Stimulation Cells Is a Potent Regulator of T Cell Adhesion Molecule 1 On
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Screening and Identification of Key Biomarkers in Clear Cell Renal Cell Carcinoma Based on Bioinformatics Analysis
bioRxiv preprint doi: https://doi.org/10.1101/2020.12.21.423889; this version posted December 23, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Screening and identification of key biomarkers in clear cell renal cell carcinoma based on bioinformatics analysis Basavaraj Vastrad1, Chanabasayya Vastrad*2 , Iranna Kotturshetti 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karanataka, India. 3. Department of Ayurveda, Rajiv Gandhi Education Society`s Ayurvedic Medical College, Ron, Karnataka 562209, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India bioRxiv preprint doi: https://doi.org/10.1101/2020.12.21.423889; this version posted December 23, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract Clear cell renal cell carcinoma (ccRCC) is one of the most common types of malignancy of the urinary system. The pathogenesis and effective diagnosis of ccRCC have become popular topics for research in the previous decade. In the current study, an integrated bioinformatics analysis was performed to identify core genes associated in ccRCC. An expression dataset (GSE105261) was downloaded from the Gene Expression Omnibus database, and included 26 ccRCC and 9 normal kideny samples. Assessment of the microarray dataset led to the recognition of differentially expressed genes (DEGs), which was subsequently used for pathway and gene ontology (GO) enrichment analysis. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

Combination Immune Therapy
Combination Immune Therapy Translational Medicine Plenary SWOG April 17, 2017 San Francisco T‐cell Activation, Proliferation, and Function is Controlled by Multiple Agonist and Antagonist Signals 1. Co‐stimulation via CD28 ligation 2. CTLA‐4 ligation on activated T 3. T cell function in tissue is subject transduces T cell activating signals cells down‐regulates T cell to feedback inhibition responses T cell Functional Block T cell Activation T cell Activation T cell Proliferative Block T cell T cell APC T cell CTLA‐4 T cell CTLA‐4 TCR TCR PD‐1 PD‐1 PD‐1 IFN‐gamma TCR CD28 Cytokines TCR CD4 MHC MHC PD‐‐L1 MHC B7 MHC CD28 B7 PD‐‐L1 Tumor or Tumor or APC APC Immune cell B7 immune cell APC NK Presence of PD-L1 or TILs1 PPD-L1 /TIL+ PD-L1/TIL + + PD-L1 /TIL+ + PD-L1 /TIL PD-L1 /TIL D-L1 /TIL+ Schalper and Rimm, Yale University NSCLC PD-L1/TIL 45% 17% 26% 12% Type 1 Type 2 Type 3 Type 4 45% 41% 13% 1% Melanoma45% 17% 26% 12% Taube et al Type 1 Type 2 Type 3 Type 4 Tumor‐specific T cells are contained in the PD‐1+ TIL population and are functional after in vitro culture Spectrum of PD‐1/PD‐L1 Antagonist Activity Active • Melanoma Minimal to no activity • Renal cancer (clear cell and non‐clear cell) • Prostate cancer • NSCLC – adenocarcinoma and squamous cell • MMR+ (MSS) colon cancer • Small cell lung cancer • Myeloma • Head and neck cancer • Pancreatic cancer • Gastric and gastroesophageal junction • MMR‐repair deficient tumors (colon, cholangiocarcinoma) • Bladder • Triple negative breast cancer • Ovarian Major PD‐1/PD‐L1 antagonists • Hepatocellular -
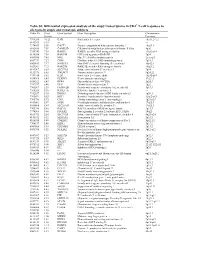
Table S1| Differential Expression Analysis of the Atopy Transcriptome
Table S1| Differential expression analysis of the atopy transcriptome in CD4+ T-cell responses to allergens in atopic and nonatopic subjects Probe ID S.test Gene Symbol Gene Description Chromosome Statistic Location 7994280 10.32 IL4R Interleukin 4 receptor 16p11.2-12.1 8143383 8.95 --- --- --- 7974689 8.50 DACT1 Dapper, antagonist of beta-catenin, homolog 1 14q23.1 8102415 7.59 CAMK2D Calcium/calmodulin-dependent protein kinase II delta 4q26 7950743 7.58 RAB30 RAB30, member RAS oncogene family 11q12-q14 8136580 7.54 RAB19B GTP-binding protein RAB19B 7q34 8043504 7.45 MAL Mal, T-cell differentiation protein 2cen-q13 8087739 7.27 CISH Cytokine inducible SH2-containing protein 3p21.3 8000413 7.17 NSMCE1 Non-SMC element 1 homolog (S. cerevisiae) 16p12.1 8021301 7.15 RAB27B RAB27B, member RAS oncogene family 18q21.2 8143367 6.83 SLC37A3 Solute carrier family 37 member 3 7q34 8152976 6.65 TMEM71 Transmembrane protein 71 8q24.22 7931914 6.56 IL2R Interleukin 2 receptor, alpha 10p15-p14 8014768 6.43 PLXDC1 Plexin domain containing 1 17q21.1 8056222 6.43 DPP4 Dipeptidyl-peptidase 4 (CD26) 2q24.3 7917697 6.40 GFI1 Growth factor independent 1 1p22 7903507 6.39 FAM102B Family with sequence similarity 102, member B 1p13.3 7968236 5.96 RASL11A RAS-like, family 11, member A --- 7912537 5.95 DHRS3 Dehydrogenase/reductase (SDR family) member 3 1p36.1 7963491 5.83 KRT1 Keratin 1 (epidermolytic hyperkeratosis) 12q12-q13 7903786 5.72 CSF1 Colony stimulating factor 1 (macrophage) 1p21-p13 8019061 5.67 SGSH N-sulfoglucosamine sulfohydrolase (sulfamidase) 17q25.3 -

