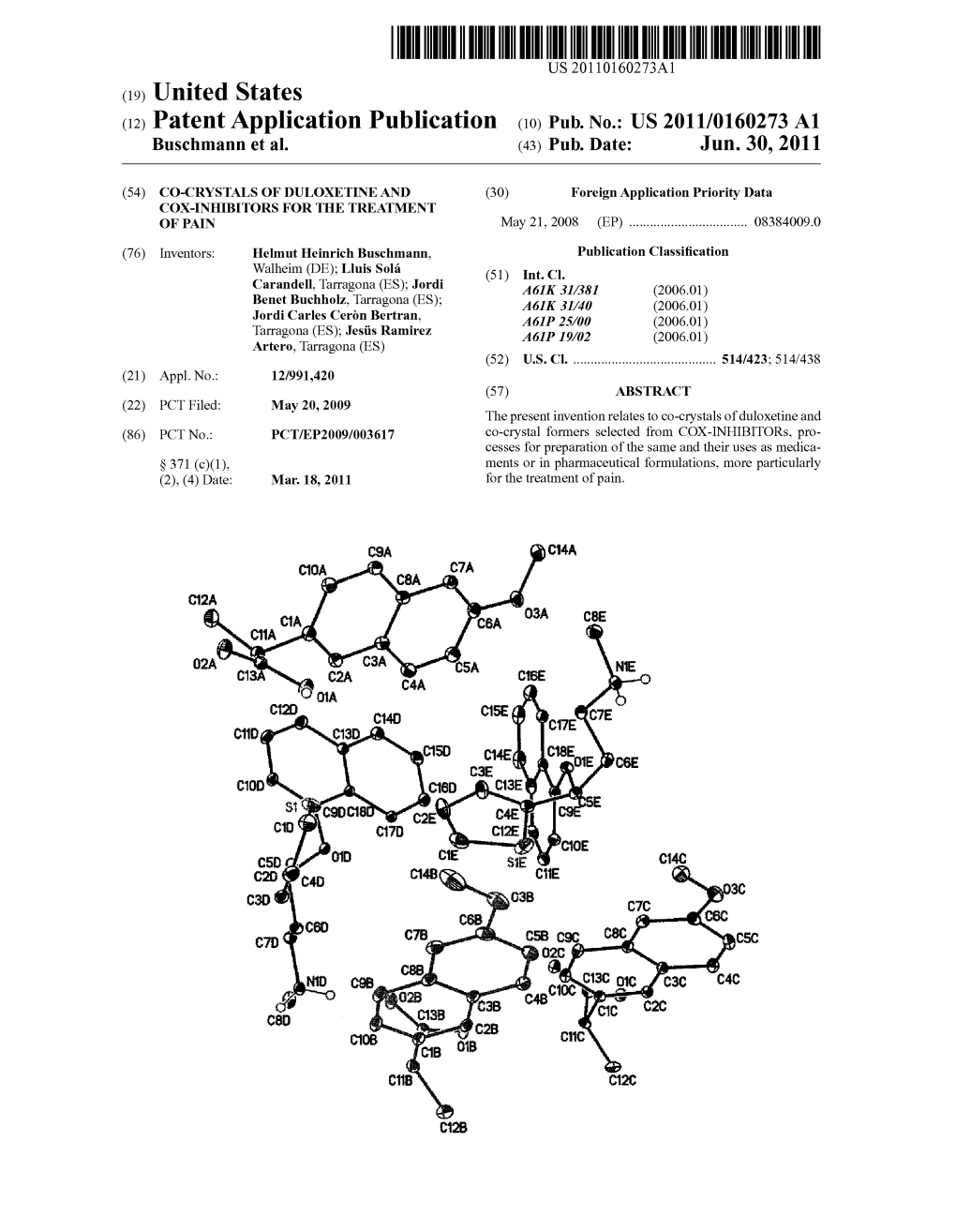
(12) Patent Application Publication (10) Pub. No.: US 2011/0160273 A1 Buschmann Et Al
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

United States Patent (19) 11 Patent Number: 5,955,504 Wechter Et Al
USOO5955504A United States Patent (19) 11 Patent Number: 5,955,504 Wechter et al. (45) Date of Patent: Sep. 21, 1999 54 COLORECTAL CHEMOPROTECTIVE Marnett, “Aspirin and the Potential Role of Prostaglandins COMPOSITION AND METHOD OF in Colon Cancer, Cancer Research, 1992; 52:5575–89. PREVENTING COLORECTAL CANCER Welberg et al., “Proliferation Rate of Colonic Mucosa in Normal Subjects and Patients with Colonic Neoplasms: A 75 Inventors: William J. Wechter; John D. Refined Immunohistochemical Method.” J. Clin Pathol, McCracken, both of Redlands, Calif. 1990; 43:453-456. Thun et al., “Aspirin Use and Reduced Risk of Fatal Colon 73 Assignee: Loma Linda University Medical Cancer." N Engl J Med 1991; 325:1593-6. Center, Loma Linda, Calif. Peleg, et al., “Aspirin and Nonsteroidal Anti-inflammatory Drug Use and the Risk of Subsequent Colorectal Cancer.” 21 Appl. No.: 08/402,797 Arch Intern Med. 1994, 154:394–399. 22 Filed: Mar 13, 1995 Gridley, et al., “Incidence of Cancer among Patients With Rheumatoid Arthritis J. Natl Cancer Inst 1993 85:307-311. 51) Int. Cl. .......................... A61K 31/19; A61K 31/40; Labayle, et al., “Sulindac Causes Regression Of Rectal A61K 31/42 Polyps. In Familial Adenomatous Polyposis” Gastroenterol 52 U.S. Cl. .......................... 514/568; 514/569; 514/428; ogy 1991 101:635-639. 514/416; 514/375 Rigau, et al., “Effects Of Long-Term Sulindac Therapy On 58 Field of Search ..................................... 514/568, 570, Colonic Polyposis” Annals of Internal Medicine 1991 514/569, 428, 416, 375 11.5:952-954. Giardiello.et al., “Treatment Of Colonic and Rectal 56) References Cited Adenomas With Sulindac In Familial Adenomatous Poly U.S. -

What Are the Acute Treatments for Migraine and How Are They Used?
2. Acute Treatment CQ II-2-1 What are the acute treatments for migraine and how are they used? Recommendation The mainstay of acute treatment for migraine is pharmacotherapy. The drugs used include (1) acetaminophen, (2) non-steroidal anti-inflammatory drugs (NSAIDs), (3) ergotamines, (4) triptans and (5) antiemetics. Stratified treatment according to the severity of migraine is recommended: use NSAIDs such as aspirin and naproxen for mild to moderate headache, and use triptans for moderate to severe headache, or even mild to moderate headache when NSAIDs were ineffective in the past. It is necessary to give guidance and cautions to patients having acute attacks, and explain the methods of using medications (timing, dose, frequency of use) and medication use during pregnancy and breast-feeding. Grade A Background and Objective The objective of acute treatment is to resolve the migraine attack completely and rapidly and restore the patient’s normal functions. An ideal treatment