Examination of Morphological and Habitat Variation Within Stenanthium
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Guide to the Flora of the Carolinas, Virginia, and Georgia, Working Draft of 17 March 2004 -- LILIACEAE
Guide to the Flora of the Carolinas, Virginia, and Georgia, Working Draft of 17 March 2004 -- LILIACEAE LILIACEAE de Jussieu 1789 (Lily Family) (also see AGAVACEAE, ALLIACEAE, ALSTROEMERIACEAE, AMARYLLIDACEAE, ASPARAGACEAE, COLCHICACEAE, HEMEROCALLIDACEAE, HOSTACEAE, HYACINTHACEAE, HYPOXIDACEAE, MELANTHIACEAE, NARTHECIACEAE, RUSCACEAE, SMILACACEAE, THEMIDACEAE, TOFIELDIACEAE) As here interpreted narrowly, the Liliaceae constitutes about 11 genera and 550 species, of the Northern Hemisphere. There has been much recent investigation and re-interpretation of evidence regarding the upper-level taxonomy of the Liliales, with strong suggestions that the broad Liliaceae recognized by Cronquist (1981) is artificial and polyphyletic. Cronquist (1993) himself concurs, at least to a degree: "we still await a comprehensive reorganization of the lilies into several families more comparable to other recognized families of angiosperms." Dahlgren & Clifford (1982) and Dahlgren, Clifford, & Yeo (1985) synthesized an early phase in the modern revolution of monocot taxonomy. Since then, additional research, especially molecular (Duvall et al. 1993, Chase et al. 1993, Bogler & Simpson 1995, and many others), has strongly validated the general lines (and many details) of Dahlgren's arrangement. The most recent synthesis (Kubitzki 1998a) is followed as the basis for familial and generic taxonomy of the lilies and their relatives (see summary below). References: Angiosperm Phylogeny Group (1998, 2003); Tamura in Kubitzki (1998a). Our “liliaceous” genera (members of orders placed in the Lilianae) are therefore divided as shown below, largely following Kubitzki (1998a) and some more recent molecular analyses. ALISMATALES TOFIELDIACEAE: Pleea, Tofieldia. LILIALES ALSTROEMERIACEAE: Alstroemeria COLCHICACEAE: Colchicum, Uvularia. LILIACEAE: Clintonia, Erythronium, Lilium, Medeola, Prosartes, Streptopus, Tricyrtis, Tulipa. MELANTHIACEAE: Amianthium, Anticlea, Chamaelirium, Helonias, Melanthium, Schoenocaulon, Stenanthium, Veratrum, Toxicoscordion, Trillium, Xerophyllum, Zigadenus. -

State of New York City's Plants 2018
STATE OF NEW YORK CITY’S PLANTS 2018 Daniel Atha & Brian Boom © 2018 The New York Botanical Garden All rights reserved ISBN 978-0-89327-955-4 Center for Conservation Strategy The New York Botanical Garden 2900 Southern Boulevard Bronx, NY 10458 All photos NYBG staff Citation: Atha, D. and B. Boom. 2018. State of New York City’s Plants 2018. Center for Conservation Strategy. The New York Botanical Garden, Bronx, NY. 132 pp. STATE OF NEW YORK CITY’S PLANTS 2018 4 EXECUTIVE SUMMARY 6 INTRODUCTION 10 DOCUMENTING THE CITY’S PLANTS 10 The Flora of New York City 11 Rare Species 14 Focus on Specific Area 16 Botanical Spectacle: Summer Snow 18 CITIZEN SCIENCE 20 THREATS TO THE CITY’S PLANTS 24 NEW YORK STATE PROHIBITED AND REGULATED INVASIVE SPECIES FOUND IN NEW YORK CITY 26 LOOKING AHEAD 27 CONTRIBUTORS AND ACKNOWLEGMENTS 30 LITERATURE CITED 31 APPENDIX Checklist of the Spontaneous Vascular Plants of New York City 32 Ferns and Fern Allies 35 Gymnosperms 36 Nymphaeales and Magnoliids 37 Monocots 67 Dicots 3 EXECUTIVE SUMMARY This report, State of New York City’s Plants 2018, is the first rankings of rare, threatened, endangered, and extinct species of what is envisioned by the Center for Conservation Strategy known from New York City, and based on this compilation of The New York Botanical Garden as annual updates thirteen percent of the City’s flora is imperiled or extinct in New summarizing the status of the spontaneous plant species of the York City. five boroughs of New York City. This year’s report deals with the City’s vascular plants (ferns and fern allies, gymnosperms, We have begun the process of assessing conservation status and flowering plants), but in the future it is planned to phase in at the local level for all species. -

