Pharmaceutical Composition for Preventing Or Treating Diabetes, Containing Zinc Salt, Cyclo-Hispro and Antidiabetic Drug As Active Ingredients
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Fixed Dose Combination of Voglibose & Repaglinide in the Management
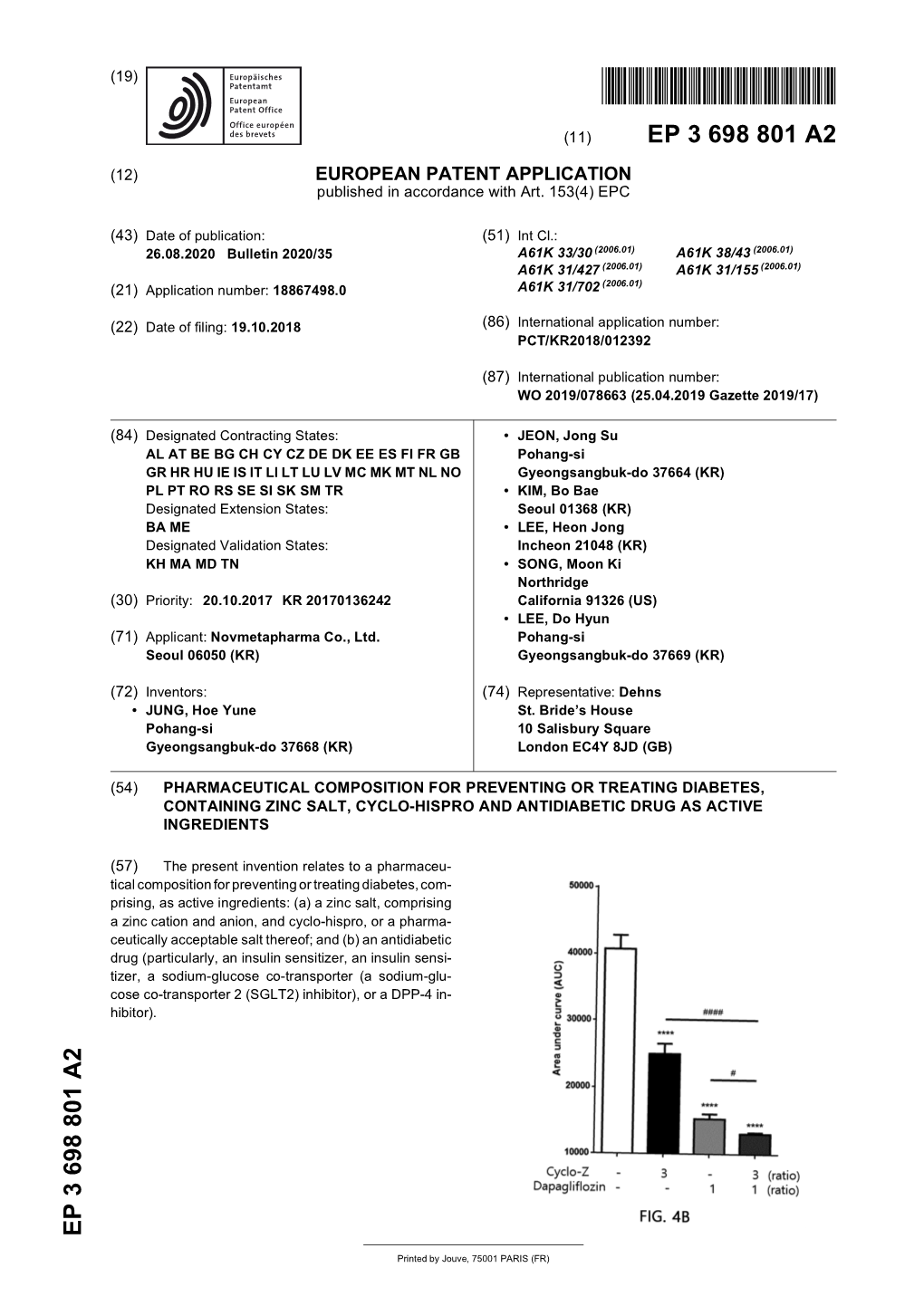
Clinical Group Global Journal of Obesity, Diabetes and Metabolic Syndrome ISSN: 2455-8583 DOI CC By Arif Faruqui* Research Article Medical Services Department, Medley Pharmaceuticals Limited, Mumbai, India Fixed Dose Combination of Voglibose Dates: Received: 09 August, 2017; Accepted: 26 August, 2017; Published: 28 August, 2017 & Repaglinide in the Management of *Corresponding author: Arif Faruqui, Medical Services Department, Medley Pharmaceuticals Limited, Mumbai, Postprandial Hyperglycemia in Indian India, E-mail: Subjects Keywords: T2DM; Postprandial hyperglycemia; Voglibose; Repaglinide https://www.peertechz.com Abstract To evaluate the effi cacy and tolerability of fi xed dose combination of voglibose and repaglinide in postprandial hyperglycemia (PPHG) in Indian subjects. A non-randomized, open labeled, non-comparative, single-centric, study was conducted in total of 20 type 2 diabetes mellitus (T2DM) patients (9 men and 11 women, mean age 69.07 ± 3.495 years). Each patient was administered a fi xed dose combination of voglibose 0.3mg and repaglinide 0.5/1mg three times a day, just before each meal for 30 days. Fasting blood glucose (FBG) and postprandial blood glucose (PPBG) was performed at baseline and at the end of study as assessment tools. At the end of study data was extractable only in 15 patients because 5 patient dropped out as they did not turn up for follow up at day 30.Postprandial blood glucose reduced signifi cantly {-61.67mg/dl (95% confi dence interval -76.79 to -46.54 p<0.0001)} from baseline at day 30. Also a signifi cant reduction (p<0.0001) was found with FBG {-42.13mg/dl (Confi dence interval -61.25 to -23.02 P value <0.0001)} at end of day 30. -

