A Close Look at Wiggly Chromosomes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
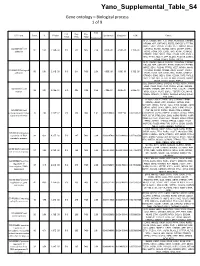
Supplemental Tables4.Pdf
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -

<I>BOC</I> Is a Modifier Gene in Holoprosencephaly
Received: 12 May 2017 Revised: 20 June 2017 Accepted: 28 June 2017 DOI: 10.1002/humu.23286 BRIEF REPORT BOC is a modifier gene in holoprosencephaly Mingi Hong1∗ Kshitij Srivastava2∗ Sungjin Kim1 Benjamin L. Allen3 Daniel J. Leahy4 Ping Hu2 Erich Roessler2 Robert S. Krauss1† Maximilian Muenke2† 1Department of Cell, Developmental, and Regenerative Biology, Icahn School of Medicine at Mount Sinai, New York, New York 2Medical Genetics Branch, National Human Genome Research Institute, National Institutes of Health, Bethesda, Maryland 3Department of Cell and Developmental Biology, University of Michigan, Ann Arbor, Michigan 4Department of Molecular Biosciences, University of Texas at Austin, Austin, Texas Correspondence Robert S. Krauss (for editorial communication), Abstract Department of Cell, Developmental and Regen- Holoprosencephaly (HPE), a common developmental defect of the forebrain and midface, has a erative Biology, Icahn School of Medicine at complex etiology. Heterozygous, loss-of-function mutations in the sonic hedgehog (SHH) pathway Mount Sinai, New York, New York 10029. Email: [email protected] are associated with HPE. However, mutation carriers display highly variable clinical presentation, Maximilian Muenke, Medical Genetics Branch, leading to an “autosomal dominant with modifier” model, in which the penetrance and expressiv- National Human Genome Research Institute, ity of a predisposing mutation is graded by genetic or environmental modifiers. Such modifiers National Institutes of Health, Bethesda, Mary- land 20892. have not been identified. Boc encodes a SHH coreceptor and is a silent HPE modifier gene in mice. Email: [email protected] Here, we report the identification of missense BOC variants in HPE patients. Consistent with these ∗Mingi Hong and Kshitij Srivastava contributed alleles functioning as HPE modifiers, individual variant BOC proteins had either loss- or gain-of- equally to this work. -

Hedgehog Signaling, Epithelial-To-Mesenchymal Transition and Mirna (Review)
271-275 1/8/08 15:18 Page 271 INTERNATIONAL JOURNAL OF MOLECULAR MEDICINE 22: 271-275, 2008 271 Hedgehog signaling, epithelial-to-mesenchymal transition and miRNA (Review) YURIKO KATOH1 and MASARU KATOH2 1M&M Medical Bioinformatics, Hongo 113-0033; 2Genetics and Cell Biology Section, National Cancer Center, Tokyo 104-0045, Japan Received May 19, 2008; Accepted June 26, 2008 DOI: 10.3892/ijmm_00000019 Abstract. SHH, IHH, and DHH are lipid-modified secreted strictly controlled before clinical application of synthetic proteins binding to Patched receptors, and CDON, BOC or miRNA. Peptide mimetic and RNA aptamer could also be GAS1 co-receptors. In the absence of Hedgehog signaling, utilized as Hedgehog signaling inhibitors or EMT suppressors. GLI1 is transcriptionally repressed, GLI2 is phosphorylated by GSK3 and CK1 for the FBXW11 (ßTRCP2)-mediated degradation, and GLI3 is processed to a cleaved repressor. In Contents the presence of Hedgehog signaling, Smoothened is relieved from Patched-mediated suppression due to the Hedgehog- 1. Introduction dependent internalization of Patched, which leads to 2. Hedgehog signaling pathway MAP3K10 (MST) activation and SUFU inactivation for the 3. Epithelial-to-mesenchymal transition (EMT) stabilization and nuclear accumulation of GLI family 4. Hedgehog and EMT members. GLI activators then upregulate CCND1, CCND2 5. MicroRNA (miRNA) for cell cycle acceleration, FOXA2, FOXC2, FOXE1, 6. Perspectives FOXF1, FOXL1, FOXP3, POU3F1, RUNX2, SOX13, TBX2 for cell fate determination, JAG2, INHBC, and INHBE for 1. Introduction stem cell signaling regulation. Hedgehog signals also up- regulate SFRP1 in mesenchymal cells for WNT signaling Hedgehog family members are key regulators of embryo- regulation. Epithelial-to-mesenchymal transition (EMT) during genesis, adult tissue homeostasis, and carcinogenesis (1-5). -