Draft Plant Propagation Protocol
Plant Propagation Protocol for Zigadenus elegans ESRM 412 – Native Plant Production TAXONOMY Family Names Family Scientific Name: Liliaceae Family Common Name: Lily family Scientific Names Genus: Zigadenus Michx. Species: Zigadenus elegans Species Authority: Pursh Variety: Sub-species: Zigadenus elegans ssp. elegans Zigadenus elegans ssp. glaucus Cultivar: Authority for Variety/Sub-species: Common Synonym(s) Anticlea coloradensis (Rydb.) Rydb. Anticlea elegans (Pursh) Rydb. Zigadenus alpinus Blank. Zigadenus elegans Pursh ssp. elegans 2 Common Name(s): Glaucous death camas, Mountain death camas, White camas 2 Species Code : ZIEL2 GENERAL INFORMATION Geographical range See above 1 Ecological distribution : Occurs in meadows, open forests and rocky slopes, at middle to high elevations in the mountains 2 Other sources indicate it can also be found in moist grasslands, river and lake shores, and bogs in coniferous forests. 6 9 It has also been listed as an indicator species for areas that have been former savanna's/woodlands. Climate and elevation range Subalpine meadows and moist screes at high elevations in the Rockies and Pacific Coast states. 12 Local habitat and abundance; may Occurs in sandy, moist soils. It can tolerate partial include commonly associated shade but also needs sunlight. 5 species It and other indicator species tend to be strongly limited to partial canopy conditions. In more heavily-wooded sites, these species are usually in a state of decline due to the increasing canopy closure above. They are therefore dependent on canopy gaps, edges, roadsides etc. in densely-wooded areas. 9 In Missouri it cam be found on the crevices and ledges of north-facing dolomite bluffs. -

Redalyc.A Conspectus of Mexican Melanthiaceae Including A
Acta Botánica Mexicana ISSN: 0187-7151 [email protected] Instituto de Ecología, A.C. México Frame, Dawn; Espejo, Adolfo; López Ferrari, Ana Rosa A conspectus of mexican Melanthiaceae including a description of new taxa of schoenocaulon and Zigadenus Acta Botánica Mexicana, núm. 48, septiembre, 1999, pp. 27 - 50 Instituto de Ecología, A.C. Pátzcuaro, México Disponible en: http://www.redalyc.org/articulo.oa?id=57404804 Cómo citar el artículo Número completo Sistema de Información Científica Más información del artículo Red de Revistas Científicas de América Latina, el Caribe, España y Portugal Página de la revista en redalyc.org Proyecto académico sin fines de lucro, desarrollado bajo la iniciativa de acceso abierto Acta Botánica Mexicana (1999), 48:27-50 A CONSPECTUS OF MEXICAN MELANTHIACEAE INCLUDING A DESCRIPTION OF NEW TAXA OF SCHOENOCAULON AND ZIGADENUS DAWN FRAME Laboratoire de Botanique, ISEM Institut de Botanique 163, rue A. Broussonet 34090 Montpellier France e-mail: [email protected] ADOLFO ESPEJO Y ANA ROSA LOPEZ-FERRARI Herbario Metropolitano Departamento de Biología, CBS Universidad Autónoma Metropolitana Unidad Iztapalapa Apartado postal 55-535 09340 México, D.F. e-mail: [email protected] ABSTRACT Seven new taxa of Schoenocaulon and one more of Zigadenus from Mexico are herein described. In addition, a brief description of the Mexican genera of Melanthiaceae and a key to genera and species known from Mexico are given. RESUMEN Se describen siete nuevos taxa de Schoenocaulon y uno de Zigadenus provenientes de diversos estados de México. Se incluyen claves para la identificación de los géneros y de las especies de la familia presentes en México y se hacen algunos comentarios breves sobre los mismos. -

