The Human Non-Visual Opsin OPN3 Regulates Pigmentation of Epidermal Melanocytes Through Interaction with MC1R
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Peropsin, a Novel Visual Pigment-Like Protein Located in the Apical Microvilli of the Retinal Pigment Epithelium
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 9893–9898, September 1997 Neurobiology Peropsin, a novel visual pigment-like protein located in the apical microvilli of the retinal pigment epithelium HUI SUN*, DEBRA J. GILBERT†,NEAL G. COPELAND†,NANCY A. JENKINS†, AND JEREMY NATHANS*‡§¶i *Department of Molecular Biology and Genetics, §Department of Neuroscience, ¶Department of Ophthalmology, ‡Howard Hughes Medical Institute, Johns Hopkins University School of Medicine, Baltimore, MD 21205; and †Mammalian Genetics Laboratory, Advanced BioScience Laboratories Basic Research Program, National Cancer Institute–Frederick Cancer Research and Development Center, Frederick, MD 21702 Contributed by Jeremy Nathans, June 19, 1997 ABSTRACT A visual pigment-like protein, referred to as bovine RPE binds to all-trans but not 11-cis retinal and absorbs peropsin, has been identified by large-scale sequencing of both visible and ultraviolet light (8, 9). The sequences of cDNAs derived from human ocular tissues. The corresponding retinochrome and RGR opsin form a distinct and highly mRNA was found only in the eye, where it is localized to the divergent branch within the visual pigment family (6, 10). retinal pigment epithelium (RPE). Peropsin immunoreactiv- Whether retinochrome and RGR act as signal-transducing ity, visualized by light and electron microscopy, localizes the light receptors, participate in the visual cycle as retinal isomer- protein to the apical face of the RPE, and most prominently ases, or function in both capacities, is not known. to the microvilli that surround the photoreceptor outer seg- In the vertebrate eye, the RPE lies adjacent to the photo- ments. These observations suggest that peropsin may play a receptor cells and performs a number of functions critical for role in RPE physiology either by detecting light directly or by the viability and activity of the retina (11). -

Peptide, Peptidomimetic and Small Molecule Based Ligands Targeting Melanocortin Receptor System
PEPTIDE, PEPTIDOMIMETIC AND SMALL MOLECULE BASED LIGANDS TARGETING MELANOCORTIN RECEPTOR SYSTEM By ALEKSANDAR TODOROVIC A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY UNIVERSITY OF FLORIDA 2006 Copyright 2006 by Aleksandar Todorovic This document is dedicated to my family for everlasting support and selfless encouragement. ACKNOWLEDGMENTS I would like to thank and sincerely express my appreciation to all members, former and past, of Haskell-Luevano research group. First of all, I would like to express my greatest satisfaction by working with my mentor, Dr. Carrie Haskell-Luevano, whose guidance, expertise and dedication to research helped me reaching the point where I will continue the science path. Secondly, I would like to thank Dr. Ryan Holder who has taught me the principles of solid phase synthesis and initial strategies for the compounds design. I would like to thank Mr. Jim Rocca for the help and all necessary theoretical background required to perform proton 1-D NMR. In addition, I would like to thank Dr. Zalfa Abdel-Malek from the University of Cincinnati for the collaboration on the tyrosinase study project. Also, I would like to thank the American Heart Association for the Predoctoral fellowship that supported my research from 2004-2006. The special dedication and thankfulness go to my fellow graduate students within the lab and the department. iv TABLE OF CONTENTS page ACKNOWLEDGMENTS ................................................................................................ -

