Essential Knowledge to Manage Malolactic Fermentation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Flavour Management by Citric Acid Negative MLF Starter Cultures Authors: Carsten Heinemeyer, 2B Fermcontrol; Ulrich Hamm DLR-RNH Bad Kreuznach, Germany; Dr
Flavour management by citric acid negative MLF starter cultures Authors: Carsten Heinemeyer, 2B FermControl; Ulrich Hamm DLR-RNH Bad Kreuznach, Germany; Dr. Jürgen Fröhlich, University of Mainz General will be completed in all fermentations. Additionally, acetic acid and a certain residual profile of the metabolic intermediates The malo-lactic fermentation (MLF) is a commonly used remain in the wine. method to convert the aggressive malic acid to lactic acid. This conversion results in a reduction of the titratable acidity, which New MLF Starter Cultures is desired mainly in red wine but also in numerous white wines. The development of a citric acid negative MLF starter culture This process will be done either by the indigenous flora of LAB gave a new opportunity to avoid diacetyl and acetic acid or by selected strains of LAB e.g. Oenococcus oeni. During the production from the citric acid degradation. The fermentation MLF Oenococcus oeni does not convert only malic acid into lactic with a citric acid negative MLF starter culture will best preserve acid, numerous amounts of aroma active by-products will also be the varietal character of the wine. produced. The best known is diacetyl, which gives buttery notes to the wine. Diacetyl will be produced during the MLF by the In a comprehensive four year study at the wine research institute conversion of the natural citric acid in the wine by Oenococcus DLR-RNH Bad Kreuznach in Rhineland-Palatinate-Germany, oeni (Jan Clair Nielsen 1999). Apart from the diacetyl formation different MLF starter strains were tested under practical by Oenococcus oeni, numerous intermediate by-products are winemaking conditions. -

Acetoin Acetate Natural
aurochemicals.com Safety Data Sheet HEALTH 0 FLAMMABILITY 2 REACTIVITY 0 Section 1: PRODUCT AND COMPANY IDENTIFICATION 1.1 Product identifiers Product Name Acetoin Acetate, Natural Product Number 0352600 CAS-No. 4906-24-5 1.2 Product Recommended Use Flavorings 1.3 Preparation Information Company Aurochemicals 7 Nicoll Street Washingtonville, NY 10992- USA Telephone 845-496-6065 Fax 845-496-6248 1.4 Emergency Telephone Number 1-800-535-5053 International - 1-352-323-3500 collect Section 2: HAZARD(s) IDENTIFICATION 2.1 Classification of substance or mixture GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) Combustible Liquid (Category 4) H227 Skin irritation (Category 2) H315 Eye Irritation (Category 2A) H319 Specific Target Organ Toxicity -Single Exposure -Respiratory irritation H335 2.2 GHS Label Elements, Including precautionary statements Pictogram Signal Statement Warning Hazard Statement(s) H227 Combustible liquid H315 Causes skin irritation H319 Causes serious eye irritation H335 May cause respiratory irritation Precautionary Statement(s) PREVENTION P210 Keep away from flames and hot surfaces - No smoking P216 Avoid breathing dust/fumes/gas/mist/vapors/spray P280 Wear protective gloves/protective clothing/face protection 3526 ACETOIN ACETATE Nat sds.doc Printed: April 17, 2019 Page 1 of 7 aurochemicals.com Safety Data Sheet RESPONSE P305+351+338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present and easy to do. Continue rinsing. STORAGE P405 Store locked up DISPOSAL P501 Dispose of contents/container to an approved waste disposal plant 2.3 HNOC (Hazards not otherwise None classified or not covered by GHS Section 3: COMPOSITION / INFORMATION ON INGREDIENTS 3.1 Substances Synonym 2-Acetoxy-3-Butanone Formula C6H10O3 Molecular Weight 130.14 g/mol CAS-No 4906-24-5 EC-No 200-580-7 Hazardous Components Does not contain any hazardous substances Section 4: FIRST AID MEASURES 4.1 Description of first aid measures General Advice Consult a physician. -

