Studies on the Inhibition of Endosome Fusion by Gtpγs-Bound ARF
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hras Intracellular Trafficking and Signal Transduction Jodi Ho-Jung Mckay Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 2007 HRas intracellular trafficking and signal transduction Jodi Ho-Jung McKay Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Biological Phenomena, Cell Phenomena, and Immunity Commons, Cancer Biology Commons, Cell Biology Commons, Genetics and Genomics Commons, and the Medical Cell Biology Commons Recommended Citation McKay, Jodi Ho-Jung, "HRas intracellular trafficking and signal transduction" (2007). Retrospective Theses and Dissertations. 13946. https://lib.dr.iastate.edu/rtd/13946 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. HRas intracellular trafficking and signal transduction by Jodi Ho-Jung McKay A dissertation submitted to the graduate faculty in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY Major: Genetics Program of Study Committee: Janice E. Buss, Co-major Professor Linda Ambrosio, Co-major Professor Diane Bassham Drena Dobbs Ted Huiatt Iowa State University Ames, Iowa 2007 Copyright © Jodi Ho-Jung McKay, 2007. All rights reserved. UMI Number: 3274881 Copyright 2007 by McKay, Jodi Ho-Jung All rights reserved. UMI Microform 3274881 Copyright 2008 by ProQuest Information and Learning Company. All rights reserved. This microform edition is protected against unauthorized copying under Title 17, United States Code. ProQuest Information and Learning Company 300 North Zeeb Road P.O. -

Coatomer-Rich Endoplasmic Reticulum LELIO ORCI*, ALAIN PERRELET*, MARIELLA RAVAZZOLA*, Myltne AMHERDT*, JAMES E
Proc. Nati. Acad. Sci. USA Vol. 91, pp. 11924-11928, December 1994 Cell Biology Coatomer-rich endoplasmic reticulum LELIO ORCI*, ALAIN PERRELET*, MARIELLA RAVAZZOLA*, MYLtNE AMHERDT*, JAMES E. ROTHMANt, AND RANDY SCHEKMAN* *Department of Morphology, University of Geneva Medical School, 1211 Geneva 4, Switzerland; tDivision of Biochemistry and Molecular Biology, Howard Hughes Medical Institute, University of California, Berkeley, CA 94720; and tCellular Biochemistry and Biophysics Program, Memorial Sloan-Kettering Cancer Center, 1275 York Avenue, New York, NY 10021 Contributed by Randy Schekman, August 30, 1994 ABSTRACT We identify in normal cells the existence of digestion and incubated at normal (370C) or low (220C, 15'C, two distinct sites of the transitional endoplasmic reticulum 40C) temperatures under continuous shaking and gassing with (ER), one housing the Sec23p protein complex (the classical 95% 02/5% CO2. Monolayer cultures ofpancreatic endocrine transitional element), the other the coatomer protein complex cells (24) were exposed to low temperatures as for isolated (the coatomer-rich ER). Experimental conditions that reduce islets. ATP depletion in both preparations was induced at transport from the ER to the Golgi complex lead to the 370C with 10 ImM antimycin or 1 mM dinitrophenol or by overexpression of this newly defined coatomer-rich ER. gassing the cells with N2 instead of the 02/CO2 mixture. At the end of the incubations, the samples were fixed with Progress in the identification and characterization of the 1% glutaraldehyde in 0.1 M sodium phosphate buffer (pH 7.4) carriers involved in membrane traffic from the endoplasmic for 1 hr at the final temperature of the incubation protocol, reticulum (ER) to and through the Golgi complex was pos- washed with buffer, and processed for Epon embedding sible by combining morphological and biochemical methods, (conventional thin sections) or for cryoultramicrotomy ac- cell-free assay systems, and yeast genetics (for review see refs. -

