OEB51 Lecture 7 Introduc\On to the Protostomes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Systema Naturae∗
Systema Naturae∗ c Alexey B. Shipunov v. 5.802 (June 29, 2008) 7 Regnum Monera [ Bacillus ] /Bacteria Subregnum Bacteria [ 6:8Bacillus ]1 Superphylum Posibacteria [ 6:2Bacillus ] stat.m. Phylum 1. Firmicutes [ 6Bacillus ]2 Classis 1(1). Thermotogae [ 5Thermotoga ] i.s. 2(2). Mollicutes [ 5Mycoplasma ] 3(3). Clostridia [ 5Clostridium ]3 4(4). Bacilli [ 5Bacillus ] 5(5). Symbiobacteres [ 5Symbiobacterium ] Phylum 2. Actinobacteria [ 6Actynomyces ] Classis 1(6). Actinobacteres [ 5Actinomyces ] Phylum 3. Hadobacteria [ 6Deinococcus ] sed.m. Classis 1(7). Hadobacteres [ 5Deinococcus ]4 Superphylum Negibacteria [ 6:2Rhodospirillum ] stat.m. Phylum 4. Chlorobacteria [ 6Chloroflexus ]5 Classis 1(8). Ktedonobacteres [ 5Ktedonobacter ] sed.m. 2(9). Thermomicrobia [ 5Thermomicrobium ] 3(10). Chloroflexi [ 5Chloroflexus ] ∗Only recent taxa. Viruses are not included. Abbreviations and signs: sed.m. (sedis mutabilis); stat.m. (status mutabilis): s., aut i. (superior, aut interior); i.s. (incertae sedis); sed.p. (sedis possibilis); s.str. (sensu stricto); s.l. (sensu lato); incl. (inclusum); excl. (exclusum); \quotes" for environmental groups; * (asterisk) for paraphyletic taxa; / (slash) at margins for major clades (\domains"). 1Incl. \Nanobacteria" i.s. et dubitativa, \OP11 group" i.s. 2Incl. \TM7" i.s., \OP9", \OP10". 3Incl. Dictyoglomi sed.m., Fusobacteria, Thermolithobacteria. 4= Deinococcus{Thermus. 5Incl. Thermobaculum i.s. 1 4(11). Dehalococcoidetes [ 5Dehalococcoides ] 5(12). Anaerolineae [ 5Anaerolinea ]6 Phylum 5. Cyanobacteria [ 6Nostoc ] Classis 1(13). Gloeobacteres [ 5Gloeobacter ] 2(14). Chroobacteres [ 5Chroococcus ]7 3(15). Hormogoneae [ 5Nostoc ] Phylum 6. Bacteroidobacteria [ 6Bacteroides ]8 Classis 1(16). Fibrobacteres [ 5Fibrobacter ] 2(17). Chlorobi [ 5Chlorobium ] 3(18). Salinibacteres [ 5Salinibacter ] 4(19). Bacteroidetes [ 5Bacteroides ]9 Phylum 7. Spirobacteria [ 6Spirochaeta ] Classis 1(20). Spirochaetes [ 5Spirochaeta ] s.l.10 Phylum 8. Planctobacteria [ 6Planctomyces ]11 Classis 1(21). -

Identical Genomic Organization of Two Hemichordate Hox Clusters
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Current Biology 22, 2053–2058, November 6, 2012 ª2012 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2012.08.052 Report Identical Genomic Organization of Two Hemichordate Hox Clusters Robert Freeman,1,12 Tetsuro Ikuta,2,12 Michael Wu,3 [10] have similar organization to the hemichordate cluster, Ryo Koyanagi,2 Takeshi Kawashima,2 Kunifumi Tagawa,4 but with different posterior genes. These results provide Tom Humphreys,5 Guang-Chen Fang,6 Asao Fujiyama,7 genomic evidence for a well-ordered complex in the deutero- Hidetoshi Saiga,8 Christopher Lowe,9 Kim Worley,10 stome ancestor for the hox1–hox9/10 region, with the Jerry Jenkins,11 Jeremy Schmutz,11 Marc Kirschner,1 number and kind of posterior genes still to be elucidated. Daniel Rokhsar,3 Nori Satoh,2,* and John Gerhart3,* 1 Department of Systems Biology, Harvard Medical School, Results and Discussion Boston, MA 02114, USA 2 Marine Genomics Unit, Okinawa Institute of Science and Here we characterize the order, transcriptional orientation, and Technology Graduate University, Onna, clustering of the Hox genes of the genomes of two widely Okinawa 904-0495, Japan studied model hemichordates, Saccoglossus kowalevskii 3 Department of Molecular and Cell Biology, University of and Ptychodera flava [4, 11, 12], that represent two major California, Berkeley, Berkeley, CA 94720-3200, USA evolutionary branches of enteropneust hemichordates that 4 Marine Biological Laboratory, Graduate School of Science, diverged an estimated 400 million years ago (MYa) [13]: Hiroshima University, Onomichi, Hiroshima 722-0073, Japan Saccoglossus, of the direct developing harimaniids, and 5 Department of Cell and Molecular Biology, University of Ptychodera, of the indirect developing ptychoderids [14]. -

