Two Sisters from Qatar with TUSC3 Genetic Mutation: Psychiatric Considerations
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Tumor Suppressor Candidate 3 (TUSC3)
IJC International Journal of Cancer Tumor suppressor candidate 3 (TUSC3) prevents the epithelial- to-mesenchymal transition and inhibits tumor growth by modulating the endoplasmic reticulum stress response in ovarian cancer cells Katerina Kratochvılova1,2, Peter Horak3, Milan Esner1, Karel Soucˇek2,4,5, Dietmar Pils6, Mariam Anees3,7, Erwin Tomasich3, Frantisek Drafi 1, Veronika Jurtıkova1, Ales Hampl1, Michael Krainer3 and Petr Vanhara 1 1 Department of Histology and Embryology, Faculty of Medicine, Masaryk University, Brno, Czech Republic 2 Center of Biomolecular and Cellular Engineering, International Clinical Research Center, St. Anne’s University Hospital, Brno, Czech Republic 3 Department of Internal Medicine I and Comprehensive Cancer Center, Medical University of Vienna, Vienna, Austria 4 Department of Cytokinetics, Institute of Biophysics, Academy of Sciences of the Czech Republic, v.v.i., Brno, Czech Republic 5 Department of Experimental Biology, Faculty of Science, Masaryk University, Brno, Czech Republic 6 Department of Obstetrics and Gynecology, Molecular Oncology Group, Medical University of Vienna, Vienna, Austria 7 Department of Biochemistry, Quaid-i-Azam University, Islamabad, Pakistan Ovarian cancer is one of the most common malignancies in women and contributes greatly to cancer-related deaths. Tumor suppressor candidate 3 (TUSC3) is a putative tumor suppressor gene located at chromosomal region 8p22, which is often lost in epithelial cancers. Epigenetic silencing of TUSC3 has been associated with poor prognosis, and hypermethylation of its pro- moter provides an independent biomarker of overall and disease-free survival in ovarian cancer patients. TUSC3 is localized to the endoplasmic reticulum in an oligosaccharyl tranferase complex responsible for the N-glycosylation of proteins. However, the precise molecular role of TUSC3 in ovarian cancer remains unclear. -

Integrating Single-Step GWAS and Bipartite Networks Reconstruction Provides Novel Insights Into Yearling Weight and Carcass Traits in Hanwoo Beef Cattle
animals Article Integrating Single-Step GWAS and Bipartite Networks Reconstruction Provides Novel Insights into Yearling Weight and Carcass Traits in Hanwoo Beef Cattle Masoumeh Naserkheil 1 , Abolfazl Bahrami 1 , Deukhwan Lee 2,* and Hossein Mehrban 3 1 Department of Animal Science, University College of Agriculture and Natural Resources, University of Tehran, Karaj 77871-31587, Iran; [email protected] (M.N.); [email protected] (A.B.) 2 Department of Animal Life and Environment Sciences, Hankyong National University, Jungang-ro 327, Anseong-si, Gyeonggi-do 17579, Korea 3 Department of Animal Science, Shahrekord University, Shahrekord 88186-34141, Iran; [email protected] * Correspondence: [email protected]; Tel.: +82-31-670-5091 Received: 25 August 2020; Accepted: 6 October 2020; Published: 9 October 2020 Simple Summary: Hanwoo is an indigenous cattle breed in Korea and popular for meat production owing to its rapid growth and high-quality meat. Its yearling weight and carcass traits (backfat thickness, carcass weight, eye muscle area, and marbling score) are economically important for the selection of young and proven bulls. In recent decades, the advent of high throughput genotyping technologies has made it possible to perform genome-wide association studies (GWAS) for the detection of genomic regions associated with traits of economic interest in different species. In this study, we conducted a weighted single-step genome-wide association study which combines all genotypes, phenotypes and pedigree data in one step (ssGBLUP). It allows for the use of all SNPs simultaneously along with all phenotypes from genotyped and ungenotyped animals. Our results revealed 33 relevant genomic regions related to the traits of interest. -

