Disease-Relevant Transcriptional Signatures Identified in Individual
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Increased Expression of the Tail-Anchored Membrane Protein SLMAP in Adipose Tissue from Type 2 Tally Ho Diabetic Mice
Hindawi Publishing Corporation Experimental Diabetes Research Volume 2011, Article ID 421982, 10 pages doi:10.1155/2011/421982 Research Article Increased Expression of the Tail-Anchored Membrane Protein SLMAP in Adipose Tissue from Type 2 Tally Ho Diabetic Mice Xiaoliang Chen1 and Hong Ding2 1 Second People Hospital of Hangzhou, No. 1 Wenzhou Road, Hangzhou 310015, China 2 Department of Pharmacology, Weill Cornell Medical College in Qatar, P.O. Box 24144, Doha, Qatar Correspondence should be addressed to Hong Ding, [email protected] Received 6 April 2011; Accepted 5 May 2011 Academic Editor: N. Cameron Copyright © 2011 X. Chen and H. Ding. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. The tail-anchored membrane protein, sarcolemmal membrane associated protein (SLMAP) is encoded to a single gene that maps to the chromosome 3p14 region and has also been reported in certain diabetic populations. Our previous studies with db/db mice shown that a deregulation of SLMAP expression plays an important role in type 2 diabetes. Male Tally Ho mice were bred to present with either normoglycemia (NG) or hyperglycemia (HG). Abdominal adipose tissue from male Tally Ho mice of the HG group was found to have a significantly lower expression of the membrane associated glucose transporter-4 (GLUT-4) and higher expression of SLMAP compared to tissue from NG mice. There were 3 isoforms expressed in the abdominal adipose tissue, but only 45 kDa isoform of SLMAP was associated with the GLUT-4 revealed by immunoprecipitation data. -

Atherosclerosis-Susceptible and Atherosclerosis-Resistant Pigeon Aortic Cells Express Different Genes in Vivo
University of New Hampshire University of New Hampshire Scholars' Repository New Hampshire Agricultural Experiment Station Publications New Hampshire Agricultural Experiment Station 7-1-2013 Atherosclerosis-susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo Janet L. Anderson University of New Hampshire, [email protected] C. M. Ashwell University of New Hampshire - Main Campus S. C. Smith University of New Hampshire - Main Campus R. Shine University of New Hampshire - Main Campus E. C. Smith University of New Hampshire - Main Campus See next page for additional authors Follow this and additional works at: https://scholars.unh.edu/nhaes Part of the Poultry or Avian Science Commons Recommended Citation J. L. Anderson, C. M. Ashwell, S. C. Smith, R. Shine, E. C. Smith and R. L. Taylor, Jr. Atherosclerosis- susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo Poultry Science (2013) 92 (10): 2668-2680 doi:10.3382/ps.2013-03306 This Article is brought to you for free and open access by the New Hampshire Agricultural Experiment Station at University of New Hampshire Scholars' Repository. It has been accepted for inclusion in New Hampshire Agricultural Experiment Station Publications by an authorized administrator of University of New Hampshire Scholars' Repository. For more information, please contact [email protected]. Authors Janet L. Anderson, C. M. Ashwell, S. C. Smith, R. Shine, E. C. Smith, and Robert L. Taylor Jr. This article is available at University of New Hampshire Scholars' Repository: https://scholars.unh.edu/nhaes/207 Atherosclerosis-susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo J. -

Protein Kinase A-Mediated Septin7 Phosphorylation Disrupts Septin Filaments and Ciliogenesis
cells Article Protein Kinase A-Mediated Septin7 Phosphorylation Disrupts Septin Filaments and Ciliogenesis Han-Yu Wang 1,2, Chun-Hsiang Lin 1, Yi-Ru Shen 1, Ting-Yu Chen 2,3, Chia-Yih Wang 2,3,* and Pao-Lin Kuo 1,2,4,* 1 Department of Obstetrics and Gynecology, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan; [email protected] (H.-Y.W.); [email protected] (C.-H.L.); [email protected] (Y.-R.S.) 2 Institute of Basic Medical Sciences, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan; [email protected] 3 Department of Cell Biology and Anatomy, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan 4 Department of Obstetrics and Gynecology, National Cheng-Kung University Hospital, Tainan 704, Taiwan * Correspondence: [email protected] (C.-Y.W.); [email protected] (P.-L.K.); Tel.: +886-6-2353535 (ext. 5338); (C.-Y.W.)+886-6-2353535 (ext. 5262) (P.-L.K.) Abstract: Septins are GTP-binding proteins that form heteromeric filaments for proper cell growth and migration. Among the septins, septin7 (SEPT7) is an important component of all septin filaments. Here we show that protein kinase A (PKA) phosphorylates SEPT7 at Thr197, thus disrupting septin filament dynamics and ciliogenesis. The Thr197 residue of SEPT7, a PKA phosphorylating site, was conserved among different species. Treatment with cAMP or overexpression of PKA catalytic subunit (PKACA2) induced SEPT7 phosphorylation, followed by disruption of septin filament formation. Constitutive phosphorylation of SEPT7 at Thr197 reduced SEPT7-SEPT7 interaction, but did not affect SEPT7-SEPT6-SEPT2 or SEPT4 interaction. -