Platyhelminthes, Nemertea, and "Aschelminthes" - A
BIOLOGICAL SCIENCE FUNDAMENTALS AND SYSTEMATICS – Vol. III - Platyhelminthes, Nemertea, and "Aschelminthes" - A. Schmidt-Rhaesa PLATYHELMINTHES, NEMERTEA, AND “ASCHELMINTHES” A. Schmidt-Rhaesa University of Bielefeld, Germany Keywords: Platyhelminthes, Nemertea, Gnathifera, Gnathostomulida, Micrognathozoa, Rotifera, Acanthocephala, Cycliophora, Nemathelminthes, Gastrotricha, Nematoda, Nematomorpha, Priapulida, Kinorhyncha, Loricifera Contents 1. Introduction 2. General Morphology 3. Platyhelminthes, the Flatworms 4. Nemertea (Nemertini), the Ribbon Worms 5. “Aschelminthes” 5.1. Gnathifera 5.1.1. Gnathostomulida 5.1.2. Micrognathozoa (Limnognathia maerski) 5.1.3. Rotifera 5.1.4. Acanthocephala 5.1.5. Cycliophora (Symbion pandora) 5.2. Nemathelminthes 5.2.1. Gastrotricha 5.2.2. Nematoda, the Roundworms 5.2.3. Nematomorpha, the Horsehair Worms 5.2.4. Priapulida 5.2.5. Kinorhyncha 5.2.6. Loricifera Acknowledgements Glossary Bibliography Biographical Sketch Summary UNESCO – EOLSS This chapter provides information on several basal bilaterian groups: flatworms, nemerteans, Gnathifera,SAMPLE and Nemathelminthes. CHAPTERS These include species-rich taxa such as Nematoda and Platyhelminthes, and as taxa with few or even only one species, such as Micrognathozoa (Limnognathia maerski) and Cycliophora (Symbion pandora). All Acanthocephala and subgroups of Platyhelminthes and Nematoda, are parasites that often exhibit complex life cycles. Most of the taxa described are marine, but some have also invaded freshwater or the terrestrial environment. “Aschelminthes” are not a natural group, instead, two taxa have been recognized that were earlier summarized under this name. Gnathifera include taxa with a conspicuous jaw apparatus such as Gnathostomulida, Micrognathozoa, and Rotifera. Although they do not possess a jaw apparatus, Acanthocephala also belong to Gnathifera due to their epidermal structure. ©Encyclopedia of Life Support Systems (EOLSS) BIOLOGICAL SCIENCE FUNDAMENTALS AND SYSTEMATICS – Vol. -

New Zealand's Genetic Diversity
1.13 NEW ZEALAND’S GENETIC DIVERSITY NEW ZEALAND’S GENETIC DIVERSITY Dennis P. Gordon National Institute of Water and Atmospheric Research, Private Bag 14901, Kilbirnie, Wellington 6022, New Zealand ABSTRACT: The known genetic diversity represented by the New Zealand biota is reviewed and summarised, largely based on a recently published New Zealand inventory of biodiversity. All kingdoms and eukaryote phyla are covered, updated to refl ect the latest phylogenetic view of Eukaryota. The total known biota comprises a nominal 57 406 species (c. 48 640 described). Subtraction of the 4889 naturalised-alien species gives a biota of 52 517 native species. A minimum (the status of a number of the unnamed species is uncertain) of 27 380 (52%) of these species are endemic (cf. 26% for Fungi, 38% for all marine species, 46% for marine Animalia, 68% for all Animalia, 78% for vascular plants and 91% for terrestrial Animalia). In passing, examples are given both of the roles of the major taxa in providing ecosystem services and of the use of genetic resources in the New Zealand economy. Key words: Animalia, Chromista, freshwater, Fungi, genetic diversity, marine, New Zealand, Prokaryota, Protozoa, terrestrial. INTRODUCTION Article 10b of the CBD calls for signatories to ‘Adopt The original brief for this chapter was to review New Zealand’s measures relating to the use of biological resources [i.e. genetic genetic resources. The OECD defi nition of genetic resources resources] to avoid or minimize adverse impacts on biological is ‘genetic material of plants, animals or micro-organisms of diversity [e.g. genetic diversity]’ (my parentheses). -