Computational Analysis and Polymorphism Study of Tumor Suppressor Candidate Gene-3 for Non Syndromic Autosomal Recessive Mental Retardation
Naveed et al., J Appl Bioinforma Comput Biol 2016, 5:2 DOI: 10.4172/2329-9533.1000127 Journal of Applied Bioinformatics & Computational Biology Research Article a SciTechnol journal social, practical adaptive skills and conceptual, originating before 18 Computational Analysis and years [1] and intellectual functioning (I.Q below 70%). It is observed that 1- 3% of general population is affected with MR [2]. Because large Polymorphism study of Tumor numbers of X-linked MR genes are involved in mental retardation which gives ratio between mentally retarded males and females which Suppressor Candidate Gene-3 seem to be quite high at 1.4-:1 to 1.6:1 [3]. Intellectual disability has a very diverse etiology [4]. The causes of ID that affect the development for Non Syndromic Autosomal and functioning of CNS prenatally or postnatally can involve both Recessive Mental Retardation genetic and environmental factors [5]. Muhammad Naveed*, Syeda Khushbakht Kazmi, Fiza Anwar, Prenatal causes of mental retardation include congenital Fatima Arshad, Tehreem Zafar Dar and Muddassar Zafar infections such as cytomegalovirus, toxoplasmosis, syphilis, rubella, herpes and human immunodeficiency virus; prolonged maternal fever in the first trimester; disclosure to alcohol or anticonvulsants; Abstract and untreated maternal phenylketonuria (PKU). Complications of prematurity especially in exceptionally low-birth-weight infants or Mental Retardation (MR) is regarded as a neuronal malfunction certain postnatal exposure can lead to mental retardation/ID [6]. characterized by a low Intellectual Quotient (IQ). To date, few genes (GRIK2, TUSC3, TRAPPC9, TECR, ST3GAL3, MED23, MAN1B1, Genetic causes of MD include genomic disorders, chromosome NSUN1 PRSS12, CRBN, CC2D1A) for autosomal-recessive non structural abnormalities, monogenic diseases and chromosome syndromic MR (NS-ARMR) have been identified and established aneusomies. -

Heterogeneity Between Primary Colon Carcinoma and Paired Lymphatic and Hepatic Metastases
MOLECULAR MEDICINE REPORTS 6: 1057-1068, 2012 Heterogeneity between primary colon carcinoma and paired lymphatic and hepatic metastases HUANRONG LAN1, KETAO JIN2,3, BOJIAN XIE4, NA HAN5, BINBIN CUI2, FEILIN CAO2 and LISONG TENG3 Departments of 1Gynecology and Obstetrics, and 2Surgical Oncology, Taizhou Hospital, Wenzhou Medical College, Linhai, Zhejiang; 3Department of Surgical Oncology, First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang; 4Department of Surgical Oncology, Sir Run Run Shaw Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang; 5Cancer Chemotherapy Center, Zhejiang Cancer Hospital, Zhejiang University of Chinese Medicine, Hangzhou, Zhejiang, P.R. China Received January 26, 2012; Accepted May 8, 2012 DOI: 10.3892/mmr.2012.1051 Abstract. Heterogeneity is one of the recognized characteris- Introduction tics of human tumors, and occurs on multiple levels in a wide range of tumors. A number of studies have focused on the Intratumor heterogeneity is one of the recognized charac- heterogeneity found in primary tumors and related metastases teristics of human tumors, which occurs on multiple levels, with the consideration that the evaluation of metastatic rather including genetic, protein and macroscopic, in a wide range than primary sites could be of clinical relevance. Numerous of tumors, including breast, colorectal cancer (CRC), non- studies have demonstrated particularly high rates of hetero- small cell lung cancer (NSCLC), prostate, ovarian, pancreatic, geneity between primary colorectal tumors and their paired gastric, brain and renal clear cell carcinoma (1). Over the past lymphatic and hepatic metastases. It has also been proposed decade, a number of studies have focused on the heterogeneity that the heterogeneity between primary colon carcinomas and found in primary tumors and related metastases with the their paired lymphatic and hepatic metastases may result in consideration that the evaluation of metastatic rather than different responses to anticancer therapies. -