Mutations in CDON, Encoding a Hedgehog Receptor, Result in Holoprosencephaly and Defective Interactions with Other Hedgehog Receptors
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector ARTICLE Mutations in CDON, Encoding a Hedgehog Receptor, Result in Holoprosencephaly and Defective Interactions with Other Hedgehog Receptors Gyu-Un Bae,1,2,4,6 Sabina Domene´,3,4,7 Erich Roessler,3 Karen Schachter,1,8 Jong-Sun Kang,2,5,* Maximilian Muenke,3,5,* and Robert S. Krauss1,5 Holoprosencephaly (HPE), a common human congenital anomaly defined by a failure to delineate the midline of the forebrain and/or midface, is associated with diminished Sonic hedgehog (SHH)-pathway activity in development of these structures. SHH signaling is regulated by a network of ligand-binding factors, including the primary receptor PTCH1 and the putative coreceptors, CDON (also called CDO), BOC, and GAS1. Although binding of SHH to these receptors promotes pathway activity, it is not known whether interactions between these receptors are important. We report here identification of missense CDON mutations in human HPE. These mutations diminish CDON’s ability to support SHH-dependent gene expression in cell-based signaling assays. The mutations occur outside the SHH-binding domain of CDON, and the encoded variant CDON proteins do not display defects in binding to SHH. In contrast, wild-type CDON associates with PTCH1 and GAS1, but the variants do so inefficiently, in a manner that parallels their activity in cell-based assays. Our findings argue that CDON must associate with both ligand and other hedgehog-receptor components, particularly PTCH1, for signaling to occur and that disruption of the latter interactions is a mechanism of HPE. -

Role of Cytonemes in Wnt Transport Eliana Stanganello and Steffen Scholpp*
© 2016. Published by The Company of Biologists Ltd | Journal of Cell Science (2016) 129, 665-672 doi:10.1242/jcs.182469 COMMENTARY Role of cytonemes in Wnt transport Eliana Stanganello and Steffen Scholpp* ABSTRACT dependent or canonical Wnt signaling (reviewed in Logan and Wnt signaling regulates a broad variety of processes during Nusse, 2004). Paracrine signaling activity is fundamental to the embryonic development and disease. A hallmark of the Wnt morphogenetic function of Wnt proteins in tissue patterning. signaling pathway is the formation of concentration gradients by However, the extracellular transport mechanism of this morphogen Wnt proteins across responsive tissues, which determines cell fate from the signal-releasing cell to the recipient cell is still debated. in invertebrates and vertebrates. To fulfill its paracrine function, Recent data suggest that Wnt proteins are distributed on long trafficking of the Wnt morphogen from an origin cell to a recipient cell signaling filopodia known as cytonemes, which allow contact- must be tightly regulated. A variety of models have been proposed to dependent, juxtacrine signaling over a considerable distance. We explain the extracellular transport of these lipid-modified signaling have recently shown that, in zebrafish, Wnt8a is transported on proteins in the aqueous extracellular space; however, there is still actin-containing cytonemes to cells, where it activates the signaling considerable debate with regard to which mechanisms allow the required for the specification of neural plate cells (Stanganello et al., precise distribution of ligand in order to generate a morphogenetic 2015). Concomitantly, the Wnt receptor Frz was identified on gradient within growing tissue. Recent evidence suggests that Wnt cytonemes to enable the retrograde transport of Wnt proteins on proteins are distributed along signaling filopodia during vertebrate these protrusions during flight muscle formation in Drosophila and invertebrate embryogenesis. -