An Arf1 Synthetic Lethal Screen Identifies a New Clathrin Heavy
Copyright 1998 by the Genetics Society of America An arf1D Synthetic Lethal Screen Identi®es a New Clathrin Heavy Chain Conditional Allele That Perturbs Vacuolar Protein Transport in Saccharomyces cerevisiae Chih-Ying Chen and Todd R. Graham Department of Molecular Biology, Vanderbilt University, Nashville, Tennessee 37235 Manuscript received March 5, 1998 Accepted for publication June 16, 1998 ABSTRACT ADP-ribosylation factor (ARF) is a small GTP-binding protein that is thought to regulate the assembly of coat proteins on transport vesicles. To identify factors that functionally interact with ARF, we have performed a genetic screen in Saccharomyces cerevisiae for mutations that exhibit synthetic lethality with an arf1D allele and de®ned seven genes by complementation tests (SWA1-7 for synthetically lethal with arf1D). Most of the swa mutants exhibit phenotypes comparable to arf1D mutants such as temperature-conditional growth, hypersensitivity to ¯uoride ions, and partial protein transport and glycosylation defects. Here, we report that swa5-1 is a new temperature-sensitive allele of the clathrin heavy chain gene (chc1-5), which carries a frameshift mutation near the 39 end of the CHC1 open reading frame. This genetic interaction between arf1 and chc1 provides in vivo evidence for a role for ARF in clathrin coat assembly. Surprisingly, strains harboring chc1-5 exhibited a signi®cant defect in transport of carboxypeptidase Y or carboxypepti- dase S to the vacuole that was not observed in other chc1 ts mutants. The kinetics of invertase secretion or transport of alkaline phosphatase to the vacuole were not signi®cantly affected in the chc1-5 mutant, further implicating clathrin speci®cally in the Golgi to vacuole transport pathway for carboxypeptidase Y. -
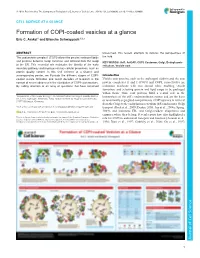
Formation of COPI-Coated Vesicles at a Glance Eric C
© 2018. Published by The Company of Biologists Ltd | Journal of Cell Science (2018) 131, jcs209890. doi:10.1242/jcs.209890 CELL SCIENCE AT A GLANCE Formation of COPI-coated vesicles at a glance Eric C. Arakel1 and Blanche Schwappach1,2,* ABSTRACT unresolved, this review attempts to refocus the perspectives of The coat protein complex I (COPI) allows the precise sorting of lipids the field. and proteins between Golgi cisternae and retrieval from the Golgi KEY WORDS: Arf1, ArfGAP, COPI, Coatomer, Golgi, Endoplasmic to the ER. This essential role maintains the identity of the early reticulum, Vesicle coat secretory pathway and impinges on key cellular processes, such as protein quality control. In this Cell Science at a Glance and accompanying poster, we illustrate the different stages of COPI- Introduction coated vesicle formation and revisit decades of research in the Vesicle coat proteins, such as the archetypal clathrin and the coat context of recent advances in the elucidation of COPI coat structure. protein complexes II and I (COPII and COPI, respectively) are By calling attention to an array of questions that have remained molecular machines with two central roles: enabling vesicle formation, and selecting protein and lipid cargo to be packaged within them. Thus, coat proteins fulfil a central role in the 1Department of Molecular Biology, Universitätsmedizin Göttingen, Humboldtallee homeostasis of the cell’s endomembrane system and are the basis 23, 37073 Göttingen, Germany. 2Max-Planck Institute for Biophysical Chemistry, 37077 Göttingen, Germany. of functionally segregated compartments. COPI operates in retrieval from the Golgi to the endoplasmic reticulum (ER) and in intra-Golgi *Author for correspondence ([email protected]) transport (Beck et al., 2009; Duden, 2003; Lee et al., 2004a; Spang, E.C.A., 0000-0001-7716-7149; B.S., 0000-0003-0225-6432 2009), and maintains ER- and Golgi-resident chaperones and enzymes where they belong. -