GENOME EVOLUTION in MONOCOTS a Dissertation
GENOME EVOLUTION IN MONOCOTS A Dissertation Presented to The Faculty of the Graduate School At the University of Missouri In Partial Fulfillment Of the Requirements for the Degree Doctor of Philosophy By Kate L. Hertweck Dr. J. Chris Pires, Dissertation Advisor JULY 2011 The undersigned, appointed by the dean of the Graduate School, have examined the dissertation entitled GENOME EVOLUTION IN MONOCOTS Presented by Kate L. Hertweck A candidate for the degree of Doctor of Philosophy And hereby certify that, in their opinion, it is worthy of acceptance. Dr. J. Chris Pires Dr. Lori Eggert Dr. Candace Galen Dr. Rose‐Marie Muzika ACKNOWLEDGEMENTS I am indebted to many people for their assistance during the course of my graduate education. I would not have derived such a keen understanding of the learning process without the tutelage of Dr. Sandi Abell. Members of the Pires lab provided prolific support in improving lab techniques, computational analysis, greenhouse maintenance, and writing support. Team Monocot, including Dr. Mike Kinney, Dr. Roxi Steele, and Erica Wheeler were particularly helpful, but other lab members working on Brassicaceae (Dr. Zhiyong Xiong, Dr. Maqsood Rehman, Pat Edger, Tatiana Arias, Dustin Mayfield) all provided vital support as well. I am also grateful for the support of a high school student, Cady Anderson, and an undergraduate, Tori Docktor, for their assistance in laboratory procedures. Many people, scientist and otherwise, helped with field collections: Dr. Travis Columbus, Hester Bell, Doug and Judy McGoon, Julie Ketner, Katy Klymus, and William Alexander. Many thanks to Barb Sonderman for taking care of my greenhouse collection of many odd plants brought back from the field. -

ISB: Atlas of Florida Vascular Plants
Longleaf Pine Preserve Plant List Acanthaceae Asteraceae Wild Petunia Ruellia caroliniensis White Aster Aster sp. Saltbush Baccharis halimifolia Adoxaceae Begger-ticks Bidens mitis Walter's Viburnum Viburnum obovatum Deer Tongue Carphephorus paniculatus Pineland Daisy Chaptalia tomentosa Alismataceae Goldenaster Chrysopsis gossypina Duck Potato Sagittaria latifolia Cow Thistle Cirsium horridulum Tickseed Coreopsis leavenworthii Altingiaceae Elephant's foot Elephantopus elatus Sweetgum Liquidambar styraciflua Oakleaf Fleabane Erigeron foliosus var. foliosus Fleabane Erigeron sp. Amaryllidaceae Prairie Fleabane Erigeron strigosus Simpson's rain lily Zephyranthes simpsonii Fleabane Erigeron vernus Dog Fennel Eupatorium capillifolium Anacardiaceae Dog Fennel Eupatorium compositifolium Winged Sumac Rhus copallinum Dog Fennel Eupatorium spp. Poison Ivy Toxicodendron radicans Slender Flattop Goldenrod Euthamia caroliniana Flat-topped goldenrod Euthamia minor Annonaceae Cudweed Gamochaeta antillana Flag Pawpaw Asimina obovata Sneezeweed Helenium pinnatifidum Dwarf Pawpaw Asimina pygmea Blazing Star Liatris sp. Pawpaw Asimina reticulata Roserush Lygodesmia aphylla Rugel's pawpaw Deeringothamnus rugelii Hempweed Mikania cordifolia White Topped Aster Oclemena reticulata Apiaceae Goldenaster Pityopsis graminifolia Button Rattlesnake Master Eryngium yuccifolium Rosy Camphorweed Pluchea rosea Dollarweed Hydrocotyle sp. Pluchea Pluchea spp. Mock Bishopweed Ptilimnium capillaceum Rabbit Tobacco Pseudognaphalium obtusifolium Blackroot Pterocaulon virgatum -