Retention Indices for Frequently Reported Compounds of Plant Essential Oils
Retention Indices for Frequently Reported Compounds of Plant Essential Oils V. I. Babushok,a) P. J. Linstrom, and I. G. Zenkevichb) National Institute of Standards and Technology, Gaithersburg, Maryland 20899, USA (Received 1 August 2011; accepted 27 September 2011; published online 29 November 2011) Gas chromatographic retention indices were evaluated for 505 frequently reported plant essential oil components using a large retention index database. Retention data are presented for three types of commonly used stationary phases: dimethyl silicone (nonpolar), dimethyl sili- cone with 5% phenyl groups (slightly polar), and polyethylene glycol (polar) stationary phases. The evaluations are based on the treatment of multiple measurements with the number of data records ranging from about 5 to 800 per compound. Data analysis was limited to temperature programmed conditions. The data reported include the average and median values of retention index with standard deviations and confidence intervals. VC 2011 by the U.S. Secretary of Commerce on behalf of the United States. All rights reserved. [doi:10.1063/1.3653552] Key words: essential oils; gas chromatography; Kova´ts indices; linear indices; retention indices; identification; flavor; olfaction. CONTENTS 1. Introduction The practical applications of plant essential oils are very 1. Introduction................................ 1 diverse. They are used for the production of food, drugs, per- fumes, aromatherapy, and many other applications.1–4 The 2. Retention Indices ........................... 2 need for identification of essential oil components ranges 3. Retention Data Presentation and Discussion . 2 from product quality control to basic research. The identifi- 4. Summary.................................. 45 cation of unknown compounds remains a complex problem, in spite of great progress made in analytical techniques over 5. -

Ctsr, the Master Regulator of Stress-Response in Oenococcus
CtsR, the Master Regulator of Stress-Response in Oenococcus oeni, Is a Heat Sensor Interacting With ClpL1 Maud Darsonval, Frédérique Julliat, Tarek Msadek, Hervé Alexandre, Cosette Grandvalet To cite this version: Maud Darsonval, Frédérique Julliat, Tarek Msadek, Hervé Alexandre, Cosette Grandvalet. CtsR, the Master Regulator of Stress-Response in Oenococcus oeni, Is a Heat Sensor Interacting With ClpL1. Frontiers in Microbiology, Frontiers Media, 2018, 9, pp.1-14. 10.3389/fmicb.2018.03135. hal-01986589 HAL Id: hal-01986589 https://hal.archives-ouvertes.fr/hal-01986589 Submitted on 18 Jan 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution| 4.0 International License fmicb-09-03135 December 15, 2018 Time: 15:10 # 1 ORIGINAL RESEARCH published: 18 December 2018 doi: 10.3389/fmicb.2018.03135 CtsR, the Master Regulator of Stress-Response in Oenococcus oeni, Is a Heat Sensor Interacting With ClpL1 Maud Darsonval1†, Frédérique Julliat1†, Tarek Msadek2,3, Hervé Alexandre1,4 and Cosette Grandvalet1,5* 1 UMR -

Evolution of Superior Lactic Acid Bacteria
Enhanced Wine-Making Efficiency Through Fool-Proof Malolactic Fermentation: Evolution of Superior Lactic Acid Bacteria Alice Livingston Betteridge A thesis submitted for the degree of Doctor of Philosophy School of Agriculture, Food and Wine Faculty of Sciences The University of Adelaide January 2015 Table of Contents Table of Contents Summary ................................................................................................................................ iv Declaration of authorship ....................................................................................................... vi Acknowledgements .............................................................................................................. vii List of Figures ........................................................................................................................ ix List of Tables .......................................................................................................................... x Abbreviations and Symbols ................................................................................................... xi Chapter 1. Strategies to improve the stress tolerance of Oenococcus oeni to enable highly efficient malolactic fermentation ........................................................... 1 1.1 Abstract .................................................................................................................... 1 1.2 Introduction ............................................................................................................. -

Oenological Impact of the Hanseniaspora/Kloeckera Yeast Genus on Wines—A Review
fermentation Review Oenological Impact of the Hanseniaspora/Kloeckera Yeast Genus on Wines—A Review Valentina Martin, Maria Jose Valera , Karina Medina, Eduardo Boido and Francisco Carrau * Enology and Fermentation Biotechnology Area, Food Science and Technology Department, Facultad de Quimica, Universidad de la Republica, Montevideo 11800, Uruguay; [email protected] (V.M.); [email protected] (M.J.V.); [email protected] (K.M.); [email protected] (E.B.) * Correspondence: [email protected]; Tel.: +598-292-481-94 Received: 7 August 2018; Accepted: 5 September 2018; Published: 10 September 2018 Abstract: Apiculate yeasts of the genus Hanseniaspora/Kloeckera are the main species present on mature grapes and play a significant role at the beginning of fermentation, producing enzymes and aroma compounds that expand the diversity of wine color and flavor. Ten species of the genus Hanseniaspora have been recovered from grapes and are associated in two groups: H. valbyensis, H. guilliermondii, H. uvarum, H. opuntiae, H. thailandica, H. meyeri, and H. clermontiae; and H. vineae, H. osmophila, and H. occidentalis. This review focuses on the application of some strains belonging to this genus in co-fermentation with Saccharomyces cerevisiae that demonstrates their positive contribution to winemaking. Some consistent results have shown more intense flavors and complex, full-bodied wines, compared with wines produced by the use of S. cerevisiae alone. Recent genetic and physiologic studies have improved the knowledge of the Hanseniaspora/Kloeckera species. Significant increases in acetyl esters, benzenoids, and sesquiterpene flavor compounds, and relative decreases in alcohols and acids have been reported, due to different fermentation pathways compared to conventional wine yeasts. -