The Requirement of SEPT2 and SEPT7 for Migration and Invasion in Human Breast Cancer Via MEK/ERK Activation
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 7, No. 38 Research Paper The requirement of SEPT2 and SEPT7 for migration and invasion in human breast cancer via MEK/ERK activation Nianzhu Zhang1,*, Lu Liu2,*, Ning Fan2, Qian Zhang1, Weijie Wang1, Mingnan Zheng3, Lingfei Ma4, Yan Li2, Lei Shi1,5 1Institute of Cancer Stem Cell, Cancer Center, Dalian Medical University, Dalian, 116044, Liaoning, P.R.China 2College of Basic Medical Sciences, Dalian Medical University, Dalian, 116044 Liaoning, P.R.China 3Department of Gynecology and Obstetrics, Dalian Municipal Central Hospital Affiliated to Dalian Medical University, Dalian, 116033, Liaoning, P.R.China 4The First Affiliated Hospital of Dalian Medical University, Dalian, 116011, Liaoning, P.R.China 5State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, P.R.China *These authors contributed equally to this work Correspondence to: Lei Shi, email: [email protected] Yan Li, email: [email protected] Lingfei Ma, email: [email protected] Keywords: septin, forchlorfenuron, breast cancer, invasion, MAPK Received: April 24, 2016 Accepted: July 28, 2016 Published: August 19, 2016 ABSTRACT Septins are a novel class of GTP-binding cytoskeletal proteins evolutionarily conserved from yeast to mammals and have now been found to play a contributing role in a broad range of tumor types. However, their functional importance in breast cancer remains largely unclear. Here, we demonstrated that pharmaceutical inhibition of global septin dynamics would greatly suppress proliferation, migration and invasiveness in breast cancer cell lines. We then examined the expression and subcellular distribution of the selected septins SEPT2 and SEPT7 in breast cancer cells, revealing a rather variable localization of the two proteins with cell cycle progression. -
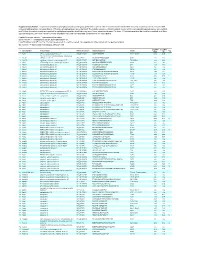
Supplementary Table 1
Supplementary Table 1. Large-scale quantitative phosphoproteomic profiling was performed on paired vehicle- and hormone-treated mTAL-enriched suspensions (n=3). A total of 654 unique phosphopeptides corresponding to 374 unique phosphoproteins were identified. The peptide sequence, phosphorylation site(s), and the corresponding protein name, gene symbol, and RefSeq Accession number are reported for each phosphopeptide identified in any one of three experimental pairs. For those 414 phosphopeptides that could be quantified in all three experimental pairs, the mean Hormone:Vehicle abundance ratio and corresponding standard error are also reported. Peptide Sequence column: * = phosphorylated residue Site(s) column: ^ = ambiguously assigned phosphorylation site Log2(H/V) Mean and SE columns: H = hormone-treated, V = vehicle-treated, n/a = peptide not observable in all 3 experimental pairs Sig. column: * = significantly changed Log 2(H/V), p<0.05 Log (H/V) Log (H/V) # Gene Symbol Protein Name Refseq Accession Peptide Sequence Site(s) 2 2 Sig. Mean SE 1 Aak1 AP2-associated protein kinase 1 NP_001166921 VGSLT*PPSS*PK T622^, S626^ 0.24 0.95 PREDICTED: ATP-binding cassette, sub-family A 2 Abca12 (ABC1), member 12 XP_237242 GLVQVLS*FFSQVQQQR S251^ 1.24 2.13 3 Abcc10 multidrug resistance-associated protein 7 NP_001101671 LMT*ELLS*GIRVLK T464, S468 -2.68 2.48 4 Abcf1 ATP-binding cassette sub-family F member 1 NP_001103353 QLSVPAS*DEEDEVPVPVPR S109 n/a n/a 5 Ablim1 actin-binding LIM protein 1 NP_001037859 PGSSIPGS*PGHTIYAK S51 -3.55 1.81 6 Ablim1 actin-binding -