Molecular Architecture Underlying Fluid Absorption by the Developing Inner
RESEARCH ARTICLE Molecular architecture underlying fluid absorption by the developing inner ear Keiji Honda1†, Sung Huhn Kim2‡, Michael C Kelly3, Joseph C Burns3§, Laura Constance2, Xiangming Li2#, Fei Zhou2, Michael Hoa4, Matthew W Kelley3, Philine Wangemann2*, Robert J Morell5, Andrew J Griffith1* 1Molecular Biology and Genetics Section, National Institute on Deafness and Other Communication Disorders, National Institutes of Health, Bethesda, United States; 2Anatomy and Physiology Department, Kansas State University, Manhattan, United States; 3Developmental Neuroscience Section, National Institute on Deafness and Other Communication Disorders, National Institutes of Health, Bethesda, United States; 4Auditory Development and Restoration Program, National Institute on *For correspondence: Deafness and Other Communication Disorders, National Institutes of Health, [email protected] (PW); Bethesda, United States; 5Genomics and Computational Biology Core, National [email protected] (AJG) Institute on Deafness and Other Communication Disorders, National Institutes of Present address: Health, Bethesda, United States †Otolaryngology Department, Tsuchiura Kyodo General Hospital, Tsuchiura, Japan; ‡Department of Abstract Mutations of SLC26A4 are a common cause of hearing loss associated with Otorhinolaryngology, Head and enlargement of the endolymphatic sac (EES). Slc26a4 expression in the developing mouse Neck Surgery, Yonsei University College of Medicine, Seoul, endolymphatic sac is required for acquisition of normal inner ear structure and function. Here, we Korea; §Decibel Therapeutics, show that the mouse endolymphatic sac absorbs fluid in an SLC26A4-dependent fashion. Fluid Cambridge, United States; absorption was sensitive to ouabain and gadolinium but insensitive to benzamil, bafilomycin and #Technique R and D-Drug S3226. Single-cell RNA-seq analysis of pre- and postnatal endolymphatic sacs demonstrates two Substance, GlaxoSmithKline types of differentiated cells. -

Comprehensive Analysis of the Long Noncoding RNA HOXA11-AS Gene
Zhang et al. Cancer Cell Int (2016) 16:89 DOI 10.1186/s12935-016-0366-6 Cancer Cell International HYPOTHESIS Open Access Comprehensive analysis of the long noncoding RNA HOXA11‑AS gene interaction regulatory network in NSCLC cells Yu Zhang1†, Rong‑quan He2†, Yi‑wu Dang1, Xiu‑ling Zhang1, Xiao Wang3, Su‑ning Huang4, Wen‑ting Huang1, Meng‑tong Jiang1, Xiao‑ning Gan1, You Xie1, Ping Li1, Dian‑zhong Luo1, Gang Chen1*‡ and Ting‑qing Gan2*‡ Abstract Background: Long noncoding RNAs (lncRNAs) are related to different biological processes in non-small cell lung cancer (NSCLC). However, the possible molecular mechanisms underlying the effects of the long noncoding RNA HOXA11-AS (HOXA11 antisense RNA) in NSCLC are unknown. Methods: HOXA11-AS was knocked down in the NSCLC A549 cell line and a high throughput microarray assay was applied to detect changes in the gene profiles of the A549 cells. Bioinformatics analyses (gene ontology (GO), path‑ way, Kyoto Encyclopedia of Genes and Genomes (KEGG), and network analyses) were performed to investigate the potential pathways and networks of the differentially expressed genes. The molecular signatures database (MSigDB) was used to display the expression profiles of these differentially expressed genes. Furthermore, the relationships between the HOXA11-AS, de-regulated genes and clinical NSCLC parameters were verified by using NSCLC patient information from The Cancer Genome Atlas (TCGA) database. In addition, the relationship between HOXA11-AS expression and clinical diagnostic value was analyzed by receiver operating characteristic (ROC) curve. Results: Among the differentially expressed genes, 277 and 80 genes were upregulated and downregulated in NSCLC, respectively (fold change 2.0, P < 0.05 and false discovery rate (FDR) < 0.05). -

Original Article Increased Expression of TUSC3 in Non-Small Cell Lung Cancer
Int J Clin Exp Pathol 2016;9(5):5552-5559 www.ijcep.com /ISSN:1936-2625/IJCEP0023217 Original Article Increased expression of TUSC3 in non-small cell lung cancer Pengfei Ren1*, Jie Lin2*, Yuanyuan Wang3*, Kaican Cai1, Hua Wu1, Siyang Feng1 1Department of Thoracic Surgery, Nanfang Hospital, Southern Medical University, Guangzhou, PR China; 2Depart- ment of Pathology, Nanfang Hospital & School of Basic Medical Science, Southern Medical University, Guang- zhou, PR China; 3Department of Oncology, Nanfang Hospital, Southern Medical University, Guangzhou, PR China. *Equal contributors. Received January 4, 2016; Accepted March 18, 2016; Epub May 1, 2016; Published May 15, 2016 Abstract: Aims: To examine the expression of tumor suppressor candidate 3 (TUSC3) in non-small cell lung can- cer (NSCLC) compared to adjacent normal tissue, and its correlation with patients’ clinicopathological features. Methods: Forty NSCLC patients were recruited, including 30 patients with adenocarcinoma and 10 patients with squamous cell carcinoma. Expression of TUSC3 in tumor tissue and matched normal tissue was determined by real- time PCR and immunohistochemistry (IHC). The epidermal growth factor receptor (EGFR) mutation was detected by amplification refractory mutation system (ARMS)-Scorpions analysis. TUSC3 expression in several NSCLC cell lines was assayed by Western blotting. Results: Expression of TUSC3 is significantly higher in tumors than their ad- jacent normal tissues in 40 paired NSCLC specimens at both mRNA and protein level (P<0.05). More importantly, increased TUSC3 expression at the mRNA level correlated significantly with gender and age (P<0.05). However, no statistical significance was found between TUSC3 expression and smoking, T status, lymph node metastasis, differentiation and EGFR mutation status at neither mRNA nor protein level (P>0.05). -

Homozygous Single Base Deletion in TUSC3 Causes Intellectual Disability with Developmental Delay in an Omani Family
This is a repository copy of Homozygous single base deletion in TUSC3 causes intellectual disability with developmental delay in an Omani family. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/102499/ Version: Accepted Version Article: Al-Amri, A, Saegh, AA, Al-Mamari, W et al. (6 more authors) (2016) Homozygous single base deletion in TUSC3 causes intellectual disability with developmental delay in an Omani family. American Journal of Medical Genetics Part A, 170 (7). pp. 1826-1831. ISSN 1552-4825 https://doi.org/10.1002/ajmg.a.37690 © 2016, Wiley. This is the peer reviewed version of the following article: "Al-Amri A, Saegh AA, Al-Mamari W, El-Asrag ME, Ivorra JL, Cardno AG, Inglehearn CF, Clapcote SJ, Ali M. 2016. Homozygous single base deletion in TUSC3 causes intellectual disability with developmental delay in an Omani family. Am J Med Genet Part A 170A:1826–1831" which has been published in final form at http://dx.doi.org/10.1002/ajmg.a.37690. This article may be used for non-commercial purposes in accordance with Wiley Terms and Conditions for Self-Archiving. Uploaded in accordance with the publisher's self-archiving policy. Reuse Unless indicated otherwise, fulltext items are protected by copyright with all rights reserved. The copyright exception in section 29 of the Copyright, Designs and Patents Act 1988 allows the making of a single copy solely for the purpose of non-commercial research or private study within the limits of fair dealing. The publisher or other rights-holder may allow further reproduction and re-use of this version - refer to the White Rose Research Online record for this item. -