should have the following characteristics: (1) resolves pain and associated symptoms rapidly; (2) is consistently effective; (3) no recurrence; (4) no need for additional use of medication; (5) no adverse effects; (6) can be administered by the patients themselves; and (7) low cost. Literature was searched to identify acute treatments that satisfy the above conditions. Comments and Evidence The acute treatment drugs for migraine generally include (1) acetaminophens, (2) non-steroidal anti-inflammatory drugs (NSAIDs), (3) ergotamines, (4) triptans, and (5) antiemetics. For severe migraines including status migrainosus and migraine attacks refractory to treatment, (6) anesthetics, and (7) corticosteroids (dexamethasone) are used (Tables 1 and 2).1)-9) There are two approaches to the selection and sequencing of these medications: “step care” and “stratified care”. -

Comparison of Ketorolac 0.4 and Nepafenac 0.1 for the Prevention Of
Downloaded from http://bjo.bmj.com/ on June 11, 2015 - Published by group.bmj.com Clinical science Comparison of ketorolac 0.4% and nepafenac 0.1% for the prevention of cystoid macular oedema after phacoemulsification: prospective placebo-controlled Editor’s choice Scan to access more free content randomised study Patrick Frensel Tzelikis,1 Monike Vieira,1 Wilson Takashi Hida,1,2 Antonio Francisco Motta,2 Celso Takashi Nakano,2 Eliane Mayumi Nakano,1 Milton Ruiz Alves2 1Brasilia Ophthalmologic ABSTRACT disposal, but there is still debate over whether to Hospital, HOB, Brasilia, DF, Purpose To compare the anti-inflammatory efficacy of use them in combination for routine cases. Brazil 2Ophthalmology Department, ketorolac of tromethamine 0.4% and nepafenac 0.1% Despite a convincing theory of pathogenesis Faculdade de Medicina, eye drops for prophylaxis of cystoid macular oedema involving migratory inflammatory mediator and Universidade de São Paulo, (CME) after small-incision cataract extraction. hyperpermeability of retinal blood vessels, particu- São Paulo, SP, Brazil Methods Patients were assigned randomly to three larly the retinal capillary bed, evidence-based medi- groups. Group 1 patients received a topical artificial tear cine still regards prophylaxis and treatment with Correspondence to fl Dr Patrick F Tzelikis, Brasilia substitute (placebo); group 2 received ketorolac anti-in ammatory agents as unproven. The present Ophthalmologic Hospital, HOB, tromethamine 0.4% (Acular LS, Allergan) and group 3 study evaluated the effects of ophthalmic solutions SQN 203, bloco K, apart 502, received nepafenac 0.1% (Nevanac, Alcon). The of the NSAIDs ketorolac 0.4% and nepafenac 0.1% Brasilia, DF 70833-110, Brazil; incidence and severity of CME were evaluated by retinal in preventing CME assessed by optical coherence [email protected] foveal thickness on optical coherence tomography (OCT) tomography (OCT) using central subfield thickness 3 Received 15 August 2014 after 1, 4 and 12 weeks. -

Short Reports