"The Genecards Suite: from Gene Data Mining to Disease Genome Sequence Analyses". In: Current Protocols in Bioinformat
The GeneCards Suite: From Gene Data UNIT 1.30 Mining to Disease Genome Sequence Analyses Gil Stelzer,1,5 Naomi Rosen,1,5 Inbar Plaschkes,1,2 Shahar Zimmerman,1 Michal Twik,1 Simon Fishilevich,1 Tsippi Iny Stein,1 Ron Nudel,1 Iris Lieder,2 Yaron Mazor,2 Sergey Kaplan,2 Dvir Dahary,2,4 David Warshawsky,3 Yaron Guan-Golan,3 Asher Kohn,3 Noa Rappaport,1 Marilyn Safran,1 and Doron Lancet1,6 1Department of Molecular Genetics, Weizmann Institute of Science, Rehovot, Israel 2LifeMap Sciences Ltd., Tel Aviv, Israel 3LifeMap Sciences Inc., Marshfield, Massachusetts 4Toldot Genetics Ltd., Hod Hasharon, Israel 5These authors contributed equally to the paper 6Corresponding author GeneCards, the human gene compendium, enables researchers to effectively navigate and inter-relate the wide universe of human genes, diseases, variants, proteins, cells, and biological pathways. Our recently launched Version 4 has a revamped infrastructure facilitating faster data updates, better-targeted data queries, and friendlier user experience. It also provides a stronger foundation for the GeneCards suite of companion databases and analysis tools. Improved data unification includes gene-disease links via MalaCards and merged biological pathways via PathCards, as well as drug information and proteome expression. VarElect, another suite member, is a phenotype prioritizer for next-generation sequencing, leveraging the GeneCards and MalaCards knowledgebase. It au- tomatically infers direct and indirect scored associations between hundreds or even thousands of variant-containing genes and disease phenotype terms. Var- Elect’s capabilities, either independently or within TGex, our comprehensive variant analysis pipeline, help prepare for the challenge of clinical projects that involve thousands of exome/genome NGS analyses. -

PRMT7 Ablation in Cardiomyocytes Causes Cardiac Hypertrophy and Fibrosis Through Β-Catenin Dysregulation
PRMT7 Ablation in Cardiomyocytes Causes Cardiac Hypertrophy and Fibrosis Through β-Catenin Dysregulation Byeong-Yun Ahn Sungkyunkwan University School of Medicine Myong-Ho Jeong Sungkyunkwan University School of Medicine Jung-Hoon Pyun Sungkyunkwan University School of Medicine Hyeon-Ju Jeong Sungkyunkwan University School of Medicine Tuan Anh Vuong Sungkyunkwan University School of Medicine Ju-Hyeon Bae Sungkyunkwan University School of Medicine Subin An Sungkyunkwan University School of Medicine Su Woo Kim Sungkyunkwan University School of Medicine Yong Kee Kim Sookmyung Women's University Dongryeol Ryu Sungkyunkwan University School of Medicine Hyun-Ji Kim Sungkyunkwan University School of Medicine Hana Cho Sungkyunkwan University School of Medicine Gyu-Un Bae ( [email protected] ) Sookmyung Women's University College of Pharmacy https://orcid.org/0000-0003-4262-1895 Jong-Sun Kang Sungkyunkwan University School of Medicine Research Article Page 1/24 Keywords: PRMT7, cardiomyopathy, Wnt, β-Catenin Posted Date: August 19th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-798227/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 2/24 Abstract Angiotensin II (AngII) has potent cardiac hypertrophic effects mediated through activation of hypertrophic signaling like Wnt/b-Catenin signaling. In the current study, we examined the role of protein arginine methyltransferase 7 (PRMT7) in cardiac function. PRMT7 was greatly decreased in hypertrophic hearts chronically infused with AngII and cardiomyocytes treated with AngII. PRMT7 depletion in rat cardiomyocytes resulted in hypertrophic responses. Consistently, mice lacking PRMT7 exhibited displayed the cardiac hypertrophy and brosis. PRMT7 overexpression abrogated the cellular hypertrophy elicited by AngII, while PRMT7 depletion exacerbated the hypertrophic response caused by AngII. -