Computationally Guided Design of Dipeptidyl Peptidase-4 Inhibitors Lauren C
bioRxiv preprint doi: https://doi.org/10.1101/772137; this version posted September 18, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Computationally Guided Design of Dipeptidyl Peptidase-4 Inhibitors Lauren C. Reynolds1, Morgan P. Connolly2,3, Justin B. Siegel1,2,4* 1. Department of Chemistry, University of California Davis, Davis, California, United States of America 2. Genome Center, University of California Davis, Davis, California, United States of America 3. Microbiology Graduate Group, University of California Davis, Davis, California, United States of America 4. Department of Biochemistry and Molecular Medicine, University of California Davis, Davis, California, United States of America ABSTRACT: The Type 2 Diabetes Mellitus (T2DM) epidemic undoubtedly creates a need for the development of new phar- maceuticals. With the goal of generating new therapeutics for this disease, computational studies were conducted to design novel dipeptidyl peptidase-4 (DPP-4) inhibitors. Two candidates, generated by chemical intuition-driven design and bioiso- steric replacement, were found to have better docking scores than anagliptin, a currently available diabetes medication. INTRODUCTION debate in recent years for other gliptins as well. A recent Type 2 Diabetes Mellitus (T2DM) is dubbed the meta-analysis seems to suggest a possible increased pan- “epidemic of the 21st century”— affecting millions of pa- creatitis risk for sitagliptin -

(Trulicity) Pen and the Semaglutide (Ozempic) Pen
Protocol H9X-MC-B021(b) Crossover Study Comparing Dulaglutide (Trulicity) Pen and the Semaglutide (Ozempic) Pen NCT03724981 Approval Date: 30-Nov-2018 H9X-MC-B021(b) Clinical Protocol Page 1 Protocol H9X-MC-B021(b): Crossover Study Comparing the Dulaglutide (Trulicity) Pen and the Semaglutide (Ozempic) Pen Confidential Information The information contained in this document is confidential and is intended for the use of clinical investigators. It is the property of Eli Lilly and Company or its subsidiaries and should not be copied by or distributed to persons not involved in the clinical investigation of dulaglutide (LY2189265), unless such persons are bound by a confidentiality agreement with Eli Lilly and Company or its subsidiaries. Note to Regulatory Authorities: This document may contain protected personal data and/or commercially confidential information exempt from public disclosure. Eli Lilly and Company requests consultation regarding release/redaction prior to any public release. In the United States, this document is subject to Freedom of Information Act (FOIA) Exemption 4 and may not be reproduced or otherwise disseminated without the written approval of Eli Lilly and Company or its subsidiaries. Dulaglutide (LY2189265) Eli Lilly and Company Indianapolis, Indiana USA 46285 Protocol Electronically Signed and Approved by Lilly on 07 Aug 2018 Amendment (a) Electronically Signed and Approved by Lilly on 11 Sep 2018 Amendment (b) Electronically Signed and Approved by Lilly on date below Approval Date: 30-Nov-2018 GMT LY2189265 H9X-MC-B021(b) Clinical Protocol Page 2 Table of Contents Section Page Protocol H9X-MC-B021(b): Crossover Study Comparing the Dulaglutide (Trulicity) Pen and the Semaglutide (Ozempic) Pen ............................................................1 Table of Contents........................................................................................................................2 1. -

