Algae-Bacteria Interactions During Commercial Production of Nannochloropsis Sp. in Closed Photobioreactors
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genomic Insights from the Oleaginous Model Alga Nannochloropsis Gaditana
ADDENDUM ADDENDUM Bioengineered 4:1, 37–43; January/February 2013; © 2013 Landes Bioscience Genomic insights from the oleaginous model alga Nannochloropsis gaditana Robert E. Jinkerson, Randor Radakovits† and Matthew C. Posewitz* Department of Chemistry and Geochemistry; Colorado School of Mines; Golden, CO USA †Current Affiliation: Synthetic Genomics, La Jolla, CA 92037 USA annochloropsis species have economically viable biofuel production Nemerged as leading phototrophic processes.1-3 A promising strategy for microorganisms for the production of bio- increasing productivities is to “domesti- fuels. Several isolates produce large quan- cate” algal strains via genetic engineering tities of triacylglycerols, grow rapidly, to improve lipid yields and to produce and can be cultivated at industrial scales. biomolecules tailored for biofuel appli- Recently, the mitochondrial, plastid and cations.4 Currently, only a few algal sys- nuclear genomes of Nannochloropsis gadi- tems with publicly available genomes tana were sequenced. Genomic interro- are genetically tractable, including the gation revealed several key features that green algae Chlamydomonas reinhardtii likely facilitate the oleaginous phenotype and Ostreococcus tauri,5 as well as the observed in Nannochloropsis, including diatoms Phaeodactylum tricornutum6 and an over-representation of genes involved Thalassiosira pseudonana.7 These organ- in lipid biosynthesis. Here we present isms were selected as model systems additional analyses on gene orientation, because of their phylogenetic -

Which Organisms Are Used for Anti-Biofouling Studies
Table S1. Semi-systematic review raw data answering: Which organisms are used for anti-biofouling studies? Antifoulant Method Organism(s) Model Bacteria Type of Biofilm Source (Y if mentioned) Detection Method composite membranes E. coli ATCC25922 Y LIVE/DEAD baclight [1] stain S. aureus ATCC255923 composite membranes E. coli ATCC25922 Y colony counting [2] S. aureus RSKK 1009 graphene oxide Saccharomycetes colony counting [3] methyl p-hydroxybenzoate L. monocytogenes [4] potassium sorbate P. putida Y. enterocolitica A. hydrophila composite membranes E. coli Y FESEM [5] (unspecified/unique sample type) S. aureus (unspecified/unique sample type) K. pneumonia ATCC13883 P. aeruginosa BAA-1744 composite membranes E. coli Y SEM [6] (unspecified/unique sample type) S. aureus (unspecified/unique sample type) graphene oxide E. coli ATCC25922 Y colony counting [7] S. aureus ATCC9144 P. aeruginosa ATCCPAO1 composite membranes E. coli Y measuring flux [8] (unspecified/unique sample type) graphene oxide E. coli Y colony counting [9] (unspecified/unique SEM sample type) LIVE/DEAD baclight S. aureus stain (unspecified/unique sample type) modified membrane P. aeruginosa P60 Y DAPI [10] Bacillus sp. G-84 LIVE/DEAD baclight stain bacteriophages E. coli (K12) Y measuring flux [11] ATCC11303-B4 quorum quenching P. aeruginosa KCTC LIVE/DEAD baclight [12] 2513 stain modified membrane E. coli colony counting [13] (unspecified/unique colony counting sample type) measuring flux S. aureus (unspecified/unique sample type) modified membrane E. coli BW26437 Y measuring flux [14] graphene oxide Klebsiella colony counting [15] (unspecified/unique sample type) P. aeruginosa (unspecified/unique sample type) graphene oxide P. aeruginosa measuring flux [16] (unspecified/unique sample type) composite membranes E. -

Extraction of the Protein Content from the Green Microalgae
Extraction of the protein content from the green microalgae Nannochloropsis gaditana and Neochloris oleoabundans Studies on high-pressure homogenization, centrifugation and ultrafiltration Thesis to obtain the Masters Science degree in Biological Engineering Pedro Miguel Oliveira Grilo E X E C U T I V E S U M M A R Y Info A B S T R A C T Experimental work done This project was conducted with the objective of extracting the protein content, free from chlorophyll, from the green microalgae in the Wageningen UR species Nannochloropsis gaditana and Neochloris oleoabundans. The combination of the high-pressure homogenization (HPH) with an Food & Biobased Center alkaline pH adjustment of the microalgae suspension was tried in order to increase the protein release. Although the pH adjustment (FBR), from 02/2017 until increased the permeability of intact cells, the results in combination with the homogenization were not satisfactory. The centrifugation 08/2017, under the supervision of Dr. Carl parameters were optimized, leading to concentration ratios in the supernatant of 42.2% and 62.4%/initial concentration for N. gaditana Safi and Dr. Ana and N. oleoabundans and yields of 31.0% and 49.4