Defective Glycosylation and Multisystem Abnormalities Characterize the Primary Immunodeficiency XMEN Disease
Defective glycosylation and multisystem abnormalities characterize the primary immunodeficiency XMEN disease Juan C. Ravell, … , Matthias Mann, Michael J. Lenardo J Clin Invest. 2020;130(1):507-522. https://doi.org/10.1172/JCI131116. Research Article Immunology Graphical abstract Find the latest version: https://jci.me/131116/pdf The Journal of Clinical Investigation RESEARCH ARTICLE Defective glycosylation and multisystem abnormalities characterize the primary immunodeficiency XMEN disease Juan C. Ravell,1 Mami Matsuda-Lennikov,1 Samuel D. Chauvin,1 Juan Zou,1 Matthew Biancalana,1 Sally J. Deeb,2 Susan Price,3 Helen C. Su,3 Giulia Notarangelo,1 Ping Jiang,1 Aaron Morawski,1 Chrysi Kanellopoulou,1 Kyle Binder,1,4 Ratnadeep Mukherjee,5 James T. Anibal,5 Brian Sellers,6 Lixin Zheng,1 Tingyan He,1,7 Alex B. George,1 Stefania Pittaluga,8 Astin Powers,9 David E. Kleiner,9 Devika Kapuria,10 Marc Ghany,10 Sally Hunsberger,11 Jeffrey I. Cohen,12 Gulbu Uzel,3 Jenna Bergerson,3 Lynne Wolfe,13 Camilo Toro,13 William Gahl,13 Les R. Folio,14 Helen Matthews,1 Pam Angelus,3,15 Ivan K. Chinn,16 Jordan S. Orange,16 Claudia M. Trujillo-Vargas,17 Jose Luis Franco,17 Julio Orrego-Arango,17 Sebastian Gutiérrez-Hincapié,17 Niraj Chandrakant Patel,18,19 Kimiyo Raymond,20 Turkan Patiroglu,21 Ekrem Unal,21 Musa Karakukcu,21 Alexandre G.R. Day,22 Pankaj Mehta,22 Evan Masutani,1 Suk S. De Ravin,3 Harry L. Malech,3 Grégoire Altan-Bonnet,5 V. Koneti Rao,3 Matthias Mann,2 and Michael J. Lenardo1 1Molecular Development of the Immune System Section, Laboratory of Immune System Biology, and Clinical Genomics Program, Division of Intramural Research, National Institute of Allergy and Infectious Diseases (NIAID), Bethesda, Maryland, USA. -
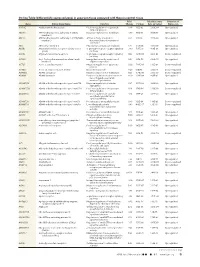
Differentially Expressed Genes in Aneurysm Tissue Compared With
On-line Table: Differentially expressed genes in aneurysm tissue compared with those in control tissue Fold False Discovery Direction of Gene Entrez Gene Name Function Change P Value Rate (q Value) Expression AADAC Arylacetamide deacetylase Positive regulation of triglyceride 4.46 1.33E-05 2.60E-04 Up-regulated catabolic process ABCA6 ATP-binding cassette, subfamily A (ABC1), Integral component of membrane 3.79 9.15E-14 8.88E-12 Up-regulated member 6 ABCC3 ATP-binding cassette, subfamily C (CFTR/MRP), ATPase activity, coupled to 6.63 1.21E-10 7.33E-09 Up-regulated member 3 transmembrane movement of substances ABI3 ABI family, member 3 Peptidyl-tyrosine phosphorylation 6.47 2.47E-05 4.56E-04 Up-regulated ACKR1 Atypical chemokine receptor 1 (Duffy blood G-protein–coupled receptor signaling 3.80 7.95E-10 4.18E-08 Up-regulated group) pathway ACKR2 Atypical chemokine receptor 2 G-protein–coupled receptor signaling 0.42 3.29E-04 4.41E-03 Down-regulated pathway ACSM1 Acyl-CoA synthetase medium-chain family Energy derivation by oxidation of 9.87 1.70E-08 6.52E-07 Up-regulated member 1 organic compounds ACTC1 Actin, ␣, cardiac muscle 1 Negative regulation of apoptotic 0.30 7.96E-06 1.65E-04 Down-regulated process ACTG2 Actin, ␥2, smooth muscle, enteric Blood microparticle 0.29 1.61E-16 2.36E-14 Down-regulated ADAM33 ADAM domain 33 Integral component of membrane 0.23 9.74E-09 3.95E-07 Down-regulated ADAM8 ADAM domain 8 Positive regulation of tumor necrosis 4.69 2.93E-04 4.01E-03 Up-regulated factor (ligand) superfamily member 11 production ADAMTS18 -