Classification of Medicinal Drugs and Driving: Co-Ordination and Synthesis Report
Project No. TREN-05-FP6TR-S07.61320-518404-DRUID DRUID Driving under the Influence of Drugs, Alcohol and Medicines Integrated Project 1.6. Sustainable Development, Global Change and Ecosystem 1.6.2: Sustainable Surface Transport 6th Framework Programme Deliverable 4.4.1 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Due date of deliverable: 21.07.2011 Actual submission date: 21.07.2011 Revision date: 21.07.2011 Start date of project: 15.10.2006 Duration: 48 months Organisation name of lead contractor for this deliverable: UVA Revision 0.0 Project co-funded by the European Commission within the Sixth Framework Programme (2002-2006) Dissemination Level PU Public PP Restricted to other programme participants (including the Commission x Services) RE Restricted to a group specified by the consortium (including the Commission Services) CO Confidential, only for members of the consortium (including the Commission Services) DRUID 6th Framework Programme Deliverable D.4.4.1 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Page 1 of 243 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Authors Trinidad Gómez-Talegón, Inmaculada Fierro, M. Carmen Del Río, F. Javier Álvarez (UVa, University of Valladolid, Spain) Partners - Silvia Ravera, Susana Monteiro, Han de Gier (RUGPha, University of Groningen, the Netherlands) - Gertrude Van der Linden, Sara-Ann Legrand, Kristof Pil, Alain Verstraete (UGent, Ghent University, Belgium) - Michel Mallaret, Charles Mercier-Guyon, Isabelle Mercier-Guyon (UGren, University of Grenoble, Centre Regional de Pharmacovigilance, France) - Katerina Touliou (CERT-HIT, Centre for Research and Technology Hellas, Greece) - Michael Hei βing (BASt, Bundesanstalt für Straßenwesen, Germany). -

Summary of Investigation Results Sodium-Glucose Co-Transporter 2 (SGLT2) Inhibitors
Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this English translation, the former shall prevail. Summary of investigation results Sodium-glucose co-transporter 2 (SGLT2) inhibitors September 15, 2015 Non-proprietary name a. Canagliflozin hydrate b. Dapagliflozin propylene glycolate hydrate c. Empagliflozin d. Ipragliflozin L-proline e. Luseogliflozin hydrate f. Tofogliflozin hydrate Brand name (Marketing authorization holder) a. Canaglu Tablets 100 mg (Mitsubishi Tanabe Pharma Corporation) b. Forxiga Tablets 5 mg and 10 mg (AstraZeneca K.K.) c. Jardiance Tablets 10 mg and 25 mg (Nippon Boehringer Ingelheim Co., Ltd.) d. Suglat Tablets 25 mg and 50 mg (Astellas Pharma Inc.) e. Lusefi Tablets 2.5 mg and 5 mg (Taisho Pharmaceutical Co., Ltd.) f. Apleway Tablets 20 mg (Sanofi K.K.) and Deberza Tablets 20 mg (Kowa Company, Ltd.) Indications Type 2 diabetes mellitus Summary of revision 1. Precautions regarding ketoacidosis should be added in the Important Precautions section for the above products from a to f. 2. “Ketoacidosis” should be newly added in the Clinically significant adverse reaction section for the above products from a to f. 3. “Sepsis” should be added to the “Pyelonephritis” subsection in the Important Precautions section for the above products from a to f. Pharmaceuticals and Medical Devices Agency Office of Safety I 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan E-mail: [email protected] Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material for the convenience of users. -

