2018 Annual Report (PDF)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Entertainment & Syndication Fitch Group Hearst Health Hearst Television Magazines Newspapers Ventures Real Estate & O
hearst properties WPBF-TV, West Palm Beach, FL SPAIN Friendswood Journal (TX) WYFF-TV, Greenville/Spartanburg, SC Hardin County News (TX) entertainment Hearst España, S.L. KOCO-TV, Oklahoma City, OK Herald Review (MI) & syndication WVTM-TV, Birmingham, AL Humble Observer (TX) WGAL-TV, Lancaster/Harrisburg, PA SWITZERLAND Jasper Newsboy (TX) CABLE TELEVISION NETWORKS & SERVICES KOAT-TV, Albuquerque, NM Hearst Digital SA Kingwood Observer (TX) WXII-TV, Greensboro/High Point/ La Voz de Houston (TX) A+E Networks Winston-Salem, NC TAIWAN Lake Houston Observer (TX) (including A&E, HISTORY, Lifetime, LMN WCWG-TV, Greensboro/High Point/ Local First (NY) & FYI—50% owned by Hearst) Winston-Salem, NC Hearst Magazines Taiwan Local Values (NY) Canal Cosmopolitan Iberia, S.L. WLKY-TV, Louisville, KY Magnolia Potpourri (TX) Cosmopolitan Television WDSU-TV, New Orleans, LA UNITED KINGDOM Memorial Examiner (TX) Canada Company KCCI-TV, Des Moines, IA Handbag.com Limited Milford-Orange Bulletin (CT) (46% owned by Hearst) KETV, Omaha, NE Muleshoe Journal (TX) ESPN, Inc. Hearst UK Limited WMTW-TV, Portland/Auburn, ME The National Magazine Company Limited New Canaan Advertiser (CT) (20% owned by Hearst) WPXT-TV, Portland/Auburn, ME New Canaan News (CT) VICE Media WJCL-TV, Savannah, GA News Advocate (TX) HEARST MAGAZINES UK (A+E Networks is a 17.8% investor in VICE) WAPT-TV, Jackson, MS Northeast Herald (TX) VICELAND WPTZ-TV, Burlington, VT/Plattsburgh, NY Best Pasadena Citizen (TX) (A+E Networks is a 50.1% investor in VICELAND) WNNE-TV, Burlington, VT/Plattsburgh, -

COMSEARCH 19700 Janelia Farm Boulevard Ashburn, VA 20147
COMSEARCH 19700 Janelia Farm Boulevard Ashburn, VA 20147 www.comsearch.com Supplemental Showing Pursuant To: 74.638 Frequency Coordination Reference: POST NEWSWEEK STATIONS ORLANDO INC Job Number: 040525COMSKD03 JYP TOWER - OC TOWER, LEESBURG CBL - COX CABLE, FL 2.0, 7.0 GHz VIDEO MICROWAVE SYSTEM Polarization & centerline changes *** Expedited Response Requested By June 16, 2004 *** The results fo the study conducted on May 25, 2004 indicated the proposed frequencies for the above referenced microwave system do not cause interference conflicts with any existing or applied-for facilities in the area. The following are the licensees of the stations considered in the analysis. Licensees ACME TELEVISION LICENSES OF FLORIDA LLC ASSOCIATED CHRISTIAN TV SYSTEM, INC BAY TELEVISION, INC. BEACH TELEVISION PARTNERS BOARD OF REGENTS UNIVERSITY OF FLORIDA BOARD OF TRUSTEES, UNIVERSITY OF FLORIDA BREVARD COMMUNITY COLLEGE BROADCASTING TELECASTING SERVICES INC Bright House Networks, LLC C-34 FCC LICENSEE SUSIDIARY, L.L.C. CHRISTIAN TELEVISION CORPORATION CLEAR CHANNEL BROADCASTING LICENSES INC COMMUNITY COMMUNICATIONS (WMFE) COMMUNITY TV FOUNDATION OF SOUTH FLORIDA COSMOS BROADCASTING CORP. (WAVE-TV) DAYTONA BEACH COMMUNITY COLLEGE DISNEY REGIONAL ENTERTAINMENT Diversified Broadcasting Inc. Diversified Broadcasting, Inc. EMMIS TELEVISION LICENSE CORPORATION ENTRAVISION HOLDINGS, LLC FLORIDA GULF COAST UNIVERSITY FLORIDA MEDIA BROADCASTERS INC FLORIDA WEST COAST PUBLIC BROADCASTING I FORT MYERS BROADCASTING CO FOX TELEVISION STATIONS INC FREEDOM BROADCASTING -

PAIGE HARRISON Vice President, News, Hearst Television