Constraints on the Timescale of Animal Evolutionary History
Palaeontologia Electronica palaeo-electronica.org Constraints on the timescale of animal evolutionary history Michael J. Benton, Philip C.J. Donoghue, Robert J. Asher, Matt Friedman, Thomas J. Near, and Jakob Vinther ABSTRACT Dating the tree of life is a core endeavor in evolutionary biology. Rates of evolution are fundamental to nearly every evolutionary model and process. Rates need dates. There is much debate on the most appropriate and reasonable ways in which to date the tree of life, and recent work has highlighted some confusions and complexities that can be avoided. Whether phylogenetic trees are dated after they have been estab- lished, or as part of the process of tree finding, practitioners need to know which cali- brations to use. We emphasize the importance of identifying crown (not stem) fossils, levels of confidence in their attribution to the crown, current chronostratigraphic preci- sion, the primacy of the host geological formation and asymmetric confidence intervals. Here we present calibrations for 88 key nodes across the phylogeny of animals, rang- ing from the root of Metazoa to the last common ancestor of Homo sapiens. Close attention to detail is constantly required: for example, the classic bird-mammal date (base of crown Amniota) has often been given as 310-315 Ma; the 2014 international time scale indicates a minimum age of 318 Ma. Michael J. Benton. School of Earth Sciences, University of Bristol, Bristol, BS8 1RJ, U.K. [email protected] Philip C.J. Donoghue. School of Earth Sciences, University of Bristol, Bristol, BS8 1RJ, U.K. [email protected] Robert J. -

Comparative Neuroanatomy of Mollusks and Nemerteans in the Context of Deep Metazoan Phylogeny
Comparative Neuroanatomy of Mollusks and Nemerteans in the Context of Deep Metazoan Phylogeny Von der Fakultät für Mathematik, Informatik und Naturwissenschaften der RWTH Aachen University zur Erlangung des akademischen Grades einer Doktorin der Naturwissenschaften genehmigte Dissertation vorgelegt von Diplom-Biologin Simone Faller aus Frankfurt am Main Berichter: Privatdozent Dr. Rudolf Loesel Universitätsprofessor Dr. Peter Bräunig Tag der mündlichen Prüfung: 09. März 2012 Diese Dissertation ist auf den Internetseiten der Hochschulbibliothek online verfügbar. Contents 1 General Introduction 1 Deep Metazoan Phylogeny 1 Neurophylogeny 2 Mollusca 5 Nemertea 6 Aim of the thesis 7 2 Neuroanatomy of Minor Mollusca 9 Introduction 9 Material and Methods 10 Results 12 Caudofoveata 12 Scutopus ventrolineatus 12 Falcidens crossotus 16 Solenogastres 16 Dorymenia sarsii 16 Polyplacophora 20 Lepidochitona cinerea 20 Acanthochitona crinita 20 Scaphopoda 22 Antalis entalis 22 Entalina quinquangularis 24 Discussion 25 Structure of the brain and nerve cords 25 Caudofoveata 25 Solenogastres 26 Polyplacophora 27 Scaphopoda 27 i CONTENTS Evolutionary considerations 28 Relationship among non-conchiferan molluscan taxa 28 Position of the Scaphopoda within Conchifera 29 Position of Mollusca within Protostomia 30 3 Neuroanatomy of Nemertea 33 Introduction 33 Material and Methods 34 Results 35 Brain 35 Cerebral organ 38 Nerve cords and peripheral nervous system 38 Discussion 38 Peripheral nervous system 40 Central nervous system 40 In search for the urbilaterian brain 42 4 General Discussion 45 Evolution of higher brain centers 46 Neuroanatomical glossary and data matrix – Essential steps toward a cladistic analysis of neuroanatomical data 49 5 Summary 53 6 Zusammenfassung 57 7 References 61 Danksagung 75 Lebenslauf 79 ii iii 1 General Introduction Deep Metazoan Phylogeny The concept of phylogeny follows directly from the theory of evolution as published by Charles Darwin in The origin of species (1859). -