ADP-Ribosylation Factor, a Small GTP-Binding Protein, Is Required for Binding of the Coatomer Protein Fl-COP to Golgi Membranes JULIE G
Proc. Natl. Acad. Sci. USA Vol. 89, pp. 6408-6412, July 1992 Biochemistry ADP-ribosylation factor, a small GTP-binding protein, is required for binding of the coatomer protein fl-COP to Golgi membranes JULIE G. DONALDSON*, DAN CASSEL*t, RICHARD A. KAHN*, AND RICHARD D. KLAUSNER* *Cell Biology and Metabolism Branch, National Institute of Child Health and Human Development, and tLaboratory of Biological Chemistry, Division of Cancer Treatment, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892 Communicated by Marc Kirschner, April 20, 1992 (receivedfor review February 11, 1992) ABSTRACT The coatomer is a cytosolic protein complex localized to the Golgi complex, although their functions have that reversibly associates with Golgi membranes and is Impli- not been defined. Distinct among these proteins is the ADP- cated in modulating Golgi membrane transport. The associa- ribosylation factor (ARF), originally identified as a cofactor tion of 13-COP, a component of coatomer, with Golgi mem- required for in vitro cholera toxin-catalyzed ADP- branes is enhanced by guanosine 5'-[v-thioltriphosphate ribosylation of the a subunit of the trimeric GTP-binding (GTP[yS]), a nonhydrolyzable analogue of GTP, and by a protein G, (G,.) (19). ARF is an abundant cytosolic protein mixture of aluminum and fluoride ions (Al/F). Here we show that reversibly associates with Golgi membranes (20, 21). that the ADP-ribosylation factor (ARF) is required for the ARF has been shown to be present on Golgi coated vesicles binding of (-COP. Thus, 13-COP contained in a coatomer generated in the presence of GTP[yS], but it is not a com- fraction that has been resolved from ARF does not bind to Golgi ponent of the cytosolic coatomer (22). -

Mechanisms of Synaptic Plasticity Mediated by Clathrin Adaptor-Protein Complexes 1 and 2 in Mice
Mechanisms of synaptic plasticity mediated by Clathrin Adaptor-protein complexes 1 and 2 in mice Dissertation for the award of the degree “Doctor rerum naturalium” at the Georg-August-University Göttingen within the doctoral program “Molecular Biology of Cells” of the Georg-August University School of Science (GAUSS) Submitted by Ratnakar Mishra Born in Birpur, Bihar, India Göttingen, Germany 2019 1 Members of the Thesis Committee Prof. Dr. Peter Schu Institute for Cellular Biochemistry, (Supervisor and first referee) University Medical Center Göttingen, Germany Dr. Hans Dieter Schmitt Neurobiology, Max Planck Institute (Second referee) for Biophysical Chemistry, Göttingen, Germany Prof. Dr. med. Thomas A. Bayer Division of Molecular Psychiatry, University Medical Center, Göttingen, Germany Additional Members of the Examination Board Prof. Dr. Silvio O. Rizzoli Department of Neuro-and Sensory Physiology, University Medical Center Göttingen, Germany Dr. Roland Dosch Institute of Developmental Biochemistry, University Medical Center Göttingen, Germany Prof. Dr. med. Martin Oppermann Institute of Cellular and Molecular Immunology, University Medical Center, Göttingen, Germany Date of oral examination: 14th may 2019 2 Table of Contents List of abbreviations ................................................................................. 5 Abstract ................................................................................................... 7 Chapter 1: Introduction ............................................................................ -

ADP-Ribosylation Factor and Coatomer Couple Fusion to Vesicle Budding Zvulun Elazar,* Lelio Orci,T Joachim Ostermann,* Myl~Nc Amherdt,* Gary Tanigawa,* and James E
ADP-Ribosylation Factor and Coatomer Couple Fusion to Vesicle Budding Zvulun Elazar,* Lelio Orci,t Joachim Ostermann,* Myl~nc Amherdt,* Gary Tanigawa,* and James E. Rothman* * Program in Cellular Biochemistry and Biophysics, Memorial Sloan Kettering Cancer Center, New York 10021; and ¢Institute of Histology and Embryology, University of Geneva Medical School, 1211 Geneva 4, Switzerland Abstract. The coat proteins required for budding pair directly without an intervening vesicle. Coupling COP-coated vesicles from Golgi membranes, coatomer may therefore result from the sequestration of fuso- and ADP-ribosylation factor (ARF) protein, are shown genic membrane proteins into assembling coated vesi- to be required to reconstitute the orderly process of cles that are only exposed when the coat is removed transport between Golgi cisternae in which fusion of after budding is complete. This mechanism of cou- transport vesicles begins only after budding ends. pling explains the phenomenon of "retrograde transport" When either coat protein is omitted, fusion is uncou- triggered by uncouplers such as the drug brefeldin A. pled from budding-donor and acceptor compartments ow is membrane fusion coupled to vesicle budding? Orci et al., 1989). Purification of COP-coated vesicles (Mal- A transport vesicle must fuse with its target only hotra et al., 1989; Serafini et al., 1991a,b) revealed that their H after its budding from the parental membrane is coats consist of a small GTP-binding protein (ADP-ribosyla- completed. Otherwise, the various membrane-bound com- tion factor, ARF) ~ and a complex of seven distinct proteins partments connected by vesicle shuttles would fuse and the termed coatomer (Waters et al., 1992a; Stenbeck et al., topological organization of the endomembrane system in 1993), whose subunits are or, B, B', 3', ~, e, and ~'-COPs. -