Functionality and Genetics of Melanocortin and Purinergic Receptors
University of Latvia Faculty of Biology Vita Ignatoviča Doctoral Thesis Functionality and genetics of melanocortin and purinergic receptors Promotion to the degree of Doctor of Biology Molecular Biology Supervisor: Dr. Biol. Jānis Kloviņš Riga, 2012 1 The doctoral thesis was carried out in University of Latvia, Faculty of Biology, Department of Molecular biology and Latvian Biomedical Reseach and Study centre. From 2007 to 2012 The research was supported by Latvian Council of Science (LZPSP10.0010.10.04), Latvian Research Program (4VPP-2010-2/2.1) and ESF funding (1DP/1.1.1.2.0/09/APIA/VIAA/150 and 1DP/1.1.2.1.2/09/IPIA/VIAA/004). The thesis contains the introduction, 9 chapters, 38 subchapters and reference list. Form of the thesis: collection of articles in biology with subdiscipline in molecular biology Supervisor: Dr. biol. Jānis Kloviņš Reviewers: 1) Dr. biol., Prof. Astrīda Krūmiņa, Latvian Biomedical Reseach and Study centre 2) Dr. biol., Prof. Ruta Muceniece, University of Latvia, Department of Medicine, Pharmacy program 3) PhD Med, Assoc.Prof.David Gloriam, University of Copenhagen, Department of Drug Design and Pharmacology The thesis will be defended at the public section of the Doctoral Commitee of Biology, University of Latvia, in the conference hall of Latvian Biomedical Research and Study centre on July 6th, 2012, at 11.00. The thesis is available at the Library of the University of Latvia, Kalpaka blvd. 4. This thesis is accepted of the commencement of the degree of Doctor of Biology on April 19th, 2012, by the Doctoral Commitee of Biology, University of Latvia. -

Constitutive Activation of G Protein-Coupled Receptors and Diseases: Insights Into Mechanisms of Activation and Therapeutics
Pharmacology & Therapeutics 120 (2008) 129–148 Contents lists available at ScienceDirect Pharmacology & Therapeutics journal homepage: www.elsevier.com/locate/pharmthera Associate editor: S. Enna Constitutive activation of G protein-coupled receptors and diseases: Insights into mechanisms of activation and therapeutics Ya-Xiong Tao ⁎ Department of Anatomy, Physiology and Pharmacology, 212 Greene Hall, College of Veterinary Medicine, Auburn University, Auburn, AL 36849, USA article info abstract The existence of constitutive activity for G protein-coupled receptors (GPCRs) was first described in 1980s. In Keywords: 1991, the first naturally occurring constitutively active mutations in GPCRs that cause diseases were reported G protein-coupled receptor Disease in rhodopsin. Since then, numerous constitutively active mutations that cause human diseases were reported Constitutively active mutation in several additional receptors. More recently, loss of constitutive activity was postulated to also cause Inverse agonist diseases. Animal models expressing some of these mutants confirmed the roles of these mutations in the Mechanism of activation pathogenesis of the diseases. Detailed functional studies of these naturally occurring mutations, combined Transgenic model with homology modeling using rhodopsin crystal structure as the template, lead to important insights into the mechanism of activation in the absence of crystal structure of GPCRs in active state. Search for inverse Abbreviations: agonists on these receptors will be critical for correcting the diseases cause by activating mutations in GPCRs. ADRP, autosomal dominant retinitis pigmentosa Theoretically, these inverse agonists are better therapeutics than neutral antagonists in treating genetic AgRP, Agouti-related protein AR, adrenergic receptor diseases caused by constitutively activating mutations in GPCRs. CAM, constitutively active mutant © 2008 Elsevier Inc. -
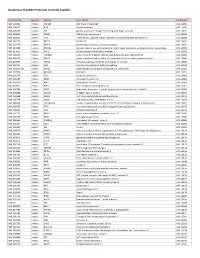
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