TELOPEA Publication Date: 13 October 1983 Til
Volume 2(4): 425–452 TELOPEA Publication Date: 13 October 1983 Til. Ro)'al BOTANIC GARDENS dx.doi.org/10.7751/telopea19834408 Journal of Plant Systematics 6 DOPII(liPi Tmst plantnet.rbgsyd.nsw.gov.au/Telopea • escholarship.usyd.edu.au/journals/index.php/TEL· ISSN 0312-9764 (Print) • ISSN 2200-4025 (Online) Telopea 2(4): 425-452, Fig. 1 (1983) 425 CURRENT ANATOMICAL RESEARCH IN LILIACEAE, AMARYLLIDACEAE AND IRIDACEAE* D.F. CUTLER AND MARY GREGORY (Accepted for publication 20.9.1982) ABSTRACT Cutler, D.F. and Gregory, Mary (Jodrell(Jodrel/ Laboratory, Royal Botanic Gardens, Kew, Richmond, Surrey, England) 1983. Current anatomical research in Liliaceae, Amaryllidaceae and Iridaceae. Telopea 2(4): 425-452, Fig.1-An annotated bibliography is presented covering literature over the period 1968 to date. Recent research is described and areas of future work are discussed. INTRODUCTION In this article, the literature for the past twelve or so years is recorded on the anatomy of Liliaceae, AmarylIidaceae and Iridaceae and the smaller, related families, Alliaceae, Haemodoraceae, Hypoxidaceae, Ruscaceae, Smilacaceae and Trilliaceae. Subjects covered range from embryology, vegetative and floral anatomy to seed anatomy. A format is used in which references are arranged alphabetically, numbered and annotated, so that the reader can rapidly obtain an idea of the range and contents of papers on subjects of particular interest to him. The main research trends have been identified, classified, and check lists compiled for the major headings. Current systematic anatomy on the 'Anatomy of the Monocotyledons' series is reported. Comment is made on areas of research which might prove to be of future significance. -

Floristic Discoveries in Delaware, Maryland, and Virginia
Knapp, W.M., R.F.C. Naczi, W.D. Longbottom, C.A. Davis, W.A. McAvoy, C.T. Frye, J.W. Harrison, and P. Stango, III. 2011. Floristic discoveries in Delaware, Maryland, and Virginia. Phytoneuron 2011-64: 1–26. Published 15 December 2011. ISSN 2153 733X FLORISTIC DISCOVERIES IN DELAWARE, MARYLAND, AND VIRGINIA WESLEY M. KNAPP 1 Maryland Department of Natural Resources Wildlife and Heritage Service Wye Mills, Maryland 21679 [email protected] ROBERT F. C. NACZI The New York Botanical Garden Bronx, New York 10458-5126 WAYNE D. LONGBOTTOM P.O. Box 634 Preston, Maryland 21655 CHARLES A. DAVIS 1510 Bellona Ave. Lutherville, Maryland 21093 WILLIAM A. MCAVOY Delaware Natural Heritage and Endangered Species Program 4876 Hay Point, Landing Rd. Smyrna, Delaware 19977 CHRISTOPHER T. FRYE Maryland Department of Natural Resources Wildlife and Heritage Service Wye Mills, Maryland 21679 JASON W. HARRISON Maryland Department of Natural Resources Wildlife and Heritage Service Wye Mills, Maryland 21679 PETER STANGO III Maryland Department of Natural Resources, Wildlife and Heritage Service, Annapolis, Maryland 21401 1 Author for correspondence ABSTRACT Over the past decade studies in the field and herbaria have yielded significant advancements in the knowledge of the floras of Delaware, Maryland, and the Eastern Shore of Virginia. We here discuss fifty-two species newly discovered or rediscovered or whose range or nativity is clarified. Eighteen are additions to the flora of Delaware ( Carex lucorum var. lucorum, Carex oklahomensis, Cyperus difformis, Cyperus flavicomus, Elymus macgregorii, Glossostigma cleistanthum, Houstonia pusilla, Juncus validus var. validus, Lotus tenuis, Melothria pendula var. pendula, Parapholis incurva, Phyllanthus caroliniensis subsp. -