Identification of Potential Key Genes and Pathway Linked with Sporadic Creutzfeldt-Jakob Disease Based on Integrated Bioinformatics Analyses
medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Identification of potential key genes and pathway linked with sporadic Creutzfeldt-Jakob disease based on integrated bioinformatics analyses Basavaraj Vastrad1, Chanabasayya Vastrad*2 , Iranna Kotturshetti 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karanataka, India. 3. Department of Ayurveda, Rajiv Gandhi Education Society`s Ayurvedic Medical College, Ron, Karnataka 562209, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Abstract Sporadic Creutzfeldt-Jakob disease (sCJD) is neurodegenerative disease also called prion disease linked with poor prognosis. The aim of the current study was to illuminate the underlying molecular mechanisms of sCJD. The mRNA microarray dataset GSE124571 was downloaded from the Gene Expression Omnibus database. Differentially expressed genes (DEGs) were screened. -

Dynamin Action at the Final Stage of Clathrin- Mediated Endocytosis
Dynamin Action at the Final Stage of Clathrin- Mediated Endocytosis Ning Fang ( [email protected] ) Georgia State University https://orcid.org/0000-0003-4710-0984 Xiaodong Cheng Georgia State University Kuangcai Chen Georgia State University https://orcid.org/0000-0002-9321-9225 Bin Dong Georgia State University https://orcid.org/0000-0002-3196-0712 Meek Yang Georgia State University Seth Filbrun Georgia State University Yong Myoung Georgia State University Teng-Xiang Huang Georgia State University Yan Gu The Bristol-Myers Squibb Company Gufeng Wang Georgia State University Article Keywords: Dynamin, clathrin-mediated endocytosis, scission, vesicle ssion Posted Date: February 5th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-134570/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/25 Abstract Dynamin plays an important role in clathrin-mediated endocytosis by cutting the neck of nascent vesicles from the cell membrane. Gold nanorods were used as imaging probes to observe dynamin action on cargo vesicles during live endocytosis events. Invariant is that at the peak of dynamin accumulation, the cargo-containing vesicle always gives abrupt, right-handed rotations that nishes in a short time (~ 0.28 s). The large and quick twist, herein named the super twist, is the result of the coordinated dynamin helix action upon GTP hydrolysis. After the super twist, the rotational freedom of the vesicle drastically increases, accompanied with simultaneous or delayed translational movement, indicating that it detaches from the cell membrane. These observations suggest that dynamin-mediated scission at the nal stage involves a large torque generated by coordinated actions of multiple dynamins in the helix, which is the main driving force for scission. -

The Structure of the COPI Coat Determined Within the Cell
RESEARCH ADVANCE The structure of the COPI coat determined within the cell Yury S Bykov1,2, Miroslava Schaffer3, Svetlana O Dodonova1†, Sahradha Albert3, Ju¨ rgen M Plitzko3, Wolfgang Baumeister3*, Benjamin D Engel3*, John AG Briggs1,2* 1Structural and Computational Biology Unit, European Molecular Biology Laboratory, Heidelberg, Germany; 2Structural Studies Division, MRC Laboratory of Molecular Biology, Cambridge, United Kingdom; 3Department of Molecular Structural Biology, Max Planck Institute of Biochemistry, Martinsried, Germany Abstract COPI-coated vesicles mediate trafficking within the Golgi apparatus and from the Golgi to the endoplasmic reticulum. The structures of membrane protein coats, including COPI, have been extensively studied with in vitro reconstitution systems using purified components. Previously we have determined a complete structural model of the in vitro reconstituted COPI coat (Dodonova et al., 2017). Here, we applied cryo-focused ion beam milling, cryo-electron tomography and subtomogram averaging to determine the native structure of the COPI coat within vitrified Chlamydomonas reinhardtii cells. The native algal structure resembles the in vitro mammalian structure, but additionally reveals cargo bound beneath b’–COP. We find that all coat components *For correspondence: disassemble simultaneously and relatively rapidly after budding. Structural analysis in situ, [email protected] (WB); maintaining Golgi topology, shows that vesicles change their size, membrane thickness, and cargo [email protected] (BDE); content as they progress from cis to trans, but the structure of the coat machinery remains [email protected] constant. (JAGB) DOI: https://doi.org/10.7554/eLife.32493.001 Present address: †Department of Molecular Biology, Max Planck Institute for Biophysical Chemistry, Go¨ ttingen, Germany Introduction Vesicular transport between cellular compartments is a fundamental mechanism of eukaryotic cells. -

