An Introduction to Thermodynamics Applied to Organic Rankine Cycles
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
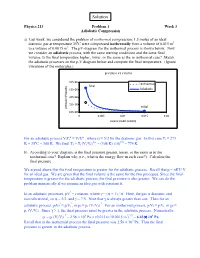
3-1 Adiabatic Compression
Solution Physics 213 Problem 1 Week 3 Adiabatic Compression a) Last week, we considered the problem of isothermal compression: 1.5 moles of an ideal diatomic gas at temperature 35oC were compressed isothermally from a volume of 0.015 m3 to a volume of 0.0015 m3. The pV-diagram for the isothermal process is shown below. Now we consider an adiabatic process, with the same starting conditions and the same final volume. Is the final temperature higher, lower, or the same as the in isothermal case? Sketch the adiabatic processes on the p-V diagram below and compute the final temperature. (Ignore vibrations of the molecules.) α α For an adiabatic process ViTi = VfTf , where α = 5/2 for the diatomic gas. In this case Ti = 273 o 1/α 2/5 K + 35 C = 308 K. We find Tf = Ti (Vi/Vf) = (308 K) (10) = 774 K. b) According to your diagram, is the final pressure greater, lesser, or the same as in the isothermal case? Explain why (i.e., what is the energy flow in each case?). Calculate the final pressure. We argued above that the final temperature is greater for the adiabatic process. Recall that p = nRT/ V for an ideal gas. We are given that the final volume is the same for the two processes. Since the final temperature is greater for the adiabatic process, the final pressure is also greater. We can do the problem numerically if we assume an idea gas with constant α. γ In an adiabatic processes, pV = constant, where γ = (α + 1) / α. -

Isobaric Expansion Engines: New Opportunities in Energy Conversion for Heat Engines, Pumps and Compressors
energies Concept Paper Isobaric Expansion Engines: New Opportunities in Energy Conversion for Heat Engines, Pumps and Compressors Maxim Glushenkov 1, Alexander Kronberg 1,*, Torben Knoke 2 and Eugeny Y. Kenig 2,3 1 Encontech B.V. ET/TE, P.O. Box 217, 7500 AE Enschede, The Netherlands; [email protected] 2 Chair of Fluid Process Engineering, Paderborn University, Pohlweg 55, 33098 Paderborn, Germany; [email protected] (T.K.); [email protected] (E.Y.K.) 3 Chair of Thermodynamics and Heat Engines, Gubkin Russian State University of Oil and Gas, Leninsky Prospekt 65, Moscow 119991, Russia * Correspondence: [email protected] or [email protected]; Tel.: +31-53-489-1088 Received: 12 December 2017; Accepted: 4 January 2018; Published: 8 January 2018 Abstract: Isobaric expansion (IE) engines are a very uncommon type of heat-to-mechanical-power converters, radically different from all well-known heat engines. Useful work is extracted during an isobaric expansion process, i.e., without a polytropic gas/vapour expansion accompanied by a pressure decrease typical of state-of-the-art piston engines, turbines, etc. This distinctive feature permits isobaric expansion machines to serve as very simple and inexpensive heat-driven pumps and compressors as well as heat-to-shaft-power converters with desired speed/torque. Commercial application of such machines, however, is scarce, mainly due to a low efficiency. This article aims to revive the long-known concept by proposing important modifications to make IE machines competitive and cost-effective alternatives to state-of-the-art heat conversion technologies. Experimental and theoretical results supporting the isobaric expansion technology are presented and promising potential applications, including emerging power generation methods, are discussed. -

Novel Hot Air Engine and Its Mathematical Model – Experimental Measurements and Numerical Analysis
POLLACK PERIODICA An International Journal for Engineering and Information Sciences DOI: 10.1556/606.2019.14.1.5 Vol. 14, No. 1, pp. 47–58 (2019) www.akademiai.com NOVEL HOT AIR ENGINE AND ITS MATHEMATICAL MODEL – EXPERIMENTAL MEASUREMENTS AND NUMERICAL ANALYSIS 1 Gyula KRAMER, 2 Gabor SZEPESI *, 3 Zoltán SIMÉNFALVI 1,2,3 Department of Chemical Machinery, Institute of Energy and Chemical Machinery University of Miskolc, Miskolc-Egyetemváros 3515, Hungary e-mail: [email protected], [email protected], [email protected] Received 11 December 2017; accepted 25 June 2018 Abstract: In the relevant literature there are many types of heat engines. One of those is the group of the so called hot air engines. This paper introduces their world, also introduces the new kind of machine that was developed and built at Department of Chemical Machinery, Institute of Energy and Chemical Machinery, University of Miskolc. Emphasizing the novelty of construction and the working principle are explained. Also the mathematical model of this new engine was prepared and compared to the real model of engine. Keywords: Hot, Air, Engine, Mathematical model 1. Introduction There are three types of volumetric heat engines: the internal combustion engines; steam engines; and hot air engines. The first one is well known, because it is on zenith nowadays. The steam machines are also well known, because their time has just passed, even the elder ones could see those in use. But the hot air engines are forgotten. Our aim is to consider that one. The history of hot air engines is 200 years old. -

