retention and retrieval ofresident ER proteins: Current models 79 Transport, processing and sorting by the GA 80 Contributions ofin vitro assays 82 The SNARE hypothesis 82 Golgi compartments and post-translational processing oftransported proteins 83 Golgi-specific functions . 83 How many Golgi compartments are there? 83 Proposed mechanisms for retention and retrieval ofresident Golgi proteins 86 Transmembrane domain retention signals 88 Formation ofinsoluble aggregates too large to enter transport vesicles 88 The kin recognition model 89 Bilayer-mediated sorting 89 Sorting at the TGN 90 Sorting oflysosomal enzymes 92 64 M.G. Farquhar and H.-P. Hauri Sorting and packaging -

Coatomer-Rich Endoplasmic Reticulum LELIO ORCI*, ALAIN PERRELET*, MARIELLA RAVAZZOLA*, Myltne AMHERDT*, JAMES E
Proc. Nati. Acad. Sci. USA Vol. 91, pp. 11924-11928, December 1994 Cell Biology Coatomer-rich endoplasmic reticulum LELIO ORCI*, ALAIN PERRELET*, MARIELLA RAVAZZOLA*, MYLtNE AMHERDT*, JAMES E. ROTHMANt, AND RANDY SCHEKMAN* *Department of Morphology, University of Geneva Medical School, 1211 Geneva 4, Switzerland; tDivision of Biochemistry and Molecular Biology, Howard Hughes Medical Institute, University of California, Berkeley, CA 94720; and tCellular Biochemistry and Biophysics Program, Memorial Sloan-Kettering Cancer Center, 1275 York Avenue, New York, NY 10021 Contributed by Randy Schekman, August 30, 1994 ABSTRACT We identify in normal cells the existence of digestion and incubated at normal (370C) or low (220C, 15'C, two distinct sites of the transitional endoplasmic reticulum 40C) temperatures under continuous shaking and gassing with (ER), one housing the Sec23p protein complex (the classical 95% 02/5% CO2. Monolayer cultures ofpancreatic endocrine transitional element), the other the coatomer protein complex cells (24) were exposed to low temperatures as for isolated (the coatomer-rich ER). Experimental conditions that reduce islets. ATP depletion in both preparations was induced at transport from the ER to the Golgi complex lead to the 370C with 10 ImM antimycin or 1 mM dinitrophenol or by overexpression of this newly defined coatomer-rich ER. gassing the cells with N2 instead of the 02/CO2 mixture. At the end of the incubations, the samples were fixed with Progress in the identification and characterization of the 1% glutaraldehyde in 0.1 M sodium phosphate buffer (pH 7.4) carriers involved in membrane traffic from the endoplasmic for 1 hr at the final temperature of the incubation protocol, reticulum (ER) to and through the Golgi complex was pos- washed with buffer, and processed for Epon embedding sible by combining morphological and biochemical methods, (conventional thin sections) or for cryoultramicrotomy ac- cell-free assay systems, and yeast genetics (for review see refs. -

Conserved and Novel Properties of Clathrin-Mediated Endocytosis in Dictyostelium Discoideum" (2012)
Rockefeller University Digital Commons @ RU Student Theses and Dissertations 2012 Conserved and Novel Properties of Clathrin- Mediated Endocytosis in Dictyostelium Discoideum Laura Macro Follow this and additional works at: http://digitalcommons.rockefeller.edu/ student_theses_and_dissertations Part of the Life Sciences Commons Recommended Citation Macro, Laura, "Conserved and Novel Properties of Clathrin-Mediated Endocytosis in Dictyostelium Discoideum" (2012). Student Theses and Dissertations. Paper 163. This Thesis is brought to you for free and open access by Digital Commons @ RU. It has been accepted for inclusion in Student Theses and Dissertations by an authorized administrator of Digital Commons @ RU. For more information, please contact [email protected]. CONSERVED AND NOVEL PROPERTIES OF CLATHRIN- MEDIATED ENDOCYTOSIS IN DICTYOSTELIUM DISCOIDEUM A Thesis Presented to the Faculty of The Rockefeller University in Partial Fulfillment of the Requirements for the degree of Doctor of Philosophy by Laura Macro June 2012 © Copyright by Laura Macro 2012 CONSERVED AND NOVEL PROPERTIES OF CLATHRIN- MEDIATED ENDOCYTOSIS IN DICTYOSTELIUM DISCOIDEUM Laura Macro, Ph.D. The Rockefeller University 2012 The protein clathrin mediates one of the major pathways of endocytosis from the extracellular milieu and plasma membrane. Clathrin functions with a network of interacting accessory proteins, one of which is the adaptor complex AP-2, to co-ordinate vesicle formation. Disruption of genes involved in clathrin-mediated endocytosis causes embryonic lethality in multicellular animals suggesting that clathrin-mediated endocytosis is a fundamental cellular process. However, loss of clathrin-mediated endocytosis genes in single cell eukaryotes, such as S.cerevisiae (yeast), does not cause lethality, suggesting that clathrin may convey specific advantages for multicellularity. -