The Role of Sept2 on Neuronal Development
THE ROLE OF SEPT2 ON NEURONAL DEVELOPMENT by HYUN JONG KIM A Dissertation submitted to the Graduate School-New Brunswick Rutgers, The State University of New Jersey and The Graduate School of Biomedical Sciences University of Medicine and Dentistry of New Jersey in partial fulfillment of the requirements for the degree of Doctor of Philosophy Graduate Program in Cell and Developmental Biology written under the direction of Shu Chan Hsu, Ph. D and approved by ________________________ ________________________ ________________________ ________________________ ________________________ New Brunswick, New Jersey May, 2009 ABSTRACT THE ROLE OF SEPT2 ON NEURONAL DEVELOPMENT By HYUN JONG KIM Dissertation Director: Shu Chan Hsu, Ph D Septins constitute a family of evolutionarily conserved proteins that were first discovered for their involvement in cell division. Recently, they are also found to be increasingly important in various neurodegenerative states, such as Parkinson’s and Alzheimer’s diseases. Molecular mechanisms underlying the function of septin proteins in the nervous system, however, remain largely unknown. Work from this thesis revealed that a member of the septin family, SEPT2, is important for exocytosis underlying neurite outgrowth in PC12 cells. SEPT2 has a similar tissue distribution and developmental expression profile as that of sec15, a subunit of the exocyst complex which has been found to be required for exocytosis. In undifferentiated PC12 cells, SEPT2 exhibited perinuclear enrichment, as observed for exocyst subunits. Upon neuronal differentiation, SEPT2, like exocyst subunits, redistributes from its perinuclear enrichment to the neurites and the growth cones. Knockdown of SEPT2 disrupted the perinuclear enrichment of exocyst subunits, decreased the protein levels of exocyst subunits. -

ID AKI Vs Control Fold Change P Value Symbol Entrez Gene Name *In
ID AKI vs control P value Symbol Entrez Gene Name *In case of multiple probesets per gene, one with the highest fold change was selected. Fold Change 208083_s_at 7.88 0.000932 ITGB6 integrin, beta 6 202376_at 6.12 0.000518 SERPINA3 serpin peptidase inhibitor, clade A (alpha-1 antiproteinase, antitrypsin), member 3 1553575_at 5.62 0.0033 MT-ND6 NADH dehydrogenase, subunit 6 (complex I) 212768_s_at 5.50 0.000896 OLFM4 olfactomedin 4 206157_at 5.26 0.00177 PTX3 pentraxin 3, long 212531_at 4.26 0.00405 LCN2 lipocalin 2 215646_s_at 4.13 0.00408 VCAN versican 202018_s_at 4.12 0.0318 LTF lactotransferrin 203021_at 4.05 0.0129 SLPI secretory leukocyte peptidase inhibitor 222486_s_at 4.03 0.000329 ADAMTS1 ADAM metallopeptidase with thrombospondin type 1 motif, 1 1552439_s_at 3.82 0.000714 MEGF11 multiple EGF-like-domains 11 210602_s_at 3.74 0.000408 CDH6 cadherin 6, type 2, K-cadherin (fetal kidney) 229947_at 3.62 0.00843 PI15 peptidase inhibitor 15 204006_s_at 3.39 0.00241 FCGR3A Fc fragment of IgG, low affinity IIIa, receptor (CD16a) 202238_s_at 3.29 0.00492 NNMT nicotinamide N-methyltransferase 202917_s_at 3.20 0.00369 S100A8 S100 calcium binding protein A8 215223_s_at 3.17 0.000516 SOD2 superoxide dismutase 2, mitochondrial 204627_s_at 3.04 0.00619 ITGB3 integrin, beta 3 (platelet glycoprotein IIIa, antigen CD61) 223217_s_at 2.99 0.00397 NFKBIZ nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, zeta 231067_s_at 2.97 0.00681 AKAP12 A kinase (PRKA) anchor protein 12 224917_at 2.94 0.00256 VMP1/ mir-21likely ortholog -