An Original Method for Producing Acetaldehyde and Diacetyl by Yeast
b r a z i l i a n j o u r n a l o f m i c r o b i o l o g y 4 7 (2 0 1 6) 949–954 ht tp://www.bjmicrobiol.com.br/ Industrial Microbiology An original method for producing acetaldehyde and diacetyl by yeast fermentation a a,∗ a b a,c Irina Rosca , Anca Roxana Petrovici , Mihai Brebu , Irina Stoica , Bogdan Minea , a Narcisa Marangoci a “Petru Poni” Institute of Macromolecular Chemistry, Advanced Research Center for Bionanoconjugates and Biopolymers, Aleea GrigoreGhica Voda, Iasi, Romania b SC Zeelandia SRL, R&D Department, Valea Lupului, Iasi, Romania c “Grigore T. Popa” University of Medicine and Pharmacy, Iasi, Romania a r a t i c l e i n f o b s t r a c t Article history: In this study a natural culture medium that mimics the synthetic yeast peptone glucose Received 2 June 2015 medium used for yeast fermentations was designed to screen and select yeasts capable Accepted 29 February 2016 of producing high levels of diacetyl and acetaldehyde. The presence of whey powder and Available online 25 July 2016 sodium citrate in the medium along with manganese and magnesium sulfate enhanced Associate Editor: Jorge Gonzalo both biomass and aroma development. A total of 52 yeasts strains were cultivated in two Farias Avendano different culture media, namely, yeast peptone glucose medium and yeast acetaldehyde- diacetyl medium. The initial screening of the strains was based on the qualitative reaction Keywords: of the acetaldehyde with Schiff’s reagent (violet color) and diacetyl with Brady’s reagent Acetaldehyde (yellow precipitate). -

An Unrecognized Hazard in E-Cigarette Vapor: Preliminary Quantification of Methylglyoxal Formation from Propylene Glycol in E-Cigarettes
International Journal of Environmental Research and Public Health Article An Unrecognized Hazard in E-Cigarette Vapor: Preliminary Quantification of Methylglyoxal Formation from Propylene Glycol in E-Cigarettes Parham Azimi 1, Zahra Keshavarz 1, Marianne Lahaie Luna 1,2, Jose Guillermo Cedeno Laurent 1 , Jose Vallarino 1, David C. Christiani 1 and Joseph G. Allen 1,* 1 Department of Environmental Health, Harvard T. H. Chan School of Public Health, Boston, MA 02115, USA; [email protected] (P.A.); [email protected] (Z.K.); [email protected] (M.L.L.); [email protected] (J.G.C.L.); [email protected] (J.V.); [email protected] (D.C.C.) 2 Occupational & Environmental Health Division, Dalla Lana School of Public Health, University of Toronto, Toronto, ON M5T 3M7, Canada * Correspondence: [email protected] Abstract: Up to 95% of the liquid volume in an e-cigarette consists of propylene glycol. Previous research has shown that propylene glycol can generate diacetyl and formaldehyde when heated. New research shows that propylene glycol can also generate methylglyoxal, an alpha di-carbonyl compound recently shown to cause epithelial necrosis at even lower concentrations than diacetyl, the flavoring chemical associated with bronchiolitis obliterans (“Popcorn Lung”). We analyzed chemical emissions from 13 JUUL pod flavors. Diacetyl and methylglyoxal was detected in 100% of samples 3 3 with median concentration (range) of 20 µg/m (less than limit of quantification: 54 µg/m ) and 4219 µg/m3 (677–15,342 µg/m3), respectively. We also detected acetaldehyde (median concentration: Citation: Azimi, P.; Keshavarz, Z.; 341 µg/m3) and propionaldehyde (median concentration: 87 µg/m3) in all samples. -