Development of the Annelid Axochord: Insights Into Notochord Evolution Antonella Lauri Et Al
RESEARCH | REPORTS ORIGIN OF NOTOCHORD by double WMISH (Fig. 2, F to L). Although none of the genes were exclusively expressed in the annelid mesodermal midline, their combined Development of the annelid coexpression was unique to these cells (implying that mesodermal midline in annelids and chor- damesoderm in vertebrates are more similar to axochord: Insights into each other than to any other tissue). It is unlikely that the molecular similarity between annelid notochord evolution and vertebrate mesodermal midline is due to in- dependent co-option of a conserved gene cas- Antonella Lauri,1*† Thibaut Brunet,1* Mette Handberg-Thorsager,1,2‡ sette, because this would require either that this Antje H.L. Fischer,1§ Oleg Simakov,1 Patrick R. H. Steinmetz,1‖ Raju Tomer,1,2¶ cassette was active elsewhere in the body (which Philipp J. Keller,2 Detlev Arendt1,3# is not the case) or that multiple identical inde- pendent events of co-option occurred (which is The origin of chordates has been debated for more than a century, with one key issue being unparsimonious). As in vertebrates, the meso- the emergence of the notochord. In vertebrates, the notochord develops by convergence dermal midline resembles the neuroectodermal and extension of the chordamesoderm, a population of midline cells of unique molecular midline, which expresses foxD, foxA, netrin, slit, identity. We identify a population of mesodermal cells in a developing invertebrate, the marine and noggin (figs. S6 and S7) but not brachyury or annelid Platynereis dumerilii, that converges and extends toward the midline and expresses a twist. However, unlike in chicken (10), the an- notochord-specific combination of genes. -

University of Copenhagen, Zoological Museum, Review Universitetsparken 15, DK-2100 Copenhagen, Denmark CN, 0000-0001-6898-7655 Cite This Article: Nielsen C
Early animal evolution a morphologist's view Nielsen, Claus Published in: Royal Society Open Science DOI: 10.1098/rsos.190638 Publication date: 2019 Document version Publisher's PDF, also known as Version of record Document license: CC BY Citation for published version (APA): Nielsen, C. (2019). Early animal evolution: a morphologist's view. Royal Society Open Science, 6(7), 1-10. [190638]. https://doi.org/10.1098/rsos.190638 Download date: 30. sep.. 2021 Early animal evolution: a morphologist’s view royalsocietypublishing.org/journal/rsos Claus Nielsen The Natural History Museum of Denmark, University of Copenhagen, Zoological Museum, Review Universitetsparken 15, DK-2100 Copenhagen, Denmark CN, 0000-0001-6898-7655 Cite this article: Nielsen C. 2019 Early animal evolution: a morphologist’s view. R. Soc. open sci. Two hypotheses for the early radiation of the metazoans are vividly discussed in recent phylogenomic studies, the ‘Porifera- 6: 190638. first’ hypothesis, which places the poriferans as the sister group http://dx.doi.org/10.1098/rsos.190638 of all other metazoans, and the ‘Ctenophora-first’ hypothesis, which places the ctenophores as the sister group to all other metazoans. It has been suggested that an analysis of morphological characters (including specific molecules) could Received: 5 April 2019 throw additional light on the controversy, and this is the aim of Accepted: 4 July 2019 this paper. Both hypotheses imply independent evolution of nervous systems in Planulozoa and Ctenophora. The Porifera- first hypothesis implies no homoplasies or losses of major characters. The Ctenophora-first hypothesis shows no important synapomorphies of Porifera, Planulozoa and Placozoa. It implies Subject Category: either independent evolution, in Planulozoa and Ctenophora, of Biology (whole organism) a new digestive system with a gut with extracellular digestion, which enables feeding on larger organisms, or the subsequent Subject Areas: loss of this new gut in the Poriferans (and the re-evolution of the evolution collar complex). -