Role of SLMAP Genetic Variants in Susceptibility Of
Upadhyay et al. Journal of Translational Medicine (2015) 13:61 DOI 10.1186/s12967-015-0411-6 RESEARCH Open Access Role of SLMAP genetic variants in susceptibility of diabetes and diabetic retinopathy in Qatari population Rohit Upadhyay1, Amal Robay2, Khalid Fakhro2, Charbel Abi Khadil2, Mahmoud Zirie3, Amin Jayyousi3, Maha El-Shafei4, Szilard Kiss5, Donald J D′Amico5, Jacqueline Salit6, Michelle R Staudt6, Sarah L O′Beirne6, Xiaoliang Chen7, Balwant Tuana8, Ronald G Crystal6 and Hong Ding1* Abstract Background: Overexpression of SLMAP gene has been associated with diabetes and endothelial dysfunction of macro- and micro-blood vessels. In this study our primary objective is to explore the role of SLMAP gene polymorphisms in the susceptibility of type 2 diabetes (T2DM) with or without diabetic retinopathy (DR) in the Qatari population. Methods: A total of 342 Qatari subjects (non-diabetic controls and T2DM patients with or without DR) were genotyped for SLMAP gene polymorphisms (rs17058639 C > T; rs1043045 C > T and rs1057719 A > G) using Taqman SNP genotyping assay. Results: SLMAP rs17058639 C > T polymorphism was associated with the presence of DR among Qataris with T2DM. One-way ANOVA and multiple logistic regression analysis showed SLMAP SNP rs17058639 C > T as an independent risk factor for DR development. SLMAP rs17058639 C > T polymorphism also had a predictive role for the severity of DR. Haplotype Crs17058639Trs1043045Ars1057719 was associated with the increased risk for DR among Qataris with T2DM. Conclusions: The data suggests the potential role of SLMAP SNPs as a risk factor for the susceptibility of DR among T2DM patients in the Qatari population. -

The Human Gene Connectome As a Map of Short Cuts for Morbid Allele Discovery
The human gene connectome as a map of short cuts for morbid allele discovery Yuval Itana,1, Shen-Ying Zhanga,b, Guillaume Vogta,b, Avinash Abhyankara, Melina Hermana, Patrick Nitschkec, Dror Friedd, Lluis Quintana-Murcie, Laurent Abela,b, and Jean-Laurent Casanovaa,b,f aSt. Giles Laboratory of Human Genetics of Infectious Diseases, Rockefeller Branch, The Rockefeller University, New York, NY 10065; bLaboratory of Human Genetics of Infectious Diseases, Necker Branch, Paris Descartes University, Institut National de la Santé et de la Recherche Médicale U980, Necker Medical School, 75015 Paris, France; cPlateforme Bioinformatique, Université Paris Descartes, 75116 Paris, France; dDepartment of Computer Science, Ben-Gurion University of the Negev, Beer-Sheva 84105, Israel; eUnit of Human Evolutionary Genetics, Centre National de la Recherche Scientifique, Unité de Recherche Associée 3012, Institut Pasteur, F-75015 Paris, France; and fPediatric Immunology-Hematology Unit, Necker Hospital for Sick Children, 75015 Paris, France Edited* by Bruce Beutler, University of Texas Southwestern Medical Center, Dallas, TX, and approved February 15, 2013 (received for review October 19, 2012) High-throughput genomic data reveal thousands of gene variants to detect a single mutated gene, with the other polymorphic genes per patient, and it is often difficult to determine which of these being of less interest. This goes some way to explaining why, variants underlies disease in a given individual. However, at the despite the abundance of NGS data, the discovery of disease- population level, there may be some degree of phenotypic homo- causing alleles from such data remains somewhat limited. geneity, with alterations of specific physiological pathways under- We developed the human gene connectome (HGC) to over- come this problem. -

A Homozygous Genome-Edited Sept2-EGFP Fibroblast Cell Line
bioRxiv preprint doi: https://doi.org/10.1101/570622; this version posted March 7, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. A homozygous genome-edited Sept2-EGFP fibroblast cell line Monika Banko2,3, Iwona Mucha-Kruczynska1,2, Christoph Weise1, Florian Heyd1 and Helge Ewers1,2,3* 1Institut für Chemie und Biochemie, Freie Universität Berlin, Thielallee 63, 14195 Berlin, Germany 2Randall Divison of Cell and Molecular Biophysics, King’s College London, London SE1 1UL, United Kingdom 3Institut für Biochemie, ETH Zürich, Switzerland *Corresponding author: Helge Ewers Institut für Biochemie Freie Universität Berlin Thielallee 63 14195 Berlin Germany Tel: +49 30 838 60644 Email: [email protected] Running Head: Genome-edited Sept2-EGFP cell line 1 bioRxiv preprint doi: https://doi.org/10.1101/570622; this version posted March 7, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract Septins are a conserved, essential family of GTPases that interact with actin, microtubules and membranes and form scaffolds and diffusion barriers in cells. Several of the 13 known mammalian septins assemble into nonpolar, multimeric complexes that can further polymerize into filamentous structures. While some GFP-coupled septins have been described, overexpression of GFP-tagged septins often leads to artifacts in localization and function. -