G Protein-Coupled Receptors: What a Difference a ‘Partner’ Makes
Int. J. Mol. Sci. 2014, 15, 1112-1142; doi:10.3390/ijms15011112 OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Review G Protein-Coupled Receptors: What a Difference a ‘Partner’ Makes Benoît T. Roux 1 and Graeme S. Cottrell 2,* 1 Department of Pharmacy and Pharmacology, University of Bath, Bath BA2 7AY, UK; E-Mail: [email protected] 2 Reading School of Pharmacy, University of Reading, Reading RG6 6UB, UK * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +44-118-378-7027; Fax: +44-118-378-4703. Received: 4 December 2013; in revised form: 20 December 2013 / Accepted: 8 January 2014 / Published: 16 January 2014 Abstract: G protein-coupled receptors (GPCRs) are important cell signaling mediators, involved in essential physiological processes. GPCRs respond to a wide variety of ligands from light to large macromolecules, including hormones and small peptides. Unfortunately, mutations and dysregulation of GPCRs that induce a loss of function or alter expression can lead to disorders that are sometimes lethal. Therefore, the expression, trafficking, signaling and desensitization of GPCRs must be tightly regulated by different cellular systems to prevent disease. Although there is substantial knowledge regarding the mechanisms that regulate the desensitization and down-regulation of GPCRs, less is known about the mechanisms that regulate the trafficking and cell-surface expression of newly synthesized GPCRs. More recently, there is accumulating evidence that suggests certain GPCRs are able to interact with specific proteins that can completely change their fate and function. These interactions add on another level of regulation and flexibility between different tissue/cell-types. -

Dimers of Serotonin Receptors: Impact on Ligand Affinity and Signaling
Dimers of serotonin receptors: impact on ligand affinity and signaling Luc Maroteaux, Catherine Béchade, Anne Roumier To cite this version: Luc Maroteaux, Catherine Béchade, Anne Roumier. Dimers of serotonin receptors: impact on ligand affinity and signaling. Biochimie, Elsevier, 2019, 161, pp.23-33. 10.1016/j.biochi.2019.01.009. hal- 01996206 HAL Id: hal-01996206 https://hal.archives-ouvertes.fr/hal-01996206 Submitted on 28 Jan 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Maroteaux et al., 1 Dimers of serotonin receptors: impact on ligand affinity and signaling. Luc Maroteaux1,2,3, Catherine Béchade1,2,3, and Anne Roumier1,2,3 Affiliations: 1: INSERM UMR-S839, S1270, Paris, 75005, France; 2Sorbonne Université, Paris, 75005, France; 3Institut du Fer à Moulin, Paris, 75005, France. Running title: Dimers of serotonin receptors Correspondence should be addressed to: Luc Maroteaux INSERM UMR-S839, S1270, 17 rue du Fer a Moulin Paris, 75005, France; E-mail : [email protected] Abstract Membrane receptors often form complexes with other membrane proteins that directly interact with different effectors of the signal transduction machinery. G-protein-coupled receptors (GPCRs) were for long time considered as single pharmacological entities. -

G Protein-Coupled Receptors
S.P.H. Alexander et al. The Concise Guide to PHARMACOLOGY 2015/16: G protein-coupled receptors. British Journal of Pharmacology (2015) 172, 5744–5869 THE CONCISE GUIDE TO PHARMACOLOGY 2015/16: G protein-coupled receptors Stephen PH Alexander1, Anthony P Davenport2, Eamonn Kelly3, Neil Marrion3, John A Peters4, Helen E Benson5, Elena Faccenda5, Adam J Pawson5, Joanna L Sharman5, Christopher Southan5, Jamie A Davies5 and CGTP Collaborators 1School of Biomedical Sciences, University of Nottingham Medical School, Nottingham, NG7 2UH, UK, 2Clinical Pharmacology Unit, University of Cambridge, Cambridge, CB2 0QQ, UK, 3School of Physiology and Pharmacology, University of Bristol, Bristol, BS8 1TD, UK, 4Neuroscience Division, Medical Education Institute, Ninewells Hospital and Medical School, University of Dundee, Dundee, DD1 9SY, UK, 5Centre for Integrative Physiology, University of Edinburgh, Edinburgh, EH8 9XD, UK Abstract The Concise Guide to PHARMACOLOGY 2015/16 provides concise overviews of the key properties of over 1750 human drug targets with their pharmacology, plus links to an open access knowledgebase of drug targets and their ligands (www.guidetopharmacology.org), which provides more detailed views of target and ligand properties. The full contents can be found at http://onlinelibrary.wiley.com/doi/ 10.1111/bph.13348/full. G protein-coupled receptors are one of the eight major pharmacological targets into which the Guide is divided, with the others being: ligand-gated ion channels, voltage-gated ion channels, other ion channels, nuclear hormone receptors, catalytic receptors, enzymes and transporters. These are presented with nomenclature guidance and summary information on the best available pharmacological tools, alongside key references and suggestions for further reading. -