Bioinformatics Method Identifies Potential Biomarkers of Dilated Cardiomyopathy in a Human Induced Pluripotent Stem Cell‑Derived Cardiomyocyte Model
EXPERIMENTAL AND THERAPEUTIC MEDICINE 14: 2771-2778, 2017 Bioinformatics method identifies potential biomarkers of dilated cardiomyopathy in a human induced pluripotent stem cell‑derived cardiomyocyte model YU ZHUANG1, YU-JIA GONG2, BEI-FEN ZHONG3, YI ZHOU1 and LI GONG4 1Department of Cardiovascular Surgery, Shanghai First People's Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200080; 2Stomatology Faculty, School of Medicine, Nantong University, Nantong, Jiangsu 226000; 3Department of Urology, Shanghai First People's Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200080; 4Department of Cardiothoracic Surgery, The Affiliated Huai'an Hospital of Xuzhou Medical University, Huai'an, Jiangsu 223002, P.R. China Received February 19, 2016; Accepted February 10, 2017 DOI: 10.3892/etm.2017.4850 Abstract. Dilated cardiomyopathy (DCM) is the most common crucial nodes in module 2, which were linked to each other. type of cardiomyopathy that account for the majority of heart In conclusion, several potential biomarkers for DCM were failure cases. The present study aimed to reveal the under- identified, such as MMP2, FLT1, CDH1, ITGB6, COL6A3, lying molecular mechanisms of DCM and provide potential COL6A1, LAMC2, PENK and APLNR. These genes may serve biomarkers for detection of this condition. The public dataset significant roles in DCM via involvement of various BPs, such of GSE35108 was downloaded, and 4 normal induced pluripo- as blood vessel and vasculature development and cell adhe- tent stem cell (iPSC)-derived cardiomyocytes (N samples) and sion, and the ECM-receptor interaction pathway. 4 DCM iPSC-derived cardiomyocytes (DCM samples) were utilized. Raw data were preprocessed, followed by identifica- Introduction tion of differentially expressed genes (DEGs) between N and DCM samples. -

