Lanthanide Doped Wide Band Gap Semiconductors: Intra-4F Luminescence and Lattice Location Studies
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Amphoteric Arsenic in Gan
Reprint from APPLIED PHYSICS LETTERS 90 (2007) 181934 Amphoteric arsenic in GaN U. Wahla Instituto Tecnológico e Nuclear, Estrada Nacional 10, 2686-953 Sacavém, Portugal, and Centro de Física Nuclear da Universidade de Lisboa, Avenida Professor Gama Pinto 2, 1649-003 Lisboa, Portugal J. G. Correia Instituto Tecnológico e Nuclear, Estrada Nacional 10, 2686-953 Sacavém, Portugal, and Centro de Física Nuclear da Universidade de Lisboa, Avenida Professor Gama Pinto 2, 1649-003 Lisboa, Portugal, and CERN-PH, 1211 Geneva 23, Switzerland J.P. Araújo Departamento de Física, Universidade do Porto, Rua do Campo Alegre 687, 4169-007 Porto, Portugal E. Rita and J.C. Soares Centro de Física Nuclear da Universidade de Lisboa, Avenida Professor Gama Pinto 2, 1649-003 Lisboa, Portugal The ISOLDE collaboration CERN-PH, 1211 Geneva 23, Switzerland (Received 15 March 2007; accepted 11 April 2007; published online 4 May 2007) We have determined the lattice location of implanted arsenic in GaN by means of conversion electron emission channel- ing from radioactive 73As. We give direct evidence that As is an amphoteric impurity, thus settling the long-standing ques- tion as to whether it prefers cation or anion sites in GaN. The amphoteric character of As and the fact that AsGa “anti- sites” are not minority defects provide additional aspects to be taken into account for an explanantion of the so-called “miscibility gap” in ternary GaAs1−x Nx compounds, which cannot be grown with a single phase for values of x in the range 0.1<x<0.99. © 2007 American Institute of Physics. [DOI: 10.1063/1.2736299] The growth and properties of ternary semiconductors of scale with the amount of radioactive 71As and 72As resulting the nitride family have been under intense investigation ever from radioactive decay. -
Mission Statement
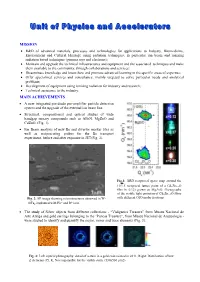
MISSION R&D of advanced materials, processes and technologies for applications to Industry, Biomedicine, Environment and Cultural Heritage using radiation techniques, in particular ion beam and ionizing radiation based techniques (gamma rays and electrons); Maintain and upgrade the technical infrastructures and equipment and the associated techniques and make them available to the community, through collaborations and services; Disseminate knowledge and know-how and promote advanced learning in the specific areas of expertise; Offer specialized services and consultancy, mainly targeted to solve particular needs and analytical problems; Development of equipment using ionizing radiation for industry and research; Technical assistance to the industry. MAIN ACHIEVEMENTS A new integrated pin-diode pre-amplifier particle detection system and the upgrade of the external ion beam line. Structural, compositional and optical studies of wide bandgap ternary compounds such as AlInN, MgZnO and CdZnO. (Fig. 1). Ion Beam analysis of new Be and divertor marker tiles as well as reciprocating probes for the Be transport experiment, before and after exposure in JET(Fig. 2). Fig.1: XRD reciprocal space map around the 10 1 5 reciprocal lattice point of a CdxZn1-xO film (x=0.12) grown on MgZnO. Photographs of the visible light emission of Cd Zn O films x 1-x Fig. 2: SE image showing microstructures observed in W- with different CdO molar fractions. + + 10Tap implanted with He and D ions. The study of Silver objects from different collections - “Vidigueira Treasure” from Museu Nacional de Arte Antiga and gold earrings belonging to the “Pancas Treasure”, from Museu Nacional de Arqueologia - were studied to identify and quantify the major, minor and trace elements (Fig. -