Br Med J (Clin Res Ed): first published as 10.1136/bmj.287.6395.798 on 17 September 1983. Downloaded from 798 BRITISH MEDICAL JOURNAL VOLUME 287 17 SEPTEMBER 1983 SHORT REPORTS analgesic properties. The usual daily dosage is 400 mg. The amount Piroxicam poisoning of fentiazac absorbed after gastric lavage would probably be small and of little importance. Piroxicam is a potent non-steroidal anti-inflammatory drug. Ex- The main signs were altered consciousness and muscle twitchings. perience with overdosage is limited. We describe a patient who took The exact causes of these were unknown, but griseofulvin has been a large dose of piroxicam. associated with dizziness, mental confusion, irritability, and impaired coordination.2 Side effects of piroxicam have occasionally included headache and dizziness, although their incidences in double blind Case report comparative studies have been lower than those reported with indomethacin.3 Thus the drowsiness and muscle twitchings could not A 25 year old Chinese man weighing 46 kg was admitted to hospital be definitely attributed to piroxicam. about two hours after ingesting 56 capsules of 10 mg piroxicam, 97 tablets Gastrointestinal bleeding is usually the major side effect of most of 125 mg griseofulvin, and about 10 tablets of 100 mg fentiazac. He had been drugs. Piroxicam seemed to be well suffering from psoriatic arthropathy affecting both knee and ankle joints non-steroidal anti-inflammatory and had been taking piroxicam 30 mg daily for nine months. Other anal- tolerated since the patient did not complain of gastric discomfort or gesics, including aspirin, paracetamol, fentiazac, and indomethacin, had gastrointestinal bleeding despite a history of gastric erosions. -

WO 2011/095814 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date _ . , 11 August 2011 (11.08.2011) WO 2011/095814 Al (51) International Patent Classification: (74) Agents: HUTTER, Anton et al; Venner Shipley LLP, 20 A61K 31/192 (2006.01) A61K 47/14 (2006.01) Little Britain, London EC1A 7DH (GB). A61K 47/44 (2006.01) (81) Designated States (unless otherwise indicated, for every (21) International Application Number: kind of national protection available): AE, AG, AL, AM, PCT/GB201 1/050189 AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (22) International Filing Date: DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, 4 February 201 1 (04.02.201 1) HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (25) Filing Language: English KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (26) Publication Language: English NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (30) Priority Data: SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, 1001 912.3 5 February 2010 (05.02.2010) GB TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. 1015878.0 22 September 2010 (22.09.2010) GB (84) Designated States (unless otherwise indicated, for every (71) Applicant (for all designated States except US): BIOCO- kind of regional protection available): ARIPO (BW, GH, PEA LIMITED [GB/GB]; CentralPoint, 45 Beech Street, GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, London EC2Y 8AD (GB). -