Rods Contribute to the Light-Induced Phase Shift of The
Rods contribute to the light-induced phase shift of the retinal clock in mammals Hugo Calligaro, Christine Coutanson, Raymond Najjar, Nadia Mazzaro, Howard Cooper, Nasser Haddjeri, Marie-Paule Felder-Schmittbuhl, Ouria Dkhissi-Benyahya To cite this version: Hugo Calligaro, Christine Coutanson, Raymond Najjar, Nadia Mazzaro, Howard Cooper, et al.. Rods contribute to the light-induced phase shift of the retinal clock in mammals. PLoS Biology, Public Library of Science, 2019, 17 (3), pp.e2006211. 10.1371/journal.pbio.2006211. inserm-02137592 HAL Id: inserm-02137592 https://www.hal.inserm.fr/inserm-02137592 Submitted on 23 May 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. RESEARCH ARTICLE Rods contribute to the light-induced phase shift of the retinal clock in mammals Hugo Calligaro1, Christine Coutanson1, Raymond P. Najjar2,3, Nadia Mazzaro4, Howard M. Cooper1, Nasser Haddjeri1, Marie-Paule Felder-Schmittbuhl4, Ouria Dkhissi- Benyahya1* 1 Univ Lyon, Universite Claude Bernard Lyon 1, Inserm, Stem Cell and Brain Research Institute, Bron, France, -

Multi-Functionality of Proteins Involved in GPCR and G Protein Signaling: Making Sense of Structure–Function Continuum with In
Cellular and Molecular Life Sciences (2019) 76:4461–4492 https://doi.org/10.1007/s00018-019-03276-1 Cellular andMolecular Life Sciences REVIEW Multi‑functionality of proteins involved in GPCR and G protein signaling: making sense of structure–function continuum with intrinsic disorder‑based proteoforms Alexander V. Fonin1 · April L. Darling2 · Irina M. Kuznetsova1 · Konstantin K. Turoverov1,3 · Vladimir N. Uversky2,4 Received: 5 August 2019 / Revised: 5 August 2019 / Accepted: 12 August 2019 / Published online: 19 August 2019 © Springer Nature Switzerland AG 2019 Abstract GPCR–G protein signaling system recognizes a multitude of extracellular ligands and triggers a variety of intracellular signal- ing cascades in response. In humans, this system includes more than 800 various GPCRs and a large set of heterotrimeric G proteins. Complexity of this system goes far beyond a multitude of pair-wise ligand–GPCR and GPCR–G protein interactions. In fact, one GPCR can recognize more than one extracellular signal and interact with more than one G protein. Furthermore, one ligand can activate more than one GPCR, and multiple GPCRs can couple to the same G protein. This defnes an intricate multifunctionality of this important signaling system. Here, we show that the multifunctionality of GPCR–G protein system represents an illustrative example of the protein structure–function continuum, where structures of the involved proteins represent a complex mosaic of diferently folded regions (foldons, non-foldons, unfoldons, semi-foldons, and inducible foldons). The functionality of resulting highly dynamic conformational ensembles is fne-tuned by various post-translational modifcations and alternative splicing, and such ensembles can undergo dramatic changes at interaction with their specifc partners. -