ISOLDE Newsletter Spring 2015
Spring 2015 ISOLDE newsletter http://isolde.web.cern.ch/ optical model and DWBA using FRESCO for Introduction several basic cases and using TWOFNR code for transfer (d,p) reactions. To learn The year 2014 was characterized by the more about the courses given in 2013 and consolidation and realisation of many 2014 one can visit, pending tasks. The year started with the http://isolde.web.cern.ch/isolde-schools- technical team working hard and counting and-courses . down the weeks until the startup. Physicists The shut down period was taken advantage followed the progress in tuning up the of by upgrading some setups, such as machine and heard with dismay about the ISOLTRAP, and building new ones. Firstly stumbling blocks encountered, mainly the ISOLDE beta Decay Studies, IDS, problems with the controls, and at the same permanently equipped with three clover time, they greeted the advances with HPGe detectors and a Miniball detector was excitement as they were eager to start. installed. Depending on the physics case In parallel the construction of building 508 IDS can incorporate Si detectors for beta- continued. The new building has more delayed charged particle studies, or space for the physicists, their laboratories LaBr3(Ce) scintillators for half-life and DAQ rooms and hosts the ISOLDE measurements of excited states. Secondly control room in a spacious and well the previous UHV ASPIC beam line was fully ventilated room outside the controlled area, redesigned to host up to three possible a long standing request from the Beams experiments including ASPIC. The first part Department. The large control room is of the new VITO (Versatile Ion-polarized ready to accomodate the future TSR Technique on-line) line was mounted and controls. -

Accepted Paper Cite As
Accepted paper Cite as: D. Nd. Faye, X. Biquard, E. Nogales, M. Felizardo, M. Peres, A. Redondo-Cubero, T. Auzelle, B. Daudin, L.H.G. Tizei, M. Kociak, P. Ruterana, W. Möller, B. Méndez, E. Alves, K. Lorenz Incorporation of Europium into GaN Nanowires by Ion Implantation J. Phys. Chem. C 123 (2019) 11874−11887 DOI: 10.1021/acs.jpcc.8b12014 Incorporation of Europium into GaN Nanowires by Ion Implantation D. Nd. Faye1, X. Biquard2, E. Nogales3, M. Felizardo1, M. Peres1, A. Redondo- Cubero1+, T. Auzelle4,5, B. Daudin4, L.H.G. Tizei5, M. Kociak5, P. Ruterana6, W. Möller7, B. Méndez3, E. Alves1, K. Lorenz1,8 1IPFN, Instituto Superior Técnico, Universidade de Lisboa, Campus Tecnológico e Nuclear, Estrada Nacional 10, 2695-066 Bobadela LRS, Portugal 2Université Grenoble Alpes, CEA, IRIG, MEM, NRS, 38000 Grenoble, France 3Departamento de Física de Materiales, Universidad Complutense, 28040 Madrid, Spain 4Université Grenoble Alpes, CEA, IRIG, PHELIQS, 38000 Grenoble, France 5Laboratoire de Physique des Solides, Université Paris-Sud, CNRS-UMR 8502, Orsay 91405, France 6Centre de recherche sur les Ions, les Matériaux et la Photonique (CIMAP), UMR 6252, CNRS-ENSICAEN, 6, Boulevard Maréchal Juin, 14050 Caen, France 7Helmholtz-Zentrum Dresden-Rossendorf, Institute of Ion Beam Physics and Materials Research, Bautzner Landstraße 400, D-01328 Dresden, Germany 8Instituto de Engenharia de Sistemas de Computadores- Microsistemas e Nanotecnologias (INESC-MN), Rua Alves Redol 9, 1000-029 Lisboa, Portugal + Current address: Departamento de Física Aplicada y Centro de Micro-Análisis de Materiales, Universidad Autónoma de Madrid, Madrid, Spain Tel. +351-219946052; fax +351 21 9946285; [email protected] 1 ABSTRACT Rare earth (RE) doped GaN nanowires (NWs), combining the well-defined and controllable optical emission lines of trivalent RE ions with the high crystalline quality, versatility and small dimension of the NW host, are promising building blocks for future nanoscale devices in optoelectronics and quantum technologies. -