An Arf1 Synthetic Lethal Screen Identifies a New Clathrin Heavy
Copyright 1998 by the Genetics Society of America An arf1D Synthetic Lethal Screen Identi®es a New Clathrin Heavy Chain Conditional Allele That Perturbs Vacuolar Protein Transport in Saccharomyces cerevisiae Chih-Ying Chen and Todd R. Graham Department of Molecular Biology, Vanderbilt University, Nashville, Tennessee 37235 Manuscript received March 5, 1998 Accepted for publication June 16, 1998 ABSTRACT ADP-ribosylation factor (ARF) is a small GTP-binding protein that is thought to regulate the assembly of coat proteins on transport vesicles. To identify factors that functionally interact with ARF, we have performed a genetic screen in Saccharomyces cerevisiae for mutations that exhibit synthetic lethality with an arf1D allele and de®ned seven genes by complementation tests (SWA1-7 for synthetically lethal with arf1D). Most of the swa mutants exhibit phenotypes comparable to arf1D mutants such as temperature-conditional growth, hypersensitivity to ¯uoride ions, and partial protein transport and glycosylation defects. Here, we report that swa5-1 is a new temperature-sensitive allele of the clathrin heavy chain gene (chc1-5), which carries a frameshift mutation near the 39 end of the CHC1 open reading frame. This genetic interaction between arf1 and chc1 provides in vivo evidence for a role for ARF in clathrin coat assembly. Surprisingly, strains harboring chc1-5 exhibited a signi®cant defect in transport of carboxypeptidase Y or carboxypepti- dase S to the vacuole that was not observed in other chc1 ts mutants. The kinetics of invertase secretion or transport of alkaline phosphatase to the vacuole were not signi®cantly affected in the chc1-5 mutant, further implicating clathrin speci®cally in the Golgi to vacuole transport pathway for carboxypeptidase Y. -
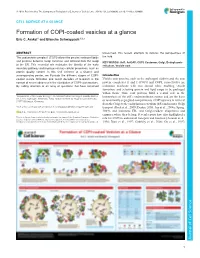
Formation of COPI-Coated Vesicles at a Glance Eric C
© 2018. Published by The Company of Biologists Ltd | Journal of Cell Science (2018) 131, jcs209890. doi:10.1242/jcs.209890 CELL SCIENCE AT A GLANCE Formation of COPI-coated vesicles at a glance Eric C. Arakel1 and Blanche Schwappach1,2,* ABSTRACT unresolved, this review attempts to refocus the perspectives of The coat protein complex I (COPI) allows the precise sorting of lipids the field. and proteins between Golgi cisternae and retrieval from the Golgi KEY WORDS: Arf1, ArfGAP, COPI, Coatomer, Golgi, Endoplasmic to the ER. This essential role maintains the identity of the early reticulum, Vesicle coat secretory pathway and impinges on key cellular processes, such as protein quality control. In this Cell Science at a Glance and accompanying poster, we illustrate the different stages of COPI- Introduction coated vesicle formation and revisit decades of research in the Vesicle coat proteins, such as the archetypal clathrin and the coat context of recent advances in the elucidation of COPI coat structure. protein complexes II and I (COPII and COPI, respectively) are By calling attention to an array of questions that have remained molecular machines with two central roles: enabling vesicle formation, and selecting protein and lipid cargo to be packaged within them. Thus, coat proteins fulfil a central role in the 1Department of Molecular Biology, Universitätsmedizin Göttingen, Humboldtallee homeostasis of the cell’s endomembrane system and are the basis 23, 37073 Göttingen, Germany. 2Max-Planck Institute for Biophysical Chemistry, 37077 Göttingen, Germany. of functionally segregated compartments. COPI operates in retrieval from the Golgi to the endoplasmic reticulum (ER) and in intra-Golgi *Author for correspondence ([email protected]) transport (Beck et al., 2009; Duden, 2003; Lee et al., 2004a; Spang, E.C.A., 0000-0001-7716-7149; B.S., 0000-0003-0225-6432 2009), and maintains ER- and Golgi-resident chaperones and enzymes where they belong. -