Increased Expression of Cell Adhesion Molecule P-Selectin in Active Inflammatory Bowel Disease Gut: First Published As 10.1136/Gut.36.3.411 on 1 March 1995
Gut 1995; 36: 411-418 411 Increased expression of cell adhesion molecule P-selectin in active inflammatory bowel disease Gut: first published as 10.1136/gut.36.3.411 on 1 March 1995. Downloaded from G M Schurmann, A E Bishop, P Facer, M Vecchio, J C W Lee, D S Rampton, J M Polak Abstract proposed, entailing margination from the The pathogenic changes of inflammatory centreline of blood flow towards the vascular bowel disease (IBD) depend on migration wall, rolling, tethering to the endothelia, stable of circulating leucocytes into intestinal adhesion, and finally, transendothelial migra- tissues. Although leucocyte rolling and tion.1 Each of these steps involves specific fam- tenuous adhesion are probably regulated ilies of adhesion molecules, which are by inducible selectins on vascular expressed on endothelial cells and on circulat- endothelia, little is known about the ing cells as their counterparts and ligands.2 3 expression of these molecules in Crohn's The selectin family of adhesion molecules, disease and ulcerative colitis. Using which comprises E-selectin, P-selectin, and L- immunohistochemistry on surgically selectin, predominantly mediates the first steps resected specimens, this study investi- of cellular adhesion4 5 and several studies have gated endothelial P-selectin (CD62, gran- shown upregulation of E-selectin on activated ular membrane protein-140) in frozen endothelial cells in a variety oftissues6-8 includ- sections of histologically uninvolved ing the gut in patients with IBD.9 10 Little tissues adjacent to inflammation (Crohn's investigation has been made, however, of P- disease= 10; ulcerative colitis= 10), from selectin in normal and diseased gut, although highly inflamed areas (Crohn's its DNA was cloned and sequenced in 1989.11 disease=20; ulcerative colitis=13), and P-selectin (also known as PADGEM, from normal bowel (n=20). -

(Lilrs) on Human Neutrophils: Modulators of Infection and Immunity
MINI REVIEW published: 13 May 2020 doi: 10.3389/fimmu.2020.00857 Leukocyte Immunoglobulin-Like Receptors (LILRs) on Human Neutrophils: Modulators of Infection and Immunity Alexander L. Lewis Marffy and Alex J. McCarthy* MRC Centre for Molecular Bacteriology and Infection, Imperial College London, London, United Kingdom Neutrophils have a crucial role in defense against microbes. Immune receptors allow neutrophils to sense their environment, with many receptors functioning to recognize signs of infection and to promote antimicrobial effector functions. However, the neutrophil Edited by: Nicole Thielens, response must be tightly regulated to prevent excessive inflammation and tissue damage, UMR5075 Institut de Biologie and regulation is achieved by expression of inhibitory receptors that can raise activation Structurale (IBS), France thresholds. The leukocyte immunoglobulin-like receptor (LILR) family contain activating Reviewed by: and inhibitory members that can up- or down-regulate immune cell activity. New ligands Debby Burshtyn, University of Alberta, Canada and functions for LILR continue to emerge. Understanding the role of LILR in neutrophil Tamás Laskay, biology is of general interest as they can activate and suppress antimicrobial responses University of Lübeck, Germany of neutrophils and because several human pathogens exploit these receptors for immune *Correspondence: Alex J. McCarthy evasion. This review focuses on the role of LILR in neutrophil biology. We focus on the [email protected] current knowledge of LILR expression -