Membrane Remodeling in Clathrin-Mediated Endocytosis Volker Haucke1,2,* and Michael M
© 2018. Published by The Company of Biologists Ltd | Journal of Cell Science (2018) 131, jcs216812. doi:10.1242/jcs.216812 REVIEW Membrane remodeling in clathrin-mediated endocytosis Volker Haucke1,2,* and Michael M. Kozlov3,* ABSTRACT membrane fission and vesicle uncoating to eventually allow fusion Clathrin-mediated endocytosis is an essential cellular mechanism by of the nascent endocytic vesicle with endosomes (Box 1). which all eukaryotic cells regulate their plasma membrane composition In spite of important advances in the characterization of these to control processes ranging from cell signaling to adhesion, migration factors, key questions regarding the endocytic process remain. and morphogenesis. The formation of endocytic vesicles and For example, different models have been proposed regarding the tubules involves extensive protein-mediated remodeling of the initiation of clathrin-coated pit (CCP) formation and the onset of plasma membrane that is organized in space and time by protein– membrane curvature acquisition. Moreover, knowledge regarding protein and protein–phospholipid interactions. Recent studies the nanoscale distribution of endocytic proteins within the bilayer combining high-resolution imaging with genetic manipulations of plane during vesicle formation and the mechanisms that guide the endocytic machinery and with theoretical approaches have led their orchestrated assembly and disassembly is only now beginning to novel multifaceted phenomenological data of the temporal and to emerge. Finally, our understanding of the nature and dynamics spatial organization of the endocytic reaction. This gave rise to of membrane phospholipids (Box 2) and their associations with various – often conflicting – models as to how endocytic proteins endocytic proteins during endocytic membrane remodeling and their association with lipids regulate the endocytic protein remains limited. -

Cryo-Tomography of Coat Complexes ISSN 2059-7983
research papers Visualizing membrane trafficking through the electron microscope: cryo-tomography of coat complexes ISSN 2059-7983 Evgenia A. Markova and Giulia Zanetti* Institute of Structural and Molecular Biology, Birkbeck College, Malet Street, London WC1E 7HX, England. Received 8 March 2019 *Correspondence e-mail: [email protected] Accepted 12 April 2019 Coat proteins mediate vesicular transport between intracellular compartments, Keywords: cryo-electron tomography; which is essential for the distribution of molecules within the eukaryotic cell. three-dimensional reconstruction; coat proteins; The global arrangement of coat proteins on the membrane is key to their vesicular transport; COPII; subtomogram function, and cryo-electron tomography and subtomogram averaging have been averaging. used to study membrane-bound coat proteins, providing crucial structural insight. This review outlines a workflow for the structural elucidation of coat proteins, incorporating recent developments in the collection and processing of cryo-electron tomography data. Recent work on coat protein I, coat protein II and retromer performed on in vitro reconstitutions or in situ is summarized. These studies have answered long-standing questions regarding the mechanisms of membrane binding, polymerization and assembly regulation of coat proteins. 1. Introduction Recent advances in cryo-electron microscopy (cryo-EM) have enabled numerous high-resolution studies which have addressed long-standing structural biology questions. The large body of cryo-EM work carried out for protein structure characterization has utilized single-particle electron micro- scopy (Cheng, 2018). Recently, developments in hardware, data collection and data processing have placed cryo-electron tomography (cryo-ET) and subtomogram averaging (STA) at the forefront of structural studies of repetitive assemblies, reaching resolutions comparable to those of single-particle EM (Schur et al., 2016; Wan et al., 2017; Hutchings et al., 2018; Dodonova et al., 2017; Himes & Zhang, 2018).