Loss of the Oligosaccharyl Transferase Subunit TUSC3 Promotes Proliferation and Migration of Ovarian Cancer Cells
INTERNATIONAL JOURNAL OF ONCOLOGY 42: 1383-1389, 2013 Loss of the oligosaccharyl transferase subunit TUSC3 promotes proliferation and migration of ovarian cancer cells PETR VAŇHARA1*, PETER HORAK2*, DIETMAR PILS3, MARIAM ANEES2, MICHAELA PETZ2, WOLFGANG GREGOR4, ROBERT ZEILLINGER3 and MICHAEL KRAINER2 1Department of Histology and Embryology, Faculty of Medicine, Masaryk University, Brno, Czech Republic; Departments of 2Internal Medicine I and Comprehensive Cancer Center and 3Obstetrics and Gynecology, Division of Gynecology, Medical University of Vienna; 4Research Group of Molecular Pharmacology and Toxicology, University of Veterinary Medicine Vienna, Vienna, Austria Received November 18, 2012; Accepted January 8, 2013 DOI: 10.3892/ijo.2013.1824 Abstract. Consequences of deregulated protein N-glycosylation detection and diagnosis of ovarian cancer presents one of the on cancer pathogenesis are poorly understood. TUSC3 is challenges of this entity and consequently a majority of cases a gene with a putative function in N-glycosylation, located are diagnosed in advanced stages (FIGO III and IV). The on the short arm of chromosome 8. This is a chromosomal prognosis for women with advanced ovarian cancer remains region of frequent genetic loss in ovarian cancer. We estab- bleak despite the advances of surgical and medical therapies lished recently that the expression of TUSC3 is epigenetically in the last decades. A better understanding of the pathogenesis decreased in epithelial ovarian cancer compared to benign of ovarian cancer may lead to earlier diagnosis and novel controls and provides prognostic information on patient therapies for this disease. survival. Therefore, we analyzed the consequences of silenced Tumor suppressor candidate 3 (TUSC3), originally named TUSC3 expression on proliferation, invasion and migration N33, was identified as a potential tumor suppressor gene in of ovarian cell lines. -

Supplemental Data
Supplementary Table 1. Gene sets from Figure 6. Lists of genes from each individual gene set defined in Figure 6, including the fold-change in expression of each gene in treatment group pair-wise comparisons. ENSEMBL: Ensembl gene identifier; Symbol: official gene symbol; logFC: log fold change; p value: significance of fold-change in a pair-wise comparison, P<0.05 cut-off; FDR: false discovery rate, expected proportion of false positives among the differentially expressed genes in a pair-wise comparison (FDR<0.25 cut-off). Sup. Table 1 SET I CP versus Sal CP versus CP+DCA DCA versus Sal ENSEMBL Symbol logFC PValue FDR logFC PValue FDR logFC PValue FDR Desc ENSMUSG00000020326 Ccng1 2.64 0.00 0.00 -0.06 0.13 0.96 0.40 0.00 0.23 cyclin G1 [Source:MGI Symbol;Acc:MGI:102890] ENSMUSG00000031886 Ces2e 3.97 0.00 0.00 -0.24 0.02 0.28 0.01 1.00 1.00 carboxylesterase 2E [Source:MGI Symbol;Acc:MGI:2443170] ENSMUSG00000041959 S100a10 2.31 0.00 0.00 -0.21 0.02 0.23 -0.11 0.53 1.00 S100 calcium binding protein A10 (calpactin) [Source:MGI Symbol;Acc:MGI:1339468] ENSMUSG00000092341 Malat1 1.09 0.00 0.00 -0.11 0.20 1.00 0.66 0.00 0.00 metastasis associated lung adenocarcinoma transcript 1 (non-coding RNA) [Source:MGI Symbol;Acc:MGI:1919539] ENSMUSG00000072949 Acot1 1.73 0.00 0.00 -0.22 0.01 0.12 -0.44 0.01 1.00 acyl-CoA thioesterase 1 [Source:MGI Symbol;Acc:MGI:1349396] ENSMUSG00000064339 mt-Rnr2 1.09 0.00 0.00 -0.08 0.17 1.00 0.67 0.00 0.07 mitochondrially encoded 16S rRNA [Source:MGI Symbol;Acc:MGI:102492] ENSMUSG00000025934 Gsta3 1.86 0.00 0.00 -0.28