Modifications to the Harmonized Tariff Schedule of the United States to Implement Changes to the Pharmaceutical Appendix
United States International Trade Commission Modifications to the Harmonized Tariff Schedule of the United States to Implement Changes to the Pharmaceutical Appendix USITC Publication 4208 December 2010 U.S. International Trade Commission COMMISSIONERS Deanna Tanner Okun, Chairman Irving A. Williamson, Vice Chairman Charlotte R. Lane Daniel R. Pearson Shara L. Aranoff Dean A. Pinkert Address all communications to Secretary to the Commission United States International Trade Commission Washington, DC 20436 U.S. International Trade Commission Washington, DC 20436 www.usitc.gov Modifications to the Harmonized Tariff Schedule of the United States to Implement Changes to the Pharmaceutical Appendix Publication 4208 December 2010 (This page is intentionally blank) Pursuant to the letter of request from the United States Trade Representative of December 15, 2010, set forth at the end of this publication, and pursuant to section 1207(a) of the Omnibus Trade and Competitiveness Act, the United States International Trade Commission is publishing the following modifications to the Harmonized Tariff Schedule of the United States (HTS) to implement changes to the Pharmaceutical Appendix, effective on January 1, 2011. Table 1 International Nonproprietary Name (INN) products proposed for addition to the Pharmaceutical Appendix to the Harmonized Tariff Schedule INN CAS Number Abagovomab 792921-10-9 Aclidinium Bromide 320345-99-1 Aderbasib 791828-58-5 Adipiplon 840486-93-3 Adoprazine 222551-17-9 Afimoxifene 68392-35-8 Aflibercept 862111-32-8 Agatolimod -

200 Adverse Drug Reactions in a Southern Hospital in Taiwan 201
200 Adverse Drug Reactions in a southern hospital in Taiwan Yuhong Lin1 1Kaohsiung Veterans General Hospital Tainan Branch, Tainan City, Taiwan Objective: The aim of this study was to evaluate the adverse drug reactions (ADRs) reporting in a southern hospital in Taiwan. Methods: It was a retrospective study of ADRs reporting in 2016. We analysed ADRs which contain gender and age, occurring sources, Anatomical Therapeutic Chemical (ATC) classification of suspected drugs, management of ADRs and so on. Results: The study collected eighty-nine ADRs reported. Most of ADRs reported were occurring in outpatient department (87.6%). The average age of ADRs reported was 67.6 years. Majority of all ADRs reported were females (55.1%). According to ATC classification system, the major classification of suspected drugs were Sensory organs (32.6%). Among the adverse reactions, Dermatologic Effects (37.1%) were the major type of ADRs. Also, the major management of ADRs was to stop using the current suspected drug (46.1%). Conclusion: Certainly, ADRs reporting are still a very important information to healthcare professionals. As a result, we put all information of ADRs reported into medical system in our hospital and it will improve the safety of medication use. In case of prescribe the suspected medicines, it can remind doctor to think of information about patient's ADRs reported. No drugs is administered without risk. Therefore, all healthcare professionals should have a responsibility to their patients, who themselves are becoming more aware of problems -

List of Union Reference Dates A
Active substance name (INN) EU DLP BfArM / BAH DLP yearly PSUR 6-month-PSUR yearly PSUR bis DLP (List of Union PSUR Submission Reference Dates and Frequency (List of Union Frequency of Reference Dates and submission of Periodic Frequency of submission of Safety Update Reports, Periodic Safety Update 30 Nov. 2012) Reports, 30 Nov. -

CDR Clinical Review Report for Soliqua
CADTH COMMON DRUG REVIEW Clinical Review Report Insulin glargine and lixisenatide injection (Soliqua) (Sanofi-Aventis) Indication: adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus inadequately controlled on basal insulin (less than 60 units daily) alone or in combination with metformin. Service Line: CADTH Common Drug Review Version: Final (with redactions) Publication Date: January 2019 Report Length: 118 Pages Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services. While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. -

Metformin Does Not Affect Cancer Risk: a Cohort Study in the U.K
2522 Diabetes Care Volume 37, September 2014 Metformin Does Not Affect Cancer Konstantinos K. Tsilidis,1,2 Despoina Capothanassi,1 Naomi E. Allen,3 Risk: A Cohort Study in the U.K. Evangelos C. Rizos,4 David S. Lopez,5 Karin van Veldhoven,6–8 Clinical Practice Research Carlotta Sacerdote,7 Deborah Ashby,9 Paolo Vineis,6,7 Ioanna Tzoulaki,1,6 and Datalink Analyzed Like an John P.A. Ioannidis10 Intention-to-Treat Trial Diabetes Care 2014;37:2522–2532 | DOI: 10.2337/dc14-0584 OBJECTIVE Meta-analyses of epidemiologic studies have suggested that metformin may re- 1Department of Hygiene and Epidemiology, Uni- duce cancer incidence, but randomized controlled trials did not support this versity of Ioannina School of Medicine, Ioannina, hypothesis. Greece 2Cancer Epidemiology Unit, University of Oxford, EPIDEMIOLOGY/HEALTH SERVICES RESEARCH RESEARCH DESIGN AND METHODS Oxford, U.K. 3 A retrospective cohort study, Clinical Practice Research Datalink, was designed to Clinical Trial Service Unit, University of Oxford, Oxford, U.K. investigate the association between use of metformin compared with other anti- 4Lipid Disorders Clinic, Department of Internal diabetes medications and cancer risk by emulating an intention-to-treat analysis Medicine, University Hospital of Ioannina, Ioan- as in a trial. A total of 95,820 participants with type 2 diabetes who started taking nina, Greece 5 metformin and other oral antidiabetes medications within 12 months of their Division of Epidemiology, University of Texas School of Public Health, Houston, TX diagnosis (initiators) were followed up for first incident cancer diagnosis without 6Department of Epidemiology and Biostatistics, regard to any subsequent changes in pharmacotherapy. -