ADP-Ribosylation Factor, a Small GTP-Binding Protein, Is Required for Binding of the Coatomer Protein Fl-COP to Golgi Membranes JULIE G
Proc. Natl. Acad. Sci. USA Vol. 89, pp. 6408-6412, July 1992 Biochemistry ADP-ribosylation factor, a small GTP-binding protein, is required for binding of the coatomer protein fl-COP to Golgi membranes JULIE G. DONALDSON*, DAN CASSEL*t, RICHARD A. KAHN*, AND RICHARD D. KLAUSNER* *Cell Biology and Metabolism Branch, National Institute of Child Health and Human Development, and tLaboratory of Biological Chemistry, Division of Cancer Treatment, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892 Communicated by Marc Kirschner, April 20, 1992 (receivedfor review February 11, 1992) ABSTRACT The coatomer is a cytosolic protein complex localized to the Golgi complex, although their functions have that reversibly associates with Golgi membranes and is Impli- not been defined. Distinct among these proteins is the ADP- cated in modulating Golgi membrane transport. The associa- ribosylation factor (ARF), originally identified as a cofactor tion of 13-COP, a component of coatomer, with Golgi mem- required for in vitro cholera toxin-catalyzed ADP- branes is enhanced by guanosine 5'-[v-thioltriphosphate ribosylation of the a subunit of the trimeric GTP-binding (GTP[yS]), a nonhydrolyzable analogue of GTP, and by a protein G, (G,.) (19). ARF is an abundant cytosolic protein mixture of aluminum and fluoride ions (Al/F). Here we show that reversibly associates with Golgi membranes (20, 21). that the ADP-ribosylation factor (ARF) is required for the ARF has been shown to be present on Golgi coated vesicles binding of (-COP. Thus, 13-COP contained in a coatomer generated in the presence of GTP[yS], but it is not a com- fraction that has been resolved from ARF does not bind to Golgi ponent of the cytosolic coatomer (22). -

Mechanisms of Synaptic Plasticity Mediated by Clathrin Adaptor-Protein Complexes 1 and 2 in Mice
Mechanisms of synaptic plasticity mediated by Clathrin Adaptor-protein complexes 1 and 2 in mice Dissertation for the award of the degree “Doctor rerum naturalium” at the Georg-August-University Göttingen within the doctoral program “Molecular Biology of Cells” of the Georg-August University School of Science (GAUSS) Submitted by Ratnakar Mishra Born in Birpur, Bihar, India Göttingen, Germany 2019 1 Members of the Thesis Committee Prof. Dr. Peter Schu Institute for Cellular Biochemistry, (Supervisor and first referee) University Medical Center Göttingen, Germany Dr. Hans Dieter Schmitt Neurobiology, Max Planck Institute (Second referee) for Biophysical Chemistry, Göttingen, Germany Prof. Dr. med. Thomas A. Bayer Division of Molecular Psychiatry, University Medical Center, Göttingen, Germany Additional Members of the Examination Board Prof. Dr. Silvio O. Rizzoli Department of Neuro-and Sensory Physiology, University Medical Center Göttingen, Germany Dr. Roland Dosch Institute of Developmental Biochemistry, University Medical Center Göttingen, Germany Prof. Dr. med. Martin Oppermann Institute of Cellular and Molecular Immunology, University Medical Center, Göttingen, Germany Date of oral examination: 14th may 2019 2 Table of Contents List of abbreviations ................................................................................. 5 Abstract ................................................................................................... 7 Chapter 1: Introduction ............................................................................ -

COPI Activity Coupled with Fatty Acid Biosynthesis Is Required for Viral Replication
COPI Activity Coupled with Fatty Acid Biosynthesis Is Required for Viral Replication Sara Cherry1*, Amit Kunte2, Hui Wang3, Carolyn Coyne4, Robert B. Rawson2, Norbert Perrimon3 1 University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, United States of America, 2 University of Texas Southwestern Medical Center, Dallas, Texas, United States of America, 3 Harvard Medical School, Howard Hughes Medical Institute, Boston, Massachusetts, United States of America, 4 Children’s Hospital of Pennsylvania, Philadelphia, Pennsylvania, United States of America During infection by diverse viral families, RNA replication occurs on the surface of virally induced cytoplasmic membranes of cellular origin. How this process is regulated, and which cellular factors are required, has been unclear. Moreover, the host–pathogen interactions that facilitate the formation of this new compartment might represent critical determinants of viral pathogenesis, and their elucidation may lead to novel insights into the coordination of vesicular trafficking events during infection. Here we show that in Drosophila cells, Drosophila C virus remodels the Golgi apparatus and forms a novel vesicular compartment, on the surface of which viral RNA replication takes place. Using genome-wide RNA interference screening, we found that this step in the viral lifecycle requires at least two host encoded pathways: the coat protein complex I (COPI) coatamer and fatty acid biosynthesis. Our results integrate, clarify, and extend numerous observations concerning the cell biology of viral replication, allowing us to conclude that the coupling of new cellular membrane formation with the budding of these vesicles from the Golgi apparatus allows for the regulated generation of this new virogenic organelle, which is essential for viral replication. -