High Efficiency and Large-Scale Subsurface Energy Storage with CO2
PROCEEDINGS, 43rd Workshop on Geothermal Reservoir Engineering Stanford University, Stanford, California, February 12-14, 2018 SGP-TR-213 High Efficiency and Large-scale Subsurface Energy Storage with CO2 Mark R. Fleming1, Benjamin M. Adams2, Jimmy B. Randolph1,3, Jonathan D. Ogland-Hand4, Thomas H. Kuehn1, Thomas A. Buscheck3, Jeffrey M. Bielicki4, Martin O. Saar*1,2 1University of Minnesota, Minneapolis, MN, USA; 2ETH-Zurich, Zurich, Switzerland; 3TerraCOH Inc., Minneapolis, MN, USA; 4The Ohio State University, Columbus, OH, USA; 5Lawrence Livermore National Laboratory, Livermore, CA, USA. *Corresponding Author: [email protected] Keywords: geothermal energy, multi-level geothermal systems, sedimentary basin, carbon dioxide, electricity generation, energy storage, power plot, CPG ABSTRACT Storing large amounts of intermittently produced solar or wind power for later, when there is a lack of sunlight or wind, is one of society’s biggest challenges when attempting to decarbonize energy systems. Traditional energy storage technologies tend to suffer from relatively low efficiencies, severe environmental concerns, and limited scale both in capacity and time. Subsurface energy storage can solve the drawbacks of many other energy storage approaches, as it can be large scale in capacity and time, environmentally benign, and highly efficient. When CO2 is used as the (pressure) energy storage medium in reservoirs underneath caprocks at depths of at least ~1 km (to ensure the CO2 is in its supercritical state), the energy generated after the energy storage operation can be greater than the energy stored. This is possible if reservoir temperatures and CO2 storage durations combine to result in more geothermal energy input into the CO2 at depth than what the CO2 pumps at the surface (and other machinery) consume. -

Section 15-6: Thermodynamic Cycles
Answer to Essential Question 15.5: The ideal gas law tells us that temperature is proportional to PV. for state 2 in both processes we are considering, so the temperature in state 2 is the same in both cases. , and all three factors on the right-hand side are the same for the two processes, so the change in internal energy is the same (+360 J, in fact). Because the gas does no work in the isochoric process, and a positive amount of work in the isobaric process, the First Law tells us that more heat is required for the isobaric process (+600 J versus +360 J). 15-6 Thermodynamic Cycles Many devices, such as car engines and refrigerators, involve taking a thermodynamic system through a series of processes before returning the system to its initial state. Such a cycle allows the system to do work (e.g., to move a car) or to have work done on it so the system can do something useful (e.g., removing heat from a fridge). Let’s investigate this idea. EXPLORATION 15.6 – Investigate a thermodynamic cycle One cycle of a monatomic ideal gas system is represented by the series of four processes in Figure 15.15. The process taking the system from state 4 to state 1 is an isothermal compression at a temperature of 400 K. Complete Table 15.1 to find Q, W, and for each process, and for the entire cycle. Process Special process? Q (J) W (J) (J) 1 ! 2 No +1360 2 ! 3 Isobaric 3 ! 4 Isochoric 0 4 ! 1 Isothermal 0 Entire Cycle No 0 Table 15.1: Table to be filled in to analyze the cycle. -

Basic Thermodynamics-17ME33.Pdf
Module -1 Fundamental Concepts & Definitions & Work and Heat MODULE 1 Fundamental Concepts & Definitions Thermodynamics definition and scope, Microscopic and Macroscopic approaches. Some practical applications of engineering thermodynamic Systems, Characteristics of system boundary and control surface, examples. Thermodynamic properties; definition and units, intensive and extensive properties. Thermodynamic state, state point, state diagram, path and process, quasi-static process, cyclic and non-cyclic processes. Thermodynamic equilibrium; definition, mechanical equilibrium; diathermic wall, thermal equilibrium, chemical equilibrium, Zeroth law of thermodynamics, Temperature; concepts, scales, fixed points and measurements. Work and Heat Mechanics, definition of work and its limitations. Thermodynamic definition of work; examples, sign convention. Displacement work; as a part of a system boundary, as a whole of a system boundary, expressions for displacement work in various processes through p-v diagrams. Shaft work; Electrical work. Other types of work. Heat; definition, units and sign convention. 10 Hours 1st Hour Brain storming session on subject topics Thermodynamics definition and scope, Microscopic and Macroscopic approaches. Some practical applications of engineering thermodynamic Systems 2nd Hour Characteristics of system boundary and control surface, examples. Thermodynamic properties; definition and units, intensive and extensive properties. 3rd Hour Thermodynamic state, state point, state diagram, path and process, quasi-static -