Exendin-4 Peptide Analogues As Dual Glp-1/Gip Receptor Agonists
(19) TZZ¥ZZ_Z_T (11) EP 3 080 150 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07K 14/575 (2006.01) C07K 14/605 (2006.01) 01.08.2018 Bulletin 2018/31 (86) International application number: (21) Application number: 14828149.6 PCT/EP2014/077336 (22) Date of filing: 11.12.2014 (87) International publication number: WO 2015/086728 (18.06.2015 Gazette 2015/24) (54) EXENDIN-4 PEPTIDE ANALOGUES AS DUAL GLP-1/GIP RECEPTOR AGONISTS EXENDIN-4-PEPTIDANALOGA ALS DUALE GLP-1/GIP-REZEPTORAGONISTEN ANALOGUES PEPTIDIQUES D’EXENDINE-4 EN TANT QU’AGONISTES DU RÉCEPTEUR GIP/GLP-1 DOUBLE (84) Designated Contracting States: (74) Representative: Weickmann & Weickmann AL AT BE BG CH CY CZ DE DK EE ES FI FR GB PartmbB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO Postfach 860 820 PL PT RO RS SE SI SK SM TR 81635 München (DE) (30) Priority: 13.12.2013 EP 13306712 (56) References cited: WO-A1-2012/167744 WO-A2-2009/035540 (43) Date of publication of application: US-A1- 2011 166 062 US-A1- 2012 196 802 19.10.2016 Bulletin 2016/42 • JOHN L. KRSTENANSKY ET AL: "Importance of (73) Proprietor: SANOFI the 10-13 region of glucagon for its receptor 75008 Paris (FR) interactionsand activation of adenylate cyclase", BIOCHEMISTRY, vol. 25, no. 13, 1 July 1986 (72) Inventors: (1986-07-01), pages 3833-3839, XP055117918, • KADEREIT, Dieter ISSN: 0006-2960, DOI: 10.1021/bi00361a014 65926 Frankfurt am Main (DE) • VICTOR A. -

Sulfonylurea Review
Human Journals Review Article February 2018 Vol.:11, Issue:3 © All rights are reserved by Farah Yousef et al. Sulfonylurea Review Keywords: Type II diabetes, Sulfonylurea, Glimipiride, Glybu- ride, Structure Activity Relationship. ABSTRACT Farah Yousef*1, Oussama Mansour2, Jehad Herbali3 Diabetes Mellitus is a chronic disease represented with high 1 Ph.D. candidate in pharmaceutical sciences, Damas- glucose blood levels. Although sulfonylurea compounds are the cus University, Damascus, Syria. second preferred drug to treat Type II Diabetes (TYIID), they are still the most used agents due to their lower cost and as a 2 Assistant Professor in pharmaceutical chimestry, Ti- mono-dosing. Literature divides these compounds according to st nd rd shreen University, Lattakia, Syria their discovery into 1 , 2 , 3 generations. However, only six sulfonylurea compounds are now available for use in the United 3 Assistant Professor in pharmaceutical chimestry, Da- States: Chlorpropamide, Glimepiride, Glipizide, Glyburide, mascus University, Damascus, Syria. Tolazamide, and Tolbutamide. They function by increasing Submission: 24 January 2018 insulin secretion from pancreatic beta cells. Their main active site is ATP sensitive potassium ion channels; Kir 6.2\SUR1; Accepted: 29 January 2018 Published: 28 February 2018 Potassium Inward Rectifier ion channel 6.2\ Sulfonylurea re- ceptor 1. They are sulfonamide derivatives. However, research- ers have declared that sulfonylurea moiety is not the only one responsible for this group efficacy. It has been known that sud- den and acute hypoglycemia incidences and weight gain are the www.ijppr.humanjournals.com two most common adverse effects TYIID the patient might face during treatment with sulfonylurea agents. This review indi- cates the historical development of sulfonylurea and the differ- ences among this group members.