Microorganisms in Fermented Apple Beverages: Current Knowledge And
Microorganisms in Fermented Apple Beverages: Current Knowledge and Future Directions Fabien Cousin, Rozenn Le Guellec, Margot Schlusselhuber, Marion Dalmasso, Jean-Marie Laplace, Marina Cretenet To cite this version: Fabien Cousin, Rozenn Le Guellec, Margot Schlusselhuber, Marion Dalmasso, Jean-Marie Laplace, et al.. Microorganisms in Fermented Apple Beverages: Current Knowledge and Future Directions. Microorganisms, MDPI, 2017, 5 (3), pp.39. 10.3390/microorganisms5030039. hal-02089085 HAL Id: hal-02089085 https://hal.archives-ouvertes.fr/hal-02089085 Submitted on 3 Apr 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. microorganisms Review Microorganisms in Fermented Apple Beverages: Current Knowledge and Future Directions Fabien J. Cousin ID , Rozenn Le Guellec, Margot Schlusselhuber, Marion Dalmasso ID , Jean-Marie Laplace and Marina Cretenet * Aliments Bioprocédés Toxicologie Environnements, Normandy University, UNICAEN, UNIROUEN, 14000 Caen, France; [email protected] (F.J.C.); [email protected] (R.L.G.); [email protected] (M.S.); [email protected] (M.D.); [email protected] (J.-M.L.) * Correspondence: [email protected]; Tel: +33-231-565-785 Received: 27 June 2017; Accepted: 21 July 2017; Published: 25 July 2017 Abstract: Production of fermented apple beverages is spread all around the world with specificities in each country. -
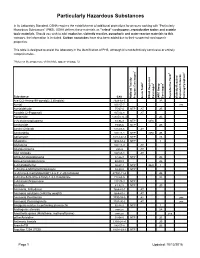
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

Changes in the Composition of the Lactic Acid Bacteria Behavior And
fermentation Article Changes in the Composition of the Lactic Acid Bacteria Behavior and the Diversity of Oenococcus oeni Isolated from Red Wines Supplemented with Selected Grape Phenolic Compounds Ingrid Collombel 1 , Francisco M. Campos 1 and Tim Hogg 1,2,* 1 CBQF—Centro de Biotecnologia e Química Fina—Laboratório Associado, Escola Superior de Biotecnologia, Universidade Católica Portuguesa, 4202-401 Porto, Portugal; [email protected] (I.C.); [email protected] (F.M.C.) 2 Plataforma de Inovação da Vinha e do Vinho, Universidade dos Tras-os-Montes e Alto Douro, 5001-801 Vila Real, Portugal * Correspondence: [email protected]; Tel. +351-225-580-066 Received: 19 November 2018; Accepted: 18 December 2018; Published: 20 December 2018 Abstract: Phenolic compounds are important components of wine and are known to have an impact on the physiology of wine microbes. The influence of specific sub-sets of phenolic compounds on the growth and metabolism of lactic acid bacteria (LAB) and on the diversity of Oenococcus oeni in inoculated and non-inoculated red wines was investigated during malolactic fermentation (MLF) and subsequent storage. Representative O. oeni strains from wines treated with flavonols and trans-resveratrol were isolated and analyzed by pulsed-field gel electrophoresis of rare restriction enzyme digests (REA-PFGE). 28 days after MLF initiation, strains from all samples had entered the death phase, except those supplemented with trans-resveratrol. In the non-inoculated samples, the onset of lactic acid production was apparently delayed by all compounds tested, except for the flavan-3-ols. Increased levels of phenolics also delayed citrate consumption in inoculated samples. -

Genomic Analysis of Oenococcus Oeni PSU-1 and Its Relevance to Winemaking
FEMS Microbiology Reviews 29 (2005) 465–475 www.fems-microbiology.org Genomic analysis of Oenococcus oeni PSU-1 and its relevance to winemaking David A. Mills a,*, Helen Rawsthorne a,1, Courtney Parker a,2, Dafna Tamir a,3, Kira Makarova b a Department of Viticulture and Enology, University of California, Davis, CA 95616, United States b National Center for Biotechnology Information, National Library of Medicine, National Institutes of Health, Bethesda, MD 20894, United States Received 24 February 2005; accepted 23 April 2005 First published online 28 August 2005 Abstract Oenococcus oeni is an acidophilic member of the Leuconostoc branch of lactic acid bacteria indigenous to wine and similar envi- ronments. O. oeni is commonly responsible for the malolactic fermentation in wine and due to its positive contribution is frequently used as a starter culture to promote malolactic fermentation. In collaboration with the Lactic Acid Bacteria Genome Consortium the genome sequence of O. oeni PSU-1 has been determined. The complete genome is 1,780,517 nt with a GC content of 38%. 1701 ORFs could be predicted from the sequence of which 75% were functionally classified. Consistent with its classification as an obli- gately heterofermentative lactic acid bacterium the PSU-1 genome encodes all the enzymes for the phosphoketolase pathway. More- over, genes related to flavor modification in wine, such as malolactic fermentation capacity and citrate utilization were readily identified. The completion of the O. oeni genome marks a significant new phase for wine-related research on lactic acid bacteria in which the physiology, genetic diversity and performance of O.