Septins Guide Microtubules
RESEARCH HIGHLIGHTS Akt-ing to control lipid LTsc1KO hepatocytes with constitutively active grew. Knockdown of SEPT2 resulted in a loss metabolism Akt2, and showed that in hepatocytes Akt2 of microtubule directionality and the microtu- represses INSIG2, an inhibitor of SREBP1c bules ended up becoming entangled. Further- The liver responds to insulin by blocking gluco- induction. These data uncover an mTORC1- more, SEPT2 knockdown increased micro- neogenesis and stimulating lipogenesis. Insulin- independent mechanism for Akt-mediated tubule shrinkage, suggesting septins inhibit induced lipid synthesis requires induction of the regulation of liver lipogenesis. AIZ microtubule depolymerization and thus main- transcription factor SREBP1c, which is mediated tain persistent microtubule growth. by the activation of mTORC1 (mammalian tar- Hence, septin control of microtubule growth get of rapamycin complex 1), through the Akt- and directionality seems to allow for correct dependent inhibition of the TSC1–TSC2 (tuber- This way up: Septins guide organization of microtubules in establishing a ous sclerosis protein 1 and 2) complex. Yecies microtubules polarized epithelial cell. GD et al. now identify an mTORC1-independent pathway that induces SREBP1c to promote lipo- Formation of a polarized epithelial cell requires genesis in the liver (Cell Metab. 14, 21–32; 2011). rearrangement of the cell’s microtubules. As the To elucidate the role of mTORC1 in the flat cell rises up into a column, the microtubules SNARE proteins regulate regulation of hepatic lipid metabolism, Yecies are formed into a complex network consisting autophagosome biogenesis et al. genetically ablated Tsc1 to generate a of bundles aligned from the top to the bottom liver-specific, insulin-independent mTORC1 of the cell, and a meshwork of shorter filaments Pre-autophagosomal structures mature into gain-of-function model (LTsc1KO). -

Lineage-Defined Leiomyosarcoma Subtypes Emerge Years Before Diagnosis and Determine Patient Survival
ARTICLE https://doi.org/10.1038/s41467-021-24677-6 OPEN Lineage-defined leiomyosarcoma subtypes emerge years before diagnosis and determine patient survival Nathaniel D. Anderson 1,2, Yael Babichev3,17, Fabio Fuligni4,17, Federico Comitani 1, Mehdi Layeghifard 1, Rosemarie E. Venier 3, Stefan C. Dentro5, Anant Maheshwari1, Sheena Guram3, Claire Wunker3,6, J. Drew Thompson1, Kyoko E. Yuki1, Huayun Hou 1, Matthew Zatzman1,2, Nicholas Light1,6, Marcus Q. Bernardini7,8, Jay S. Wunder 3,9,10, Irene L. Andrulis 2,3,11, Peter Ferguson7,9,10, Albiruni R. Abdul Razak7, Carol J. Swallow3,6,9,12, James J. Dowling1,11, Rima S. Al-Awar13,14, Richard Marcellus13, 1234567890():,; Marjan Rouzbahman2,7, Moritz Gerstung 5, Daniel Durocher 3,11, Ludmil B. Alexandrov 15, ✉ ✉ Brendan C. Dickson 2,3,16, Rebecca A. Gladdy 3,6,9,12 & Adam Shlien 1,2,4 Leiomyosarcomas (LMS) are genetically heterogeneous tumors differentiating along smooth muscle lines. Currently, LMS treatment is not informed by molecular subtyping and is associated with highly variable survival. While disease site continues to dictate clinical management, the contribution of genetic factors to LMS subtype, origins, and timing are unknown. Here we analyze 70 genomes and 130 transcriptomes of LMS, including multiple tumor regions and paired metastases. Molecular profiling highlight the very early origins of LMS. We uncover three specific subtypes of LMS that likely develop from distinct lineages of smooth muscle cells. Of these, dedifferentiated LMS with high immune infiltration and tumors primarily of gynecological origin harbor genomic dystrophin deletions and/or loss of dystrophin expression, acquire the highest burden of genomic mutation, and are associated with worse survival.