Estonian Statistics on Medicines 2016 1/41
Estonian Statistics on Medicines 2016 ATC code ATC group / Active substance (rout of admin.) Quantity sold Unit DDD Unit DDD/1000/ day A ALIMENTARY TRACT AND METABOLISM 167,8985 A01 STOMATOLOGICAL PREPARATIONS 0,0738 A01A STOMATOLOGICAL PREPARATIONS 0,0738 A01AB Antiinfectives and antiseptics for local oral treatment 0,0738 A01AB09 Miconazole (O) 7088 g 0,2 g 0,0738 A01AB12 Hexetidine (O) 1951200 ml A01AB81 Neomycin+ Benzocaine (dental) 30200 pieces A01AB82 Demeclocycline+ Triamcinolone (dental) 680 g A01AC Corticosteroids for local oral treatment A01AC81 Dexamethasone+ Thymol (dental) 3094 ml A01AD Other agents for local oral treatment A01AD80 Lidocaine+ Cetylpyridinium chloride (gingival) 227150 g A01AD81 Lidocaine+ Cetrimide (O) 30900 g A01AD82 Choline salicylate (O) 864720 pieces A01AD83 Lidocaine+ Chamomille extract (O) 370080 g A01AD90 Lidocaine+ Paraformaldehyde (dental) 405 g A02 DRUGS FOR ACID RELATED DISORDERS 47,1312 A02A ANTACIDS 1,0133 Combinations and complexes of aluminium, calcium and A02AD 1,0133 magnesium compounds A02AD81 Aluminium hydroxide+ Magnesium hydroxide (O) 811120 pieces 10 pieces 0,1689 A02AD81 Aluminium hydroxide+ Magnesium hydroxide (O) 3101974 ml 50 ml 0,1292 A02AD83 Calcium carbonate+ Magnesium carbonate (O) 3434232 pieces 10 pieces 0,7152 DRUGS FOR PEPTIC ULCER AND GASTRO- A02B 46,1179 OESOPHAGEAL REFLUX DISEASE (GORD) A02BA H2-receptor antagonists 2,3855 A02BA02 Ranitidine (O) 340327,5 g 0,3 g 2,3624 A02BA02 Ranitidine (P) 3318,25 g 0,3 g 0,0230 A02BC Proton pump inhibitors 43,7324 A02BC01 Omeprazole -

(12) United States Patent (10) Patent No.: US 6,221,377 B1 Meyer (45) Date of Patent: Apr
USOO6221377B1 (12) United States Patent (10) Patent No.: US 6,221,377 B1 Meyer (45) Date of Patent: Apr. 24, 2001 (54) ADMINISTRATION MEDIA FOR (56) References Cited ANALGESIC, ANTI-INFLAMMATORY AND ANT-PYRETC DRUGS CONTAINING FOREIGN PATENT DOCUMENTS NITROUS OXDE AND PHARMACEUTICAL 2 277 264 10/1994 (GB). COMPOSITIONS CONTAINING SUCH MEDIA AND DRUGS OTHER PUBLICATIONS (75) Inventor: Petrus Johannes Meyer, Randburg Chemical ABstracts AN 1985:464783, Hertz et al., Jan. (ZA) 1985.* Chemical Abstracts AN 1978:44978, Berkowitz et al., Jan. (73) Assignee: Pitmy International N.V., Bonaire 1977.* (NL) International Publication No. WO93/25213, published Dec. 23, 1993. (*) Notice: Subject to any disclaimer, the term of this Mikrochim. Acta, vol. 2, No. 5-6, 1980 pp. 464–474, Stahl patent is extended or adjusted under 35 et al., Extraction of natural Substances using Supercritical U.S.C. 154(b) by 0 days. and liquified gases. (21) Appl. No.: 09/068,543 Reynolds J.E.F. et al., “Martindale', 1989, The Pharmaceu tical Press. (22) PCT Filed: Nov. 13, 1996 Science, vol. 194, No. 4268, 1976, pp. 967–968, Berkowitz (86) PCT No.: PCT/IB96/01366 et al., "Nitrous