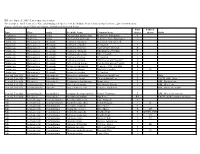
Threatened and Endangered Species List
Effective April 15, 2009 - List is subject to revision For a complete list of Tennessee's Rare and Endangered Species, visit the Natural Areas website at http://tennessee.gov/environment/na/ Aquatic and Semi-aquatic Plants and Aquatic Animals with Protected Status State Federal Type Class Order Scientific Name Common Name Status Status Habit Amphibian Amphibia Anura Gyrinophilus gulolineatus Berry Cave Salamander T Amphibian Amphibia Anura Gyrinophilus palleucus Tennessee Cave Salamander T Crustacean Malacostraca Decapoda Cambarus bouchardi Big South Fork Crayfish E Crustacean Malacostraca Decapoda Cambarus cymatilis A Crayfish E Crustacean Malacostraca Decapoda Cambarus deweesae Valley Flame Crayfish E Crustacean Malacostraca Decapoda Cambarus extraneus Chickamauga Crayfish T Crustacean Malacostraca Decapoda Cambarus obeyensis Obey Crayfish T Crustacean Malacostraca Decapoda Cambarus pristinus A Crayfish E Crustacean Malacostraca Decapoda Cambarus williami "Brawley's Fork Crayfish" E Crustacean Malacostraca Decapoda Fallicambarus hortoni Hatchie Burrowing Crayfish E Crustacean Malocostraca Decapoda Orconectes incomptus Tennessee Cave Crayfish E Crustacean Malocostraca Decapoda Orconectes shoupi Nashville Crayfish E LE Crustacean Malocostraca Decapoda Orconectes wrighti A Crayfish E Fern and Fern Ally Filicopsida Polypodiales Dryopteris carthusiana Spinulose Shield Fern T Bogs Fern and Fern Ally Filicopsida Polypodiales Dryopteris cristata Crested Shield-Fern T FACW, OBL, Bogs Fern and Fern Ally Filicopsida Polypodiales Trichomanes boschianum -

Ecological Checklist of the Missouri Flora for Floristic Quality Assessment
Ladd, D. and J.R. Thomas. 2015. Ecological checklist of the Missouri flora for Floristic Quality Assessment. Phytoneuron 2015-12: 1–274. Published 12 February 2015. ISSN 2153 733X ECOLOGICAL CHECKLIST OF THE MISSOURI FLORA FOR FLORISTIC QUALITY ASSESSMENT DOUGLAS LADD The Nature Conservancy 2800 S. Brentwood Blvd. St. Louis, Missouri 63144 [email protected] JUSTIN R. THOMAS Institute of Botanical Training, LLC 111 County Road 3260 Salem, Missouri 65560 [email protected] ABSTRACT An annotated checklist of the 2,961 vascular taxa comprising the flora of Missouri is presented, with conservatism rankings for Floristic Quality Assessment. The list also provides standardized acronyms for each taxon and information on nativity, physiognomy, and wetness ratings. Annotated comments for selected taxa provide taxonomic, floristic, and ecological information, particularly for taxa not recognized in recent treatments of the Missouri flora. Synonymy crosswalks are provided for three references commonly used in Missouri. A discussion of the concept and application of Floristic Quality Assessment is presented. To accurately reflect ecological and taxonomic relationships, new combinations are validated for two distinct taxa, Dichanthelium ashei and D. werneri , and problems in application of infraspecific taxon names within Quercus shumardii are clarified. CONTENTS Introduction Species conservatism and floristic quality Application of Floristic Quality Assessment Checklist: Rationale and methods Nomenclature and taxonomic concepts Synonymy Acronyms Physiognomy, nativity, and wetness Summary of the Missouri flora Conclusion Annotated comments for checklist taxa Acknowledgements Literature Cited Ecological checklist of the Missouri flora Table 1. C values, physiognomy, and common names Table 2. Synonymy crosswalk Table 3. Wetness ratings and plant families INTRODUCTION This list was developed as part of a revised and expanded system for Floristic Quality Assessment (FQA) in Missouri. -