Melanopsin: an Opsin in Melanophores, Brain, and Eye
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 340–345, January 1998 Neurobiology Melanopsin: An opsin in melanophores, brain, and eye IGNACIO PROVENCIO*, GUISEN JIANG*, WILLEM J. DE GRIP†,WILLIAM PA¨R HAYES‡, AND MARK D. ROLLAG*§ *Department of Anatomy and Cell Biology, Uniformed Services University of the Health Sciences, Bethesda, MD 20814; †Institute of Cellular Signaling, University of Nijmegen, 6500 HB Nijmegen, The Netherlands; and ‡Department of Biology, The Catholic University of America, Washington, DC 20064 Edited by Jeremy Nathans, Johns Hopkins University School of Medicine, Baltimore, MD, and approved November 5, 1997 (received for review September 16, 1997) ABSTRACT We have identified an opsin, melanopsin, in supernatants subjected to SDSyPAGE analysis and subse- photosensitive dermal melanophores of Xenopus laevis. Its quent electroblotting onto a poly(vinylidene difluoride) mem- deduced amino acid sequence shares greatest homology with brane. The blot was probed with a 1:2,000 dilution of antisera cephalopod opsins. The predicted secondary structure of (CERN 886) raised against bovine rhodopsin and detected by melanopsin indicates the presence of a long cytoplasmic tail enhanced chemiluminescence. with multiple putative phosphorylation sites, suggesting that cDNA Library Screen. A X. laevis dermal melanophore this opsin’s function may be finely regulated. Melanopsin oligo(dT) cDNA library was screened with a mixture of mRNA is expressed in hypothalamic sites thought to contain 32P-labeled probes. Probes were synthesized by random prim- deep brain photoreceptors and in the iris, a structure known ing of TA-cloned (Invitrogen) fragments of X. laevis rhodopsin to be directly photosensitive in amphibians. Melanopsin mes- (7) (759 bp) and violet opsin (8) (279 bp) cDNAs. -

Journal 37.Pdf
Biomaterials 34 (2013) 1911e1920 Contents lists available at SciVerse ScienceDirect Biomaterials journal homepage: www.elsevier.com/locate/biomaterials The activation of directional stem cell motility by green light-emitting diode irradiation Wei-Kee Ong a,1, How-Foo Chen b,1, Cheng-Ting Tsai b, Yun-Ju Fu a, Yi-Shan Wong a, Da-Jen Yen c, Tzu-Hao Chang d,e, Hsien-Da Huang d, Oscar Kuang-Sheng Lee f, Shu Chien g, Jennifer Hui-Chun Ho a,h,i,* a Center for Stem Cell Research, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan b Institute of Biophotonics, National Yang-Ming University, Taipei, Taiwan c Department of Material Science and Engineering, National Tsing Hua University, HsinChu, Taiwan d Institute of Bioinformatics and Systems Biology, National Chiao Tung University, HsinChu, Taiwan e Graduate Institute of Biomedical Informatics, Taipei Medical University, Taiwan f Institute of Clinical Medicine, National Yang-Ming University, Taiwan g Departments of Bioengineering and Medicine, Institute of Engineering in Medicine, UC San Diego, La Jolla, CA, USA h Graduate Institute of Clinical Medicine, Taipei Medical University, Taipei, Taiwan i Department of Ophthalmology, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan article info abstract Article history: Light-emitting diode (LED) irradiation is potentially a photostimulator to manipulate cell behavior by Received 12 November 2012 opsin-triggered phototransduction and thermal energy supply in living cells. Directional stem cell Accepted 29 November 2012 motility is critical for the efficiency and specificity of stem cells in tissue repair. We explored that green Available online 19 December 2012 LED (530 nm) irradiation directed the human orbital fat stem cells (OFSCs) to migrate away from the LED light source through activation of extracellular signal-regulated kinases (ERK)/MAP kinase/p38 signaling Keywords: pathway.