Role of Cell Adhesion Molecules of the Nectin-Like Family in Peripheral Nerve Myelination
Role of Cell Adhesion Molecules of the Nectin-like family in Peripheral Nerve Myelination: Nectin-like 1 and Nectin-like 2 are negative regulators of myelination by Schwann cells By MING-SHUO CHEN A dissertation submitted to the Graduate School - Newark Rutgers, The State University of New Jersey In partial fulfillment of the requirements For the Degree of Doctor of Philosophy Graduate Program in Biological Sciences Written under the direction of Patrice Maurel, Ph.D. And approved by Newark, New Jersey October 2017 © [2017] Ming-Shuo Chen ALL RIGHTS RESERVED ABSTRACT OF THE DISSERTATION Role of Cell Adhesion Molecules of the Nectin-like family in Peripheral Nerve Myelination: Nectin-like 1 and Nectin-like 2 are negative regulators of myelination by Schwann cells By Ming-Shuo Chen Dissertation director: Dr. Patrice Maurel Axo-glial interactions are critical for myelination and the domain organization of myelinated fibers. Cell adhesion molecules belonging to the Nectin like family, and in particular Necl-1 (axonal) and its heterophilic binding partner Necl-4 (Schwann cell), mediate these interactions along the internode. Using targeted shRNA-mediated knockdown, we show that the removal of axonal Necl- 1 promotes Schwann cell myelination in the in vitro DRG neuron/Schwann cell myelinating system. Conversely, over-expressing Necl-1 on the surface of DRG neuron axons results in an almost complete inability by Schwann cells to form myelin segments. Axons of superior cervical ganglion (SCG) neurons, which do not normally support the formation of myelin segments by Schwann cells, express higher levels of Necl-1 compared to DRG neurons. Knocking down Necl-1 in SCG neurons promotes myelination. -

The Poliovirus Receptor (CD155)
Cutting Edge: CD96 (Tactile) Promotes NK Cell-Target Cell Adhesion by Interacting with the Poliovirus Receptor (CD155) This information is current as Anja Fuchs, Marina Cella, Emanuele Giurisato, Andrey S. of September 27, 2021. Shaw and Marco Colonna J Immunol 2004; 172:3994-3998; ; doi: 10.4049/jimmunol.172.7.3994 http://www.jimmunol.org/content/172/7/3994 Downloaded from References This article cites 19 articles, 8 of which you can access for free at: http://www.jimmunol.org/content/172/7/3994.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 27, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2004 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. THE JOURNAL OF IMMUNOLOGY CUTTING EDGE Cutting Edge: CD96 (Tactile) Promotes NK Cell-Target Cell Adhesion by Interacting with the Poliovirus Receptor (CD155) Anja Fuchs, Marina Cella, Emanuele Giurisato, Andrey S. Shaw, and Marco Colonna1 The poliovirus receptor (PVR) belongs to a large family of activating receptor DNAM-1, also called CD226 (6, 7). -

Supplemental Text and Figures
Supplemental Materials and Methods Experimental bone metastasis assay Primary PCa cells were sorted by GFP marker from mTmG+ tumors, and 105 cells in 20μL PBS were injected using Hamilton syringe into the tibia of 6-week old NSG mice. Mice were monitored biweekly for moribund signs for euthanasia and organ harvest. Noninvasive mouse and ex vivo imaging MRI imaging with Bruker ICON and fluorescence imaging of fresh organs with metastasis enumeration were recently described (Lu et al. 2017). Primary prostate cell sphere formation assay Isolate of primary cells from prostate, culture and counting of prostatospheres on Matrigel were performed as described (Lukacs et al. 2010). For organoid culture assay, we followed a published matrigel embedding method (Chua et al. 2014). RNA-Seq and differential gene expression Total RNA was isolated from prostate tumors using Direct-zol RNA MiniPrep Kit (Zymo Research) and processed for stranded total RNA-Seq using Illumina HiSeq 4000 at Sequencing and Microarray Facility at MD Anderson Cancer Center. The differential expression analysis was performed using the DESeq2 package of R. P-values obtained after multiple binomial tests were adjusted using BH method. Significant genes are defined by using a cut-off of 0.05 on the BH corrected p-value and an absolute log2 fold change value of at least 1.5. Histology and western blot H&E stain, immunohistochemical (IHC) and western blot were performed as previously described (Ding et al. 2011; Wang et al. 2016). Primary antibodies for IHC include Ki67 (Fisher, RM-9106-S1), cleaved caspase 3 (Cell Signaling Technology aka CST, 9661), cyclin D1 (Fisher, clone SP4), TGFBR2 (Abcam, ab61213), BMPR2 (Abcam, ab130206), AR (EMD Millipore, 06-680), phospho- Akt (CST, 4060), GFP (CST, 2956), E-Cadherin (CST, 14472). -