ADP-Ribosylation Factor and Coatomer Couple Fusion to Vesicle Budding Zvulun Elazar,* Lelio Orci,T Joachim Ostermann,* Myl~Nc Amherdt,* Gary Tanigawa,* and James E
ADP-Ribosylation Factor and Coatomer Couple Fusion to Vesicle Budding Zvulun Elazar,* Lelio Orci,t Joachim Ostermann,* Myl~nc Amherdt,* Gary Tanigawa,* and James E. Rothman* * Program in Cellular Biochemistry and Biophysics, Memorial Sloan Kettering Cancer Center, New York 10021; and ¢Institute of Histology and Embryology, University of Geneva Medical School, 1211 Geneva 4, Switzerland Abstract. The coat proteins required for budding pair directly without an intervening vesicle. Coupling COP-coated vesicles from Golgi membranes, coatomer may therefore result from the sequestration of fuso- and ADP-ribosylation factor (ARF) protein, are shown genic membrane proteins into assembling coated vesi- to be required to reconstitute the orderly process of cles that are only exposed when the coat is removed transport between Golgi cisternae in which fusion of after budding is complete. This mechanism of cou- transport vesicles begins only after budding ends. pling explains the phenomenon of "retrograde transport" When either coat protein is omitted, fusion is uncou- triggered by uncouplers such as the drug brefeldin A. pled from budding-donor and acceptor compartments ow is membrane fusion coupled to vesicle budding? Orci et al., 1989). Purification of COP-coated vesicles (Mal- A transport vesicle must fuse with its target only hotra et al., 1989; Serafini et al., 1991a,b) revealed that their H after its budding from the parental membrane is coats consist of a small GTP-binding protein (ADP-ribosyla- completed. Otherwise, the various membrane-bound com- tion factor, ARF) ~ and a complex of seven distinct proteins partments connected by vesicle shuttles would fuse and the termed coatomer (Waters et al., 1992a; Stenbeck et al., topological organization of the endomembrane system in 1993), whose subunits are or, B, B', 3', ~, e, and ~'-COPs. -

Sec13 Safeguards the Integrity of the Endoplasmic Reticulum and Organogenesis of the Digestive System in Zebrafish
Developmental Biology 367 (2012) 197–207 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Sec13 safeguards the integrity of the endoplasmic reticulum and organogenesis of the digestive system in zebrafish Xubo Niu a,1, Chuan Gao b,1, Li Jan Lo a, Yue Luo a, Chunmei Meng c, Jian Hong c, Wanjin Hong d,e, Jinrong Peng a,n a Key Laboratory for Molecular Animal Nutrition, Ministry of Education, College of Animal Sciences, Zhejiang University, Hangzhou, PR China b Department of Biological Sciences, National University of Singapore, Singapore c Institute of Biotechnology, Zhejiang University, Hangzhou, PR China d School of Pharmaceutical Sciences, Xiamen University, Xiamen, PR China e Institute of Molecular and Cell Biology, Agency for Science and Technology Research (A*STAR), Singapore article info abstract Article history: The Sec13-Sec31 heterotetramer serves as the outer coat in the COPII complex, which mediates protein Received 25 October 2011 trafficking from the endoplasmic reticulum (ER) to the Golgi apparatus. Although it has been studied in Received in revised form depth in yeast and cultured cells, the role of COPII in organogenesis in a multicellular organism has not. 12 April 2012 We report here that a zebrafish sec13sq198 mutant, which exhibits a phenotype of hypoplastic digestive Accepted 4 May 2012 organs, has a mutation in the sec13 gene. The mutant gene encodes a carboxyl-terminus-truncated Available online 15 May 2012 Sec13 that loses its affinity to Sec31a, which leads to disintegration of the ER structure in various Keywords: differentiated cells in sec13sq198, including chondrocytes, intestinal epithelial cells and hepatocytes.