Defining Phyla: Evolutionary Pathways to Metazoan Body Plans
EVOLUTION & DEVELOPMENT 3:6, 432-442 (2001) Defining phyla: evolutionary pathways to metazoan body plans Allen G. Collins^ and James W. Valentine* Museum of Paleontology and Department of Integrative Biology, University of California, Berkeley, CA 94720, USA 'Author for correspondence (email: [email protected]) 'Present address: Section of Ecology, Befiavior, and Evolution, Division of Biology, University of California, San Diego, La Jolla, CA 92093-0116, USA SUMMARY Phyla are defined by two sets of criteria, one pothesis of Nielsen; the clonal hypothesis of Dewel; the set- morphological and the other historical. Molecular evidence aside cell hypothesis of Davidson et al.; and a benthic hy- permits the grouping of animals into clades and suggests that pothesis suggested by the fossil record. It is concluded that a some groups widely recognized as phyla are paraphyletic, benthic radiation of animals could have supplied the ances- while some may be polyphyletic; the phyletic status of crown tral lineages of all but a few phyla, is consistent with molecu- phyla is tabulated. Four recent evolutionary scenarios for the lar evidence, accords well with fossil evidence, and accounts origins of metazoan phyla and of supraphyletic clades are as- for some of the difficulties in phylogenetic analyses of phyla sessed in the light of a molecular phylogeny: the trochaea hy- based on morphological criteria. INTRODUCTION Molecules have provided an important operational ad- vance to addressing questions about the origins of animal Concepts of animal phyla have changed importantly from phyla. Molecular developmental and comparative genomic their origins in the six Linnaean classis and four Cuvieran evidence offer insights into the genetic bases of body plan embranchements. -

Giant Pacific Octopus (Enteroctopus Dofleini) Care Manual
Giant Pacific Octopus Insert Photo within this space (Enteroctopus dofleini) Care Manual CREATED BY AZA Aquatic Invertebrate Taxonomic Advisory Group IN ASSOCIATION WITH AZA Animal Welfare Committee Giant Pacific Octopus (Enteroctopus dofleini) Care Manual Giant Pacific Octopus (Enteroctopus dofleini) Care Manual Published by the Association of Zoos and Aquariums in association with the AZA Animal Welfare Committee Formal Citation: AZA Aquatic Invertebrate Taxon Advisory Group (AITAG) (2014). Giant Pacific Octopus (Enteroctopus dofleini) Care Manual. Association of Zoos and Aquariums, Silver Spring, MD. Original Completion Date: September 2014 Dedication: This work is dedicated to the memory of Roland C. Anderson, who passed away suddenly before its completion. No one person is more responsible for advancing and elevating the state of husbandry of this species, and we hope his lifelong body of work will inspire the next generation of aquarists towards the same ideals. Authors and Significant Contributors: Barrett L. Christie, The Dallas Zoo and Children’s Aquarium at Fair Park, AITAG Steering Committee Alan Peters, Smithsonian Institution, National Zoological Park, AITAG Steering Committee Gregory J. Barord, City University of New York, AITAG Advisor Mark J. Rehling, Cleveland Metroparks Zoo Roland C. Anderson, PhD Reviewers: Mike Brittsan, Columbus Zoo and Aquarium Paula Carlson, Dallas World Aquarium Marie Collins, Sea Life Aquarium Carlsbad David DeNardo, New York Aquarium Joshua Frey Sr., Downtown Aquarium Houston Jay Hemdal, Toledo -
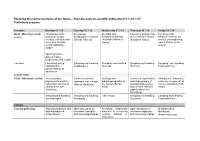