Keep Your Friends Close: Cell–Cell Contact and Skeletal Myogenesis
Downloaded from http://cshperspectives.cshlp.org/ on October 2, 2021 - Published by Cold Spring Harbor Laboratory Press Keep Your Friends Close: Cell–Cell Contact and Skeletal Myogenesis Robert S. Krauss, Giselle A. Joseph, and Aviva J. Goel Department of Cell, Developmental, and Regenerative Biology, Graduate School of Biomedical Sciences, Icahn School of Medicine at Mount Sinai, New York, New York 10029 Correspondence: [email protected] Development of skeletal muscle is a multistage process that includes lineage commitment of multipotent progenitor cells, differentiation and fusion of myoblasts into multinucleated myofibers, and maturation of myofibers into distinct types. Lineage-specific transcriptional regulation lies at the core of this process, but myogenesis is also regulated by extracellular cues. Some of these cues are initiated by direct cell–cell contact between muscle precursor cells themselves or between muscle precursors and cells of other lineages. Examples of the latter include interaction of migrating neural crest cells with multipotent muscle progenitor cells, muscle interstitial cells with myoblasts, and neurons with myofibers. Among the sig- naling factors involved are Notch ligands and receptors, cadherins, Ig superfamily members, and Ephrins and Eph receptors. In this article we describe recent progress in this area and highlight open questions raised by the findings. keletal muscle is the most abundant tissue in based on expression of distinct myosin heavy Sthe vertebrate body and necessary for breath- chain (MyHC) isoforms and metabolic capabil- ing, metabolic homeostasis, and locomotion. ities (Schiaffino and Reggiani 2011; Talbot and Development of skeletal muscle occurs in a se- Maves 2016). quential, multistage process that includes spec- Myogenic specification and differentiation ification of multipotent progenitors to lineage- are coordinated by the myogenic basic helix– committed myoblasts, differentiation of myo- loop–helix (bHLH) transcription factors Myf5, blasts into multinucleated myofibers, and mat- MyoD, myogenin, and MRF4. -

Sex-Specific Transcriptome Differences in Human Adipose
G C A T T A C G G C A T genes Article Sex-Specific Transcriptome Differences in Human Adipose Mesenchymal Stem Cells 1, 2, 3 1,3 Eva Bianconi y, Raffaella Casadei y , Flavia Frabetti , Carlo Ventura , Federica Facchin 1,3,* and Silvia Canaider 1,3 1 National Laboratory of Molecular Biology and Stem Cell Bioengineering of the National Institute of Biostructures and Biosystems (NIBB)—Eldor Lab, at the Innovation Accelerator, CNR, Via Piero Gobetti 101, 40129 Bologna, Italy; [email protected] (E.B.); [email protected] (C.V.); [email protected] (S.C.) 2 Department for Life Quality Studies (QuVi), University of Bologna, Corso D’Augusto 237, 47921 Rimini, Italy; [email protected] 3 Department of Experimental, Diagnostic and Specialty Medicine (DIMES), University of Bologna, Via Massarenti 9, 40138 Bologna, Italy; fl[email protected] * Correspondence: [email protected]; Tel.: +39-051-2094114 These authors contributed equally to this work. y Received: 1 July 2020; Accepted: 6 August 2020; Published: 8 August 2020 Abstract: In humans, sexual dimorphism can manifest in many ways and it is widely studied in several knowledge fields. It is increasing the evidence that also cells differ according to sex, a correlation still little studied and poorly considered when cells are used in scientific research. Specifically, our interest is on the sex-related dimorphism on the human mesenchymal stem cells (hMSCs) transcriptome. A systematic meta-analysis of hMSC microarrays was performed by using the Transcriptome Mapper (TRAM) software. This bioinformatic tool was used to integrate and normalize datasets from multiple sources and allowed us to highlight chromosomal segments and genes differently expressed in hMSCs derived from adipose tissue (hADSCs) of male and female donors. -

Duke University Dissertation Template
Gene-Environment Interactions in Cardiovascular Disease by Cavin Keith Ward-Caviness Graduate Program in Computational Biology and Bioinformatics Duke University Date:_______________________ Approved: ___________________________ Elizabeth R. Hauser, Supervisor ___________________________ William E. Kraus ___________________________ Sayan Mukherjee ___________________________ H. Frederik Nijhout Dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate Program in Computational Biology and Bioinformatics in the Graduate School of Duke University 2014 i v ABSTRACT Gene-Environment Interactions in Cardiovascular Disease by Cavin Keith Ward-Caviness Graduate Program in Computational Biology and Bioinformatics Duke University Date:_______________________ Approved: ___________________________ Elizabeth R. Hauser, Supervisor ___________________________ William E. Kraus ___________________________ Sayan Mukherjee ___________________________ H. Frederik Nijhout An abstract of a dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate Program in Computational Biology and Bioinformatics in the Graduate School of Duke University 2014 Copyright by Cavin Keith Ward-Caviness 2014 Abstract In this manuscript I seek to demonstrate the importance of gene-environment interactions in cardiovascular disease. This manuscript contains five studies each of which contributes to our understanding of the joint impact of genetic variation -