Thermodynamics the Goal of Thermodynamics Is to Understand How Heat Can Be Converted to Work
Thermodynamics The goal of thermodynamics is to understand how heat can be converted to work Main lesson: Not all the heat energy can be converted to mechanical energy This is because heat energy comes with disorder (entropy), and overall disorder cannot decrease Temperature v T Random directions of velocity Higher temperature means higher velocities v Add up energy of all molecules: Internal Energy of gas U Mechanical energy: all atoms move in the same direction 1 Mv2 2 Statistical mechanics For one atom 1 1 1 1 1 1 3 E = mv2 + mv2 + mv2 = kT + kT + kT = kT h i h2 xi h2 yi h2 z i 2 2 2 2 Ideal gas: No Potential energy from attraction between atoms 3 U = NkT h i 2 v T Pressure v Pressure is caused because atoms T bounce off the wall Lx p − x px ∆px =2px 2L ∆t = x vx ∆p 2mv2 mv2 F = x = x = x ∆t 2Lx Lx ∆p 2mv2 mv2 F = x = x = x ∆t 2Lx Lx 1 mv2 =2 kT = kT h xi ⇥ 2 kT F = Lx Pressure v F kT 1 kT A = LyLz P = = = A Lx LyLz V NkT Many particles P = V Lx PV = NkT Volume V = LxLyLz Work Lx ∆V = A ∆Lx v A = LyLz Work done BY the gas ∆W = F ∆Lx We can write this as ∆W =(PA) ∆Lx = P ∆V This is useful because the body could have a generic shape Internal energy of gas decreases U U ∆W ! − Gas expands, work is done BY the gas P dW = P dV Work done is Area under curve V P Gas is pushed in, work is done ON the gas dW = P dV − Work done is negative of Area under curve V By convention, we use POSITIVE sign for work done BY the gas Getting work from Heat Gas expands, work is done BY the gas P dW = P dV V Volume in increases Internal energy decreases .. -

Thermodynamics of Power Generation
THERMAL MACHINES AND HEAT ENGINES Thermal machines ......................................................................................................................................... 1 The heat engine ......................................................................................................................................... 2 What it is ............................................................................................................................................... 2 What it is for ......................................................................................................................................... 2 Thermal aspects of heat engines ........................................................................................................... 3 Carnot cycle .............................................................................................................................................. 3 Gas power cycles ...................................................................................................................................... 4 Otto cycle .............................................................................................................................................. 5 Diesel cycle ........................................................................................................................................... 8 Brayton cycle ..................................................................................................................................... -

Thermodynamics I - Enthalpy
CHEM 2880 - Kinetics Thermodynamics I - Enthalpy Tinoco Chapter 2 Secondary Reference: J.B. Fenn, Engines, Energy and Entropy, Global View Publishing, Pittsburgh, 2003. 1 CHEM 2880 - Kinetics Thermodynamics • An essential foundation for understanding physical and biological sciences. • Relationships and interconversions between various forms of energy (mechanical, electrical, chemical, heat, etc) • An understanding of the maximum efficiency with which we can transform one form of energy into another • Preferred direction by which a system evolves, i.e. will a conversion (reaction) go or not • Understanding of equilibrium • It is not based on the ideas of molecules or atoms. A linkage between these and thermo can be achieved using statistical methods. • It does not tell us about the rate of a process (how fast). Domain of kinetics. 2 CHEM 2880 - Kinetics Surroundings, boundaries, system When considering energy relationships it is important to define your point of reference. 3 CHEM 2880 - Kinetics Types of Systems Open: both mass and Closed: energy can be energy may leave and enter exchanged no matter can enter or leave Isolated: neither mass nor energy can enter or leave. 4 CHEM 2880 - Kinetics Energy Transfer Energy can be transferred between the system and the surroundings as heat (q) or work (w). This leads to a change in the internal energy (E or U) of the system. Heat • the energy transfer that occurs when two bodies at different temperatures come in contact with each other - the hotter body tends to cool while the cooler one warms -