Exploring the Marine Meiofauna of the Azores – from Discovery to Scientific Publication (15.7.-24.7.19) Preliminary Program
Exploring the marine meiofauna of the Azores – from discovery to scientific publication (15.7.-24.7.19) Preliminary program: Schedule Monday 15.7.19 Tuesday 16.7.19 Wednesday 17.7.19 Thursday 18.7.19 Friday 19.7.19 09.00 - Morning session Introduction to the Meiofaunal Intertidal and Taxonomy and diversity The Proseriata Lecture workshop (people, Scalidophora (Andreas meiofaunal annelids of meiofaunal molluscs (Platyhelminthes): the facilities, schedule and Schmidt-Rhaesa) (Alejandro Martínez (Katharina Jörger) interest of unappealing aims; Ana Ricardo Garcia) worms (Marco Curini- Costa/ Katharina Galetti) Jörger) Opening lecture: Azores marine biodiversity (Ana Costa) Lab work Laboratory set-up, Sampling and handling Sampling and handling Sampling and handling Sampling and handling handling and Scalidophora Annelida Mollusca Platyhelminthes documentation of specimens LUNCH TIME 14.00 - Afternoon session Acoelomorpha – Marine nematodes: Geology and Taxonomy, systematics Rhabdocoel flatworms, phylogenetic position, taxonomy and ecology paleobiogeography of and biogeography of a diverse component of systematic and how to (Alberto Navarrete) the Azores (Sérgio meiofaunal Nemertea marine meiofauna (Tom identify them (Ulf Avila) and relevant research Artois) Jondelius) opportunities (Jon Norenburg) Sampling and handling Sampling and handling Lab session Sampling and handling Sampling and handling Acoelomorpha Nematoda Nemertea Platyhelminthes DINNER Evening gathering Sampling strategies and Open discussion on Adressing biodiversity Open discussion -

Hemichordate Phylogeny: a Molecular, and Genomic Approach By
Hemichordate Phylogeny: A molecular, and genomic approach by Johanna Taylor Cannon A dissertation submitted to the Graduate Faculty of Auburn University in partial fulfillment of the requirements for the Degree of Doctor of Philosophy Auburn, Alabama May 4, 2014 Keywords: phylogeny, evolution, Hemichordata, bioinformatics, invertebrates Copyright 2014 by Johanna Taylor Cannon Approved by Kenneth M. Halanych, Chair, Professor of Biological Sciences Jason Bond, Associate Professor of Biological Sciences Leslie Goertzen, Associate Professor of Biological Sciences Scott Santos, Associate Professor of Biological Sciences Abstract The phylogenetic relationships within Hemichordata are significant for understanding the evolution of the deuterostomes. Hemichordates possess several important morphological structures in common with chordates, and they have been fixtures in hypotheses on chordate origins for over 100 years. However, current evidence points to a sister relationship between echinoderms and hemichordates, indicating that these chordate-like features were likely present in the last common ancestor of these groups. Therefore, Hemichordata should be highly informative for studying deuterostome character evolution. Despite their importance for understanding the evolution of chordate-like morphological and developmental features, relationships within hemichordates have been poorly studied. At present, Hemichordata is divided into two classes, the solitary, free-living enteropneust worms, and the colonial, tube- dwelling Pterobranchia. The objective of this dissertation is to elucidate the evolutionary relationships of Hemichordata using multiple datasets. Chapter 1 provides an introduction to Hemichordata and outlines the objectives for the dissertation research. Chapter 2 presents a molecular phylogeny of hemichordates based on nuclear ribosomal 18S rDNA and two mitochondrial genes. In this chapter, we suggest that deep-sea family Saxipendiidae is nested within Harrimaniidae, and Torquaratoridae is affiliated with Ptychoderidae.