Cell Adhesion Molecules in Normal Skin and Melanoma
biomolecules Review Cell Adhesion Molecules in Normal Skin and Melanoma Cian D’Arcy and Christina Kiel * Systems Biology Ireland & UCD Charles Institute of Dermatology, School of Medicine, University College Dublin, D04 V1W8 Dublin, Ireland; [email protected] * Correspondence: [email protected]; Tel.: +353-1-716-6344 Abstract: Cell adhesion molecules (CAMs) of the cadherin, integrin, immunoglobulin, and selectin protein families are indispensable for the formation and maintenance of multicellular tissues, espe- cially epithelia. In the epidermis, they are involved in cell–cell contacts and in cellular interactions with the extracellular matrix (ECM), thereby contributing to the structural integrity and barrier for- mation of the skin. Bulk and single cell RNA sequencing data show that >170 CAMs are expressed in the healthy human skin, with high expression levels in melanocytes, keratinocytes, endothelial, and smooth muscle cells. Alterations in expression levels of CAMs are involved in melanoma propagation, interaction with the microenvironment, and metastasis. Recent mechanistic analyses together with protein and gene expression data provide a better picture of the role of CAMs in the context of skin physiology and melanoma. Here, we review progress in the field and discuss molecular mechanisms in light of gene expression profiles, including recent single cell RNA expression information. We highlight key adhesion molecules in melanoma, which can guide the identification of pathways and Citation: D’Arcy, C.; Kiel, C. Cell strategies for novel anti-melanoma therapies. Adhesion Molecules in Normal Skin and Melanoma. Biomolecules 2021, 11, Keywords: cadherins; GTEx consortium; Human Protein Atlas; integrins; melanocytes; single cell 1213. https://doi.org/10.3390/ RNA sequencing; selectins; tumour microenvironment biom11081213 Academic Editor: Sang-Han Lee 1. -

CEACAM1 As a Multi-Purpose Target for Cancer Immunotherapy
CEACAM1 as a multi-purpose target for cancer immunotherapy The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Dankner, Matthew, Scott D. Gray-Owen, Yu-Hwa Huang, Richard S. Blumberg, and Nicole Beauchemin. 2017. “CEACAM1 as a multi- purpose target for cancer immunotherapy.” Oncoimmunology 6 (7): e1328336. doi:10.1080/2162402X.2017.1328336. http:// dx.doi.org/10.1080/2162402X.2017.1328336. Published Version doi:10.1080/2162402X.2017.1328336 Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:34375168 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA ONCOIMMUNOLOGY 2017, VOL. 6, NO. 7, e1328336 (16 pages) https://doi.org/10.1080/2162402X.2017.1328336 REVIEW CEACAM1 as a multi-purpose target for cancer immunotherapy Matthew Dankner a, Scott D. Gray-Owen b, Yu-Hwa Huangc, Richard S. Blumberg c, and Nicole Beauchemin a aGoodman Cancer Research Centre, McGill University, Montreal, QC, Canada; bDepartment of Molecular Genetics, University of Toronto, Toronto, ON, Canada; cDivision of Gastroenterology, Hepatology, and Endoscopy, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, USA ABSTRACT ARTICLE HISTORY CEACAM1 is an extensively studied cell surface molecule with established functions in multiple cancer Received 17 February 2017 types, as well as in various compartments of the immune system. Due to its multi-faceted role as a Revised 3 May 2017 recently appreciated immune checkpoint inhibitor and tumor marker, CEACAM1 is an attractive target for Accepted 5 May 2017 cancer immunotherapy.