ESCI 241 – Meteorology Lesson 8 - Thermodynamic Diagrams Dr
ESCI 241 – Meteorology Lesson 8 - Thermodynamic Diagrams Dr. DeCaria References: The Use of the Skew T, Log P Diagram in Analysis And Forecasting, AWS/TR-79/006, U.S. Air Force, Revised 1979 An Introduction to Theoretical Meteorology, Hess GENERAL Thermodynamic diagrams are used to display lines representing the major processes that air can undergo (adiabatic, isobaric, isothermal, pseudo- adiabatic). The simplest thermodynamic diagram would be to use pressure as the y-axis and temperature as the x-axis. The ideal thermodynamic diagram has three important properties The area enclosed by a cyclic process on the diagram is proportional to the work done in that process As many of the process lines as possible be straight (or nearly straight) A large angle (90 ideally) between adiabats and isotherms There are several different types of thermodynamic diagrams, all meeting the above criteria to a greater or lesser extent. They are the Stuve diagram, the emagram, the tephigram, and the skew-T/log p diagram The most commonly used diagram in the U.S. is the Skew-T/log p diagram. The Skew-T diagram is the diagram of choice among the National Weather Service and the military. The Stuve diagram is also sometimes used, though area on a Stuve diagram is not proportional to work. SKEW-T/LOG P DIAGRAM Uses natural log of pressure as the vertical coordinate Since pressure decreases exponentially with height, this means that the vertical coordinate roughly represents altitude. Isotherms, instead of being vertical, are slanted upward to the right. Adiabats are lines that are semi-straight, and slope upward to the left. -

Lecture 8: Maximum and Minimum Work, Thermodynamic Inequalities
Lecture 8: Maximum and Minimum Work, Thermodynamic Inequalities Chapter II. Thermodynamic Quantities A.G. Petukhov, PHYS 743 October 4, 2017 Chapter II. Thermodynamic Quantities Lecture 8: Maximum and Minimum Work, ThermodynamicA.G. Petukhov,October Inequalities PHYS 4, 743 2017 1 / 12 Maximum Work If a thermally isolated system is in non-equilibrium state it may do work on some external bodies while equilibrium is being established. The total work done depends on the way leading to the equilibrium. Therefore the final state will also be different. In any event, since system is thermally isolated the work done by the system: jAj = E0 − E(S); where E0 is the initial energy and E(S) is final (equilibrium) one. Le us consider the case when Vinit = Vfinal but can change during the process. @ jAj @E = − = −Tfinal < 0 @S @S V The entropy cannot decrease. Then it means that the greater is the change of the entropy the smaller is work done by the system The maximum work done by the system corresponds to the reversible process when ∆S = Sfinal − Sinitial = 0 Chapter II. Thermodynamic Quantities Lecture 8: Maximum and Minimum Work, ThermodynamicA.G. Petukhov,October Inequalities PHYS 4, 743 2017 2 / 12 Clausius Theorem dS 0 R > dS < 0 R S S TA > TA T > T B B A δQA > 0 B Q 0 δ B < The system following a closed path A: System receives heat from a hot reservoir. Temperature of the thermostat is slightly larger then the system temperature B: System dumps heat to a cold reservoir. Temperature of the system is slightly larger then that of the thermostat Chapter II. -

Thermodynamics for Cryogenics
Thermodynamics for Cryogenics Ram C. Dhuley USPAS – Cryogenic Engineering June 21 – July 2, 2021 Goals of this lecture • Revise important definitions in thermodynamics – system and surrounding, state properties, derived properties, processes • Revise laws of thermodynamics and learn how to apply them to cryogenic systems • Learn about ‘ideal’ thermodynamic processes and their application to cryogenic systems • Prepare background for analyzing real-world helium liquefaction and refrigeration cycles. 2 Ram C. Dhuley | USPAS Cryogenic Engineering (Jun 21 - Jul 2, 2021) Introduction • Thermodynamics deals with the relations between heat and other forms of energy such as mechanical, electrical, or chemical energy. • Thermodynamics has its basis in attempts to understand combustion and steam power but is still “state of the art” in terms of practical engineering issues for cryogenics, especially in process efficiency. James Dewar (invented vacuum flask in 1892) https://physicstoday.scitation.org/doi/10.1063/1.881490 3 Ram C. Dhuley | USPAS Cryogenic Engineering (Jun 21 - Jul 2, 2021) Definitions • Thermodynamic system is the specified region in which heat, work, and mass transfer are being studied. • Surrounding is everything else other than the system. • Thermodynamic boundary is a surface separating the system from the surrounding. Boundary System Surrounding 4 Ram C. Dhuley | USPAS Cryogenic Engineering (Jun 21 - Jul 2, 2021) Definitions (continued) Types of thermodynamic systems • Isolated system has no exchange of mass or energy with its surrounding • Closed system exchanges heat but not mass • Open system exchanges both heat and mass with its surrounding Thermodynamic State • Thermodynamic state is the condition of the system at a given time, defined by ‘state’ properties • More details on next slides • Two state properties define a state of a “homogenous” system • Usually, three state properties are needed to define a state of a non-homogenous system (example: two phase, mixture) 5 Ram C.