PAIGE HARRISON Vice President, News, Hearst Television Paige Harrison is vice president, news at Hearst Television. She was appointed to this position in June 2021. Previously, Harrison served as the news director of Hearst Television’s flagship station and perennial TV news leader WCVB-TV, Boston since 2017. While there, she managed all operations of the news department, which produces more than 45 hours of newscasts weekly, including breaking news, weather and sports, special coverage, the station’s 5 Investigates series, NewsCenter 5 Primetime specials and all of the station’s digital and social media news initiatives. In her four years leading the unit, WCVB-TV’s news department earned dozens of awards for journalism and community service, including regional Emmy Awards, regional Edward R. Murrow Awards, National Headliner Awards, a Gracie Award and a Gabriel Award — frequently earning “Station of the Year” honors and often sweeping the regional competition. In Harrison’s 18 years with Hearst, she has served within news operations for five Hearst Television stations across the country. Before joining WCVB-TV, she was the news director at WPBF-TV, Hearst Television’s ABC affiliate serving the West Palm Beach, Florida, TV market. She also worked in news management positions at WESH-TV in Orlando, Florida, KOCO-TV in Oklahoma City, Oklahoma, and WTAE-TV in Pittsburgh, Pennsylvania. A Michigan native, Harrison was raised in Halifax, Nova Scotia, in Canada and began her television career there as a producer and director. She has also worked in Toronto, Ottawa, and Barrie, Ontario. . -

Hearst Television Inc
HEARST TELEVISION INC 175 198 194 195 203 170 197 128 201 DR. OZ 3RD QUEEN QUEEN MIND OF A SEINFELD 4TH SEINFELD 5TH DR. OZ CYCLE LATIFAH LATIFAH MAN CYCLE CYCLE KING 2nd Cycle KING 3rd Cycle RANK MARKET %US STATION 2011-2014 2014-2015 2013-2014 2014-2015 2015-2016 4th Cycle 5th Cycle 2nd Cycle 3rd Cycle 7 BOSTON (MANCHESTER) MA 2.13% WCVB/WMUR WFXT WFXT WBZ/WSBK WBZ/WSBK WBZ/WSBK WBZ/WSBK WBZ/WSBK WBZ/WSBK 13 TAMPA-ST PETERSBURG (SARASOTA) FL 1.60% WMOR WFLA WFTS WTOG WTOG WTTA WTTA WTOG WTOG 18 ORLANDO-DAYTONA BEACH-MELBOURNE FL 1.29% WESH/WKCF WFTV/WRDQ WOFL/WRBW WKMG WKMG WESH/WKCF WFTV/WRDQ WFTV/WRDQ WFTV/WRDQ 20 SACRAMENTO-STOCKTON-MODESTO CA 1.18% KCRA/KCRA-DT2/KQCA KCRA/KQCA KCRA/KQCA KMAX/KOVR KMAX/KOVR KTXL KTXL KMAX/KOVR 22 PITTSBURGH PA 1.03% WTAE WTAE WTAE KDKA/WPCW KDKA/WPCW WPGH/WPMY WPGH/WPMY KDKA/WPCW KDKA/WPCW 26 BALTIMORE MD 0.96% WBAL/WBAL-DT2 WBAL WBAL WBFF/WNUV/WUTB WBFF/WNUV/WUTB WBFF/WNUV/WUTB WBFF/WNUV WBFF/WNUV 31 KANSAS CITY MO 0.81% KCWE/KMBC KCWE/KMBC KCWE/KMBC WDAF WDAF WDAF WDAF KMCI/KSHB KMCI 35 MILWAUKEE WI 0.79% WISN WISN WISN WDJT/WMLW WDJT/WMLW WITI WITI WCGV/WVTV 36 CINCINNATI OH 0.77% WLWT WLWT WLWT WLWT WKRC/WSTR EKRC/WKRC EKRC/WKRC/WSTR WXIX WXIX 37 GREENVILLE-SPARTANBURG (SC)-ASHEVILLE 0.74% WYFF WYFF WYFF WLOS/WMYA WLOS/WMYA WSPA/WYCW WSPA/WYCW WYCW WSPA/WYCW 38 WEST PALM BEACH-FT PIERCE FL 0.69% WPBF WPBF WPBF WPTV WPTV WFLX WFLX WTCN/WTVX 43 BIRMINGHAM (ANNISTON-TUSCALOOSA) AL 0.62% WVTM WBMA WBMA WBRC WBRC WABM/WTTO WABM/WTTO WABM/WTTO 44 OKLAHOMA CITY OK 0.62% KOCO KOCO KOCO KOCB/KOKH KOCB/KOKH -

HEARST PROPERTIES HUNGARY HEARST MAGAZINES UK Hearst Central Kft