3.1 Ion Implantation
CHAPTER 3 SAMPLE PREPARATION AND CHARACTERIZATION 35 3.1 Ion implantation This work’s implanted ions were produced in an ion source by sputtering a solid target or by ionising a vapour. They are extracted from the source and accelerated by an electrostatic field to energies of a few thousand electron volt (keV). Subsequently, the ions are separated by mass, using an analyzing magnet. After passing through electrostatic or magnetic quadrupole for focusing, the ions become implanted in the near-surface region of the desired material. In SrTiO3 typical implanted layers have depths of about 200-300 Å if the energy of the ion beam ranges in between 40-60 keV. The ions come to rest by dissipating the kinetic energy through interactions (i.e, collisions) with nuclei and electrons in the solid. The cross sections for both scattering processes depend on the incident ion energy, its nuclear charge and mass and the mass of the solid atoms. The second process, called electronic stopping, is dominant if high ion energies and small atomic numbers Z are present. With decreasing kinetic energy of the ions or increasing atomic number Z, the first process, called nuclear stopping (at the Coulomb potential of the nuclei) becomes dominant. Nuclear stopping leads to the generation of defects in the solid crystalline lattice, if the energy transferred to the target atom is sufficient for a displacement. Both electronic stopping and nuclear stopping determine the implantation profile, i.e. the depth distribution of the implanted ions. The implantation depth and corresponding straggling (equal to the variance of the distribution) can be calculated with SRIM (the Stopping Power and Range of Ions in Matter) code, which is a Monte Carlo computer simulation program [1]. -

Notes on Channeling
NOTES ON CHANNELING J.U. Andersen LIST OF CONTENTS AND FIGURES Chapters Page Figures 1. Introduction 2 2. Directional effects 3 1-3 3. Deflection by atom 6 4 4. Continuum potential 8 5 5. Atomic potential 10 6 6. Critical angles 13 7, 8 7. Thermal vibrations 18 9 8. Merging of axial and planar effects 21 10-12 9. Reversibility and blocking 24 10. Experimental observation 26 19-21a 11. Statistical treatment 33 22 12. Trend towards equilibrium 37 23, 24 13. Three stages in particle motion 40 25-29 14. Standard model 45 30-34 15. Corrections to 휒min and 휓1/2 53 35-41 16. Compensation rules 60 42, 43 17. Scattering of random and aligned beam 66 44-46 18. Dechanneling 82 47-56 19. Stopping and atomic processes in channelled ions 102 57-66 20. Applications of channeling and blocking 112 67-72 21. Electrons and positrons, channeling radiation 118 73-87 22. References 130 1. INTRODUCTION Ion channeling in crystals was discovered in the early 1960s [Davies 1983]. Systematic studies of the range of low-energy heavy ions revealed significant discrepancies from theoretical expectations. The average range was found to be in good agreement with theory, but there was in polycrystalline materials a nearly exponential tail of ions with much longer range. Experiments with amorphous and single-crystalline materials soon proved that the effect was caused by enhanced penetration along major axes in the crystals [Piercy et al. 1963], [Lutz and Sizmann 1963]. Also computer simulations of ion penetration showed this effect [Robinson and Oen 1963]. -

Thermal Stability of Te-Hyperdoped Si: Atomic-Scale Correlation of the Structural, Electrical and Optical Properties
Thermal stability of Te-hyperdoped Si: Atomic-scale correlation of the structural, electrical and optical properties Mao Wang1,2,*, R. Hübner,1 Chi Xu1,2, Yufang Xie1,2, Y. Berencén1, R. Heller1, L. Rebohle1, M. Helm1,2, S. Prucnal1, and Shengqiang Zhou1 1Helmholtz-Zentrum Dresden-Rossendorf, Institute of Ion Beam Physics and Materials Research, Bautzner Landstr. 400, 01328 Dresden, Germany 2Technische Universität Dresden, 01062 Dresden, Germany Abstract Si hyperdoped with chalcogens (S, Se, Te) is well-known to possess unique properties such as an insulator-to-metal transition and a room-temperature sub-bandgap absorption. These properties are expected to be sensitive to a post-synthesis thermal annealing, since hyperdoped Si is a thermodynamically metastable material. Thermal stability of the as-fabricated hyperdoped Si is of great importance for the device fabrication process involving temperature-dependent steps like ohmic contact formation. Here, we report on the thermal stability of the as-fabricated Te- hyperdoped Si subjected to isochronal furnace anneals from 250 °C to 1200 °C. We demonstrate that Te-hyperdoped Si exhibits thermal stability up to 400 °C with a duration of 10 minutes that even helps to further improve the crystalline quality, the electrical activation of Te dopants and the room-temperature sub-band gap absorption. At higher temperatures, however, Te atoms are found to move out from the substitutional sites with a migration energy of EM = 2.1 ± 0.1 eV forming inactive clusters and precipitates that impair the structural, electrical and optical properties. These results provide further insight into the underlying physical state transformation of Te dopants in a metastable compositional regime caused by post-synthesis thermal annealing as well as pave the way for the fabrication of advanced hyperdoped Si-based devices.