Deregulation of Cell Adhesion Molecules Is Associated with Progression and Poor Outcomes in Endometrial Cancer: Analysis of the Cancer Genome Atlas Data
1906 ONCOLOGY LETTERS 19: 1906-1914, 2020 Deregulation of cell adhesion molecules is associated with progression and poor outcomes in endometrial cancer: Analysis of The Cancer Genome Atlas data XIANGJUN HE1, SHU LEI1,2, QI ZHANG1, LIPING MA1, NA LI1 and JIANLIU WANG2 1Central Laboratory and Institute of Clinical Molecular Biology; 2Department of Obstetrics and Gynecology, Peking University People's Hospital, Beijing 100044, P.R. China Received June 18, 2019; Accepted November 15, 2019 DOI: 10.3892/ol.2020.11295 Abstract. Cell adhesion molecules (CAMs) determine the 11 times of that in those without elevation of these 6 CAMs. behavior of cancer cells during metastasis. Although some Similar results were obtained when relapse-free survival (RFS) CAMs are dysregulated in certain types of cancer and time was used during the analysis. Prognostic reliability of the are associated with cancer progression, to the best of our six-gene model was validated using data of an independent knowledge, a comprehensive study of CAMs has not been cohort from the International Cancer Genome Consortium. In undertaken, particularly in endometrial cancer (EC). In the conclusion, a combination of CAM alterations contributed to present study the expression of 225 CAMs in EC patients progression and aggressiveness of EC. The six-gene signature with various clinicopathological phenotypes were evaluated was effective for predicting worse OS and RFS in patients by statistical analysis using publicly available data from The with EC and could be complementary to the present clinical Cancer Genome Atlas database. The Kaplan-Meier method, prognostic criteria. and univariate and multivariate Cox proportional hazards regression models were used for survival analyses. -

Cdon Acts As a Hedgehog Decoy Receptor During Proximal-Distal Patterning of the Optic Vesicle
ARTICLE Received 10 Dec 2013 | Accepted 26 May 2014 | Published 8 Jul 2014 DOI: 10.1038/ncomms5272 OPEN Cdon acts as a Hedgehog decoy receptor during proximal-distal patterning of the optic vesicle Marcos Julia´n Cardozo1,2, Luisa Sa´nchez-Arrones1,2,A´frica Sandonis1,2, Cristina Sa´nchez-Camacho2,w, Gaia Gestri3, Stephen W. Wilson3, Isabel Guerrero1 & Paola Bovolenta1,2 Patterning of the vertebrate optic vesicle into proximal/optic stalk and distal/neural retina involves midline-derived Hedgehog (Hh) signalling, which promotes stalk specification. In the absence of Hh signalling, the stalks are not specified, causing cyclopia. Recent studies showed that the cell adhesion molecule Cdon forms a heteromeric complex with the Hh receptor Patched 1 (Ptc1). This receptor complex binds Hh and enhances signalling activation, indicating that Cdon positively regulates the pathway. Here we show that in the developing zebrafish and chick optic vesicle, in which cdon and ptc1 are expressed with a complementary pattern, Cdon acts as a negative Hh signalling regulator. Cdon predominantly localizes to the basolateral side of neuroepithelial cells, promotes the enlargement of the neuroepithelial basal end-foot and traps Hh protein, thereby limiting its dispersion. This Ptc-independent function protects the retinal primordium from Hh activity, defines the stalk/retina boundary and thus the correct proximo-distal patterning of the eye. 1 Centro de Biologı´a Molecular Severo Ochoa, Consejo Superior de Investigaciones Cientı´ficas—Universidad Auto´noma de Madrid, 28049 Madrid Spain. 2 Ciber de Enfermedades Raras (CIBERER), ISCIII, c/ Nicolas Cabrera 1, 28049 Madrid Spain. 3 Department of Cell and Developmental Biology, University College London, Gower Street, London WC1E 6BT, UK.