oxide “analgesia': resemblance to opiate action'. S371 Date: May 13, 1998 Steinegger et al., “Lehrbuch der Pharmakognosie und Phy S 102(e) Date: May 13, 1998 topharmazie', Edition 4, 1988. (87) PCT Pub. No.: WO97/17978 * cited by examiner PCT Pub. Date: May 22, 1997 Primary Examiner Jyothsna Venkat (30) Foreign Application Priority Data ASSistant Examiner-Grace Hsu (74) Attorney, Agent, or Firm-Arent Fox Kintner Plotkin Nov. 13, 1995 (ZA) ................................................... -

2021 Formulary List of Covered Prescription Drugs
2021 Formulary List of covered prescription drugs This drug list applies to all Individual HMO products and the following Small Group HMO products: Sharp Platinum 90 Performance HMO, Sharp Platinum 90 Performance HMO AI-AN, Sharp Platinum 90 Premier HMO, Sharp Platinum 90 Premier HMO AI-AN, Sharp Gold 80 Performance HMO, Sharp Gold 80 Performance HMO AI-AN, Sharp Gold 80 Premier HMO, Sharp Gold 80 Premier HMO AI-AN, Sharp Silver 70 Performance HMO, Sharp Silver 70 Performance HMO AI-AN, Sharp Silver 70 Premier HMO, Sharp Silver 70 Premier HMO AI-AN, Sharp Silver 73 Performance HMO, Sharp Silver 73 Premier HMO, Sharp Silver 87 Performance HMO, Sharp Silver 87 Premier HMO, Sharp Silver 94 Performance HMO, Sharp Silver 94 Premier HMO, Sharp Bronze 60 Performance HMO, Sharp Bronze 60 Performance HMO AI-AN, Sharp Bronze 60 Premier HDHP HMO, Sharp Bronze 60 Premier HDHP HMO AI-AN, Sharp Minimum Coverage Performance HMO, Sharp $0 Cost Share Performance HMO AI-AN, Sharp $0 Cost Share Premier HMO AI-AN, Sharp Silver 70 Off Exchange Performance HMO, Sharp Silver 70 Off Exchange Premier HMO, Sharp Performance Platinum 90 HMO 0/15 + Child Dental, Sharp Premier Platinum 90 HMO 0/20 + Child Dental, Sharp Performance Gold 80 HMO 350 /25 + Child Dental, Sharp Premier Gold 80 HMO 250/35 + Child Dental, Sharp Performance Silver 70 HMO 2250/50 + Child Dental, Sharp Premier Silver 70 HMO 2250/55 + Child Dental, Sharp Premier Silver 70 HDHP HMO 2500/20% + Child Dental, Sharp Performance Bronze 60 HMO 6300/65 + Child Dental, Sharp Premier Bronze 60 HDHP HMO -

Fentiazac (BAN, USAN, Rinn) Preparations Mate Aluminium); 16449-54-0 (Flufenamate Aluminium)
60 Analgesics Anti-inflammatory Drugs and Antipyretics sia after caesarean section confirmed an additive analgesic effect Fepradinol (rINN) Floctafenine (BAN, USAN, rINN) for the combination, there was no demonstrable clinical benefit ± α Floctafenina; Floctafénine; Floctafeninum; R-4318; RU-15750. compared with fentanyl alone in this patient group who expect Fépradinol; Fepradinolum. ( )- -{[(2-Hydroxy-1,1-dimethyle- N early mobilisation. However, the combination may be of greater thyl)amino]methyl}benzyl alcohol. 2,3-Dihydroxypropyl -(8-trifluoromethyl-4-quinolyl)anthrani- benefit in patients for whom early ambulation is not routine. Фепрадинол late. Флоктафенин Fentanyl has also been given by epidural injection to children for C12H19NO2 = 209.3. 7 postoperative analgesia. CAS — 63075-47-8. C20H17F3N2O4 = 406.4. Fentanyl has been tried by intrathecal injection for postoperative CAS — 23779-99-9. pain.8 ATC — N02BG04. As mentioned in Administration, Transdermal Route, above, an OH ATC Vet — QN02BG04. iontophoretic transdermal system for postoperative pain is also available.9-11 CH 3 OH 1. Mitchell RWD, Smith G. The control of acute postoperative N pain. Br J Anaesth 1989; 63: 147–58. HO O O 2. Morgan M. The rational use of intrathecal and extradural opio- H CH3 ids. Br J Anaesth 1989; 63: 165–88. OH H 3. Grass JA, et al. A randomized, double-blind, dose-response comparison of epidural fentanyl versus sufentanil analgesia af- N ter cesarean section. Anesth Analg 1997; 85: 365–71. Profile CF3 4. Swarm RA, et al. Pain treatment in the perioperative period. Fepradinol is an NSAID (p.96) that has been used topically in a N Curr Probl Surg 2001; 38: 835–920. -

Protocol Synopsis
Risk of Upper Gastrointestinal Complications in Users of Nonsteroidal Anti-inflammatory Drugs Study Protocol—V2.0 (Final) October 19, 2009 Prepared by Jordi Castellsague Susana Perez-Gutthann RTI Health Solutions E-mail: [email protected] Federica Pisa, MD, MStat Fabio Barbone, MD, DrPH Udine University, AOUD Udine Institute of Hygiene and Epidemiology E-mail: [email protected] RTI-HS Project No.: 0301655 PROTOCOL APPROVAL SIGNATURE PAGE Project Title : Risk of Upper Gastrointestinal Complications in Users ofNonsteroidal Anti-inflammatory Drugs RTI-HS Project 0301655 Final Appro val Date: October 22,2009 Authors: Jordi Castellsague, MD, MPH; Susana Perez-Gutthann, MD, PhD (RTI HS) Fede rica Pisa, MD, MStat; Fabio Barbone, MD, DrPH (Udine University, AOUD Udine) Version: Version 2.0 Final Version Date: October 19, 2009 The following people have reviewed the protocol and give their approval: Susana Perez-Gutthann, MD, PhD Date Global Head Epidemiology RTI Health Solutions / Fabio Barbone, MD, D H Signature Date Director, Institute of Hygiene and Epidemiology University of Udine ii TABLE OF CONTENTS 1 STUDY SYNOPSIS ............................................................................................ 1 2 BACKGROUND ................................................................................................. 4 3 KEY ROLES AND RESPONSIBILITIES ............................................................ 5 3.1.1 Financial Sponsorship .................................................................................................... -

Marrakesh Agreement Establishing the World Trade Organization
No. 31874 Multilateral Marrakesh Agreement establishing the World Trade Organ ization (with final act, annexes and protocol). Concluded at Marrakesh on 15 April 1994 Authentic texts: English, French and Spanish. Registered by the Director-General of the World Trade Organization, acting on behalf of the Parties, on 1 June 1995. Multilat ral Accord de Marrakech instituant l©Organisation mondiale du commerce (avec acte final, annexes et protocole). Conclu Marrakech le 15 avril 1994 Textes authentiques : anglais, français et espagnol. Enregistré par le Directeur général de l'Organisation mondiale du com merce, agissant au nom des Parties, le 1er juin 1995. Vol. 1867, 1-31874 4_________United Nations — Treaty Series • Nations Unies — Recueil des Traités 1995 Table of contents Table des matières Indice [Volume 1867] FINAL ACT EMBODYING THE RESULTS OF THE URUGUAY ROUND OF MULTILATERAL TRADE NEGOTIATIONS ACTE FINAL REPRENANT LES RESULTATS DES NEGOCIATIONS COMMERCIALES MULTILATERALES DU CYCLE D©URUGUAY ACTA FINAL EN QUE SE INCORPOR N LOS RESULTADOS DE LA RONDA URUGUAY DE NEGOCIACIONES COMERCIALES MULTILATERALES SIGNATURES - SIGNATURES - FIRMAS MINISTERIAL DECISIONS, DECLARATIONS AND UNDERSTANDING DECISIONS, DECLARATIONS ET MEMORANDUM D©ACCORD MINISTERIELS DECISIONES, DECLARACIONES Y ENTEND MIENTO MINISTERIALES MARRAKESH AGREEMENT ESTABLISHING THE WORLD TRADE ORGANIZATION ACCORD DE MARRAKECH INSTITUANT L©ORGANISATION MONDIALE DU COMMERCE ACUERDO DE MARRAKECH POR EL QUE SE ESTABLECE LA ORGANIZACI N MUND1AL DEL COMERCIO ANNEX 1 ANNEXE 1 ANEXO 1 ANNEX -

The Effect of Prophylactic Nepafenac 0.1% Eye Drops
Ophthalmol Ina 2015;41(3):265-271 265 Original Article The Effect of Prophylactic Nepafenac 0.1% Eye Drops on Macular Changes after Phacoemulsiication in Non- Proliferative Diabetic Retinopathy Patients Using Spectral Domain Optical Coherence Tomography (SD-OCT) Soeiandi Soedarman *, Ari Djatikusumo *, Syska Widyawati *, Arini Setiawati ** * Department of Ophthalmology, Faculty of Medicine, Indonesia University Cipto Mangunkusumo Hospital, Jakarta ** Department of Pharmacology & Therapeutic, Faculty of Medicine, Indonesia University Cipto Manungkusumo Hospital, Jakarta ABSTRACT Background: To evaluate the effect of prophylactic nepafenac eye drops on macular thickness changes after phacoemulsiication surgery in mild to moderate NPDR patients. Method: This study is an open label randomized clinical trial. Thirty-six subjects who met the inclusion criteria underwent phacoemulsiication. One group (18 subjects) were given nepafenac 0.1% eye drops and the rest were given placebo. Foveal thickness was measured by SD-OCT before surgery and the fourth week after phacoemulsiication. Best corrected visual acuity (BCVA) and degree of inlammation in the anterior chamber were also being assessed. Result: There was a statistically signiicant increase foveal thickness in the placebo group 4 weeks after phacoemulsiication (p=0.022). Clinically, percentage degree of inlammation in anterior chamber in placebo group was higher than nepafenac group (38.9% : 5.6%) but not signiicantly different between 2 groups (p=0.27). Nepafenac group achieved clinically better BCVA than the placebo group 4 weeks after phacoemulsiication, although statistically there was no signiicant difference between 2 groups (p=0.991). Conclusion: Nepafenac 0.1% eye drops could prevent foveal thickening 4 weeks after phacoemulsiication in mild to moderate NPDR patients.