Vascular Flora of The
VASCULAR FLORA OF THE UPPER ETOWAH RIVER WATERSHED, GEORGIA by LISA MARIE KRUSE (Under the Direction of DAVID E. GIANNASI) ABSTRACT The Etowah River Basin in North Georgia is a biologically diverse Southern Appalachian River system, threatened by regional population growth. This is a two-part botanical study in the Upper Etowah watershed. The primary component is a survey of vascular flora. Habitats include riparian zones, lowland forest, tributary drainages, bluffs, and uplands. A total of 662 taxa were inventoried, and seventeen reference communities were described and mapped. Small streams, remote public land, and forested private land are important for plant conservation in this watershed. In the second component, cumulative plant species richness was sampled across six floodplain sites to estimate optimal widths for riparian buffer zones. To include 90% of floodplain species in a buffer, 60-75% of the floodplain width must be protected, depending on the stream size. Soil moisture influences species richness, and is dependent on upland water sources. An optimal buffer would protect hydrologic connections between floodplains and uplands. INDEX WORDS: Etowah River, Southeastern United States, floristic inventory, riparian buffer zone, floodplain plant species, plant habitat conservation VASCULAR FLORA OF THE UPPER ETOWAH RIVER WATERSHED, GEORGIA by LISA MARIE KRUSE B.S., Emory University, 1996 A Thesis Submitted to the Graduate Faculty of The University of Georgia in Partial Fulfillment of the Requirements for the Degree MASTER OF SCIENCE -

Phylogenetic Relationships of Monocots Based on the Highly Informative Plastid Gene Ndhf Thomas J
Aliso: A Journal of Systematic and Evolutionary Botany Volume 22 | Issue 1 Article 4 2006 Phylogenetic Relationships of Monocots Based on the Highly Informative Plastid Gene ndhF Thomas J. Givnish University of Wisconsin-Madison J. Chris Pires University of Wisconsin-Madison; University of Missouri Sean W. Graham University of British Columbia Marc A. McPherson University of Alberta; Duke University Linda M. Prince Rancho Santa Ana Botanic Gardens See next page for additional authors Follow this and additional works at: http://scholarship.claremont.edu/aliso Part of the Botany Commons Recommended Citation Givnish, Thomas J.; Pires, J. Chris; Graham, Sean W.; McPherson, Marc A.; Prince, Linda M.; Patterson, Thomas B.; Rai, Hardeep S.; Roalson, Eric H.; Evans, Timothy M.; Hahn, William J.; Millam, Kendra C.; Meerow, Alan W.; Molvray, Mia; Kores, Paul J.; O'Brien, Heath W.; Hall, Jocelyn C.; Kress, W. John; and Sytsma, Kenneth J. (2006) "Phylogenetic Relationships of Monocots Based on the Highly Informative Plastid Gene ndhF," Aliso: A Journal of Systematic and Evolutionary Botany: Vol. 22: Iss. 1, Article 4. Available at: http://scholarship.claremont.edu/aliso/vol22/iss1/4 Phylogenetic Relationships of Monocots Based on the Highly Informative Plastid Gene ndhF Authors Thomas J. Givnish, J. Chris Pires, Sean W. Graham, Marc A. McPherson, Linda M. Prince, Thomas B. Patterson, Hardeep S. Rai, Eric H. Roalson, Timothy M. Evans, William J. Hahn, Kendra C. Millam, Alan W. Meerow, Mia Molvray, Paul J. Kores, Heath W. O'Brien, Jocelyn C. Hall, W. John Kress, and Kenneth J. Sytsma This article is available in Aliso: A Journal of Systematic and Evolutionary Botany: http://scholarship.claremont.edu/aliso/vol22/iss1/ 4 Aliso 22, pp.