HEARST PROPERTIES HUNGARY HEARST MAGAZINES UK Hearst Central Kft. (50% owned by Hearst) All About Soap ITALY Best Cosmopolitan NEWSPAPERS MAGAZINES Hearst Magazines Italia S.p.A. Country Living Albany Times Union (NY) H.M.C. Italia S.r.l. (49% owned by Hearst) Car and Driver ELLE Beaumont Enterprise (TX) Cosmopolitan JAPAN ELLE Decoration Connecticut Post (CT) Country Living Hearst Fujingaho Co., Ltd. Esquire Edwardsville Intelligencer (IL) Dr. Oz THE GOOD LIFE Greenwich Time (CT) KOREA Good Housekeeping ELLE Houston Chronicle (TX) Hearst JoongAng Y.H. (49.9% owned by Hearst) Harper’s BAZAAR ELLE DECOR House Beautiful Huron Daily Tribune (MI) MEXICO Laredo Morning Times (TX) Esquire Inside Soap Hearst Expansion S. de R.L. de C.V. Midland Daily News (MI) Food Network Magazine Men’s Health (50.1% owned by Hearst UK) (51% owned by Hearst) Midland Reporter-Telegram (TX) Good Housekeeping Prima Plainview Daily Herald (TX) Harper’s BAZAAR NETHERLANDS Real People San Antonio Express-News (TX) HGTV Magazine Hearst Magazines Netherlands B.V. Red San Francisco Chronicle (CA) House Beautiful Reveal The Advocate, Stamford (CT) NIGERIA Marie Claire Runner’s World (50.1% owned by Hearst UK) The News-Times, Danbury (CT) HMI Africa, LLC O, The Oprah Magazine Town & Country WEBSITES Popular Mechanics NORWAY Triathlete’s World Seattlepi.com Redbook HMI Digital, LLC (50.1% owned by Hearst UK) Road & Track POLAND Women’s Health WEEKLY NEWSPAPERS Seventeen Advertiser North (NY) Hearst-Marquard Publishing Sp.z.o.o. (50.1% owned by Hearst UK) Town & Country Advertiser South (NY) (50% owned by Hearst) VERANDA MAGAZINE DISTRIBUTION Ballston Spa/Malta Pennysaver (NY) Woman’s Day RUSSIA Condé Nast and National Magazine Canyon News (TX) OOO “Fashion Press” (50% owned by Hearst) Distributors Ltd. -
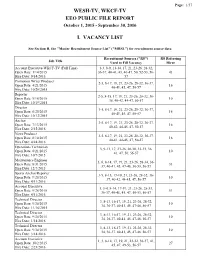
Wesh-Tv, Wkcf-Tv Eeo Public File Report I. Vacancy List
Page: 1/57 WESH-TV, WKCF-TV EEO PUBLIC FILE REPORT October 1, 2015 - September 30, 2016 I. VACANCY LIST See Section II, the "Master Recruitment Source List" ("MRSL") for recruitment source data Recruitment Sources ("RS") RS Referring Job Title Used to Fill Vacancy Hiree Account Executive-WKCF-TV (Full Time) 1-3, 8-9, 11-14, 17, 21, 23-26, 28-32, Open Date: 1/14/2015 36-37, 40-41, 43, 46-47, 50, 52-53, 56- 43 Hire Date: 9/14/2016 57 Promotion Writer Producer 2-5, 8-17, 19, 21, 23-26, 28-32, 36-37, Open Date: 4/21/2015 16 40-41, 45, 47, 50-57 Hire Date: 10/26/2015 Reporter 2-5, 8-15, 17, 19, 21, 23-26, 28-32, 36- Open Date: 5/18/2015 10 38, 40-42, 44-47, 50-57 Hire Date: 10/19/2015 Director 3-5, 8-17, 19, 21, 23-26, 28-32, 36-37, Open Date: 6/25/2015 16 40-41, 45, 47, 50-57 Hire Date: 10/12/2015 Anchor 3-5, 8-17, 19, 21, 23-26, 28-32, 36-37, Open Date: 7/13/2015 16 40-42, 44-45, 47, 50-57 Hire Date: 2/15/2016 News Producer 3-5, 8-17, 19, 21, 23-26, 28-32, 36-37, Open Date: 8/10/2015 16 40-41, 44-45, 47, 50-57 Hire Date: 4/24/2016 Operations Technician 3, 9-13, 17, 23-26, 28-30, 32-33, 36, Open Date: 8/21/2015 10 41, 47, 50, 56-57 Hire Date: 10/7/2015 Maintenance Engineer 3, 6, 8-14, 17, 19, 21, 23-26, 28-34, 36- Open Date: 8/31/2015 33 37, 40-41, 45, 47-48, 50-53, 56-57 Hire Date: 12/7/2015 Sports Anchor/Reporter 3-5, 8-15, 17-19, 21, 23-26, 28-32, 36- Open Date: 9/28/2015 10 37, 40-42, 44-45, 47, 50-57 Hire Date: 4/11/2016 Account Executive 1, 3-4, 8-14, 17-19, 21, 23-26, 28-33, Open Date: 9/28/2015 51 36-37, 40-41, 45, 47, 50-53, 56-57 -

Kcci, Ketv, Khbs/Khog, Kmbc, Koat, Koco
KCCI, KETV, KHBS/KHOG, KMBC, KOAT, KOCO, KSBW, WAPT, WBAL, WDSU, WESH, WJCL, WLKY, WLWT, WMTW, WMUR, WNNE/WPTZ, WPBF, WTAE, WVTM, WYFF Ellen 12 Days Sweepstakes Official Rules 1. GENERAL: No purchase necessary. Making a purchase will not increase your chances of winning. Void where prohibited or restricted by law. All federal, state, and local regulations apply. This Sweepstakes is in no way sponsored, administered by or associated with The Ellen DeGeneres Show, WAD Productions Inc., or its affiliated entities. 2. ELIGIBILITY: The Ellen 12 Days Sweepstakes (“Sweepstakes”) is offered only to legal U.S. residents 18 years old or older at the time of entry. One winner per household. One winner per family. Prizes are non-transferable. Sweepstakes is offered only to: Alabama, Arkansas, Maryland, Kentucky, New York, Vermont, Maine, Georgia, Louisiana, Oklahoma, South Carolina, New Hampshire, Nebraska, Florida, Pennsylvania, Iowa, Ohio, Missouri, Mississippi, New Mexico and Massachusetts residents who reside within the Birmingham, AL, Baltimore, MD, Ft. Smith/Fayetteville, AR, Louisville, KY, Plattsburgh, NY/Burlington, VT, Portland-Auburn, ME, Monterey-Salinas, CA, New Orleans, LA, Oklahoma City, OK, Greenville, SC, Omaha, NB, West Palm, FL, Pittsburgh, PA, Orlando, FL, Des Moines, IA, Cincinnati, OH, Kansas City, MO, Savannah, GA, Jackson, MS, Albuquerque, NM, Boston, MA designated Market Areas (“DMAs”) as determined by Nielsen Media Research Services. Winners of a contest or sweepstakes hosted or sponsored by KCCI, KETV, KHBS/KHOG, KMBC, KOAT, KOCO, KSBW, WAPT, WBAL, WDSU, WESH, WJCL, WLKY, WLWT, WMTW, WMUR, WNNE/WPTZ, WPBF, WTAE, WVTM, WYFF, or its affiliates within the past calendar year are not eligible. -
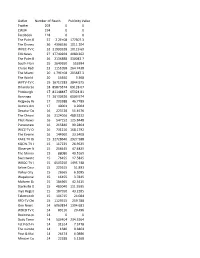
Number of Articles and Outlets.Pdf
Outlet Number of ClipsReach Publicity Value Twitter 203 0 0 CW34 194 0 0 Facebook 118 0 0 The Palm Beach Post57 Online2.2E+08 177307.3 The Disney Cruise Line36 Blog4396536 1011.204 WPEC-TV Online 32 21999328 10119.69 EIN News 27 17740269 4080.262 The Palm Beach Post26 2136888 330043.7 South Florida Sun Sentinel25 2649650 553994 Cruise Radio 23 1151058 264.7438 The Miami Herald Online20 1.79E+08 205687.3 The World News 20 14640 3.368 WPTV-TV Online 19 16717283 3844.975 Orlando Sentinel Online18 85873374 69128.07 Pittsburgh Post-Gazette17 81148837 Online 65324.81 Benzinga 17 26130326 6009.974 Ridgway Record 17 203388 46.7789 Antlers American Online17 40001 9.2004 Decatur Daily Democrat16 Online223728 51.4576 The Chronicle-Journal16 Online2124016 488.5232 Pilot-News Online 16 547152 125.8448 Punxsutawney Spirit16 215840 99.2864 WICZ-TV ONLINE 16 731216 168.1792 The Evening Leader 16Online144960 33.3408 KAKE TV Online 15 12728640 2927.588 KQCW-TV Online 15 117225 26.9625 Observer News Enterprise15 206445 - Online 47.4825 The Morning News 15 88080 40.5165 Sweetwater Reporter15 - Online76455 17.5845 WBOC-TV Online 15 6503250 1495.748 Saline Courier Online15 225615 51.891 Valley City Times-Record15 Online26565 6.1095 Wapakoneta Daily News15 Online16455 3.7845 Malvern Daily Record15 184965 42.5415 Starkville Daily News15 Online485040 111.5595 Inyo Register - Online15 187950 43.2285 Telemundo Lubbock15 104715 24.084 RFD-TV Online 15 1129515 259.788 One News Page 14 6063834 1394.681 WBCB TV Online 14 89110 20.496 Business.poteaudailynews14 0 0 Daily -

Hearst Television Station WPBF Completes Production Control Room Upgrade with Becktv
Agency Contact: BeckTV Contact: Susan Warren Rich Hajdu Wall Street Communications Vice President, Sales and Marketing Tel: +1 (631) 261-1013 Tel: +1 (330) 962-6502 Email: [email protected] Email: [email protected] Photo Link: www.wallstcom.com/BeckTV/BeckTV-WPBFControlRoom.jpg For Immediate Release Hearst Television Station WPBF Completes Production Control Room Upgrade With BeckTV BeckTV Helps West Palm Beach Station to Transition Smoothly and Painlessly to New Facilities and Equipment Without Disrupting Broadcast Operations AUSTIN, Texas — April 29, 2014 — BeckTV, one of the United States’ premier design-build systems integrators, today announced that it has completed an upgrade of the control room and technical core at Hearst Television station WPBF in West Palm Beach, Florida. BeckTV worked closely with WPBF engineers to ensure a smooth transition from old facilities and equipment to new future-proof facilities and systems without disruption of the station’s on-air broadcasts. “BeckTV was involved in every step of this project, from planning through implementation, to ensure that we could meet our new technical and operational goals,” said Cliff Thomas, chief engineer at WPBF. “With their help, we have built a new facility that will withstand the test of time, and we did so while staying on budget and on schedule.” Completed through a phased approach, the WPBF facility upgrade brought station operations into a new engineering wing with new control rooms and offices, a new technical core, and a more robust HVAC system. Together WPBF and BeckTV chose new rack systems and designed and built a new electrical distribution system to support the demands of today’s IT- heavy broadcast infrastructure. -

Ellen 12 Days 2019 Sweepstakes Official Rules 1. GENERAL
Ellen 12 Days 2019 Sweepstakes Official Rules 1. GENERAL: No purchase necessary. Making a purchase will not increase your chances of winning. Void where prohibited or restricted by law. All federal, state, and local regulations apply. This Sweepstakes is in no way sponsored, administered by or associated with The Ellen DeGeneres Show, WAD Productions Inc., or its affiliated entities. 2. ELIGIBILITY: The Ellen 12 Days 2019 Sweepstakes (“Sweepstakes”) is offered only to legal U.S. residents 18 years old or older at the time of entry (19 or older if residing within Alabama or Nebraska and 21 or older if residing in Mississippi). One winner per household. One winner per family. Prizes are non-transferable. Sweepstakes is offered only to residents of the following states who live within the Designated Market Areas as determined by Nielsen Media Research Services (“DMA”s) that follow: Alabama: Birmingham-Anniston-Tuscaloosa DMA, Arkansas & Oklahoma: Ft. Smith/Fayetteville DMA, California: Monterey-Salinas DMA, Florida: West Palm Beach/Ft. Pierce DMA, Florida: Orlando-Daytona Beach-Melbourne DMA, Georgia: Savannah DMA (SC counties excluded), Iowa: Des Moines DMA, Kansas & Missouri: Kansas City DMA, Kentucky & Indiana: Louisville DMA, Louisiana: Greater New Orleans DMA, Maine: Portland- Auburn DMA (NH counties excluded), Maryland: Baltimore DMA, Mississippi: Jackson DMA, Nebraska & Iowa: Omaha/Council Bluffs DMA, New Hampshire: Boston/Manchester DMA and any NH county (MA and VT counties excluded), New Mexico: Albuquerque/Santa Fe DMA, New York & Vermont: Plattsburgh/Burlington DMA (NH counties excluded), Ohio, Kentucky & Indiana: Cincinnati DMA, Oklahoma: Oklahoma City DMA, Pennsylvania, Ohio & West Virginia: Pittsburgh DMA (MD counties excluded), South Carolina & North Carolina: Greenville- Spartanburg-Asheville-Anderson DMA (GA counties excluded). -

Eeo Public File Report for Wpbf-Tv
EEO PUBLIC FILE REPORT FOR WPBF-TV This EEO Public File Report Covers the One-Year Period Ending on September 30, 2019 EEO Annual Public File Report WPBF-TV This EEO Public File Report is filed in Station WPBF-TV’s public inspection file pursuant to Section 73.2080(c)(6) of the Federal Communications Commission’s EEO Rule. The information contained in this Report covers the one year time period ending September 30, 2019 (the “Reporting Period”). Attachments 1 through 3 are intended to provide the information required by the FCC’s EEO Rule. Attachments 1 and 2 contain the following information for each full- time vacancy: * The full-time vacancies over the Reporting Period, the recruitment sources used to fill each vacancy, and the recruitment source that referred the hiree for each full-time vacancy; * The master list of recruitment sources utilized by the station, identified by name, address, contact person and telephone number; and * The total number of persons interviewed for each recruiting source over the Reporting Period. Attachment 3 contains a list and brief description of outreach initiatives undertaken pursuant to the FCC’s EEO Rule during the Reporting Period. Questions concerning this Report should be directed to Caroline Taplett, President and General Manager, WPBF-TV, 3970 RCA Blvd, Suite 7007, Palm Beach Gardens, FL 33410. * * * * * * Page: 1/16 WPBF-TV EEO PUBLIC FILE REPORT October 1, 2018 - September 30, 2019 I. VACANCY LIST See Section II, the "Master Recruitment Source List" ("MRSL") for recruitment source data Recruitment -

Wpbf 25 and the Florida Press Association Announce Panelists in "The Race for Governor" Democratic Primary Televised Debate
WPBF 25 AND THE FLORIDA PRESS ASSOCIATION ANNOUNCE PANELISTS IN "THE RACE FOR GOVERNOR" DEMOCRATIC PRIMARY TELEVISED DEBATE West Palm Beach, FL. July 19, 2018 -- The Florida Press Association and WPBF 25 News will present “The Race for Governor” -- The Democratic Primary Debate, live on Thursday, August 2nd from 7-8 p.m. WPBF 25, the Hearst Television-owned ABC affiliate in West Palm Beach, is the producing television station and will originate the one-hour debate from the WPBF 25 News studios for statewide broadcast. The debate will air in all 10 major Florida markets through a consortium of participating broadcast partners. Moderating the debate will be veteran WPBF 25 News anchor and multi-Emmy Award winner Todd McDermott. McDermott previously did national correspondent work for CBS in New York, as well as anchored at the CBS flagship station there. Prior to that, he anchored and reported at stations in Washington D.C., Baltimore and Wichita. McDermott also moderated the 2016 U.S. Senate Debate that was broadcast statewide, on C-Span and NPR. George Bennett, reporter, The Palm Beach Post, and Nancy Ancrum, Editorial Page Editor, the Miami Herald, will serve as media panelists. Bennett has been a reporter at Post since 1992 and has covered politics from city commission races to presidential campaigns. He has crisscrossed Florida to report on every governor’s race in this century and has appeared on CNN, Fox News and other outlets. Ancrum is a New Yorker who has finally come around. She began her career at the now-closed Baltimore Evening Sun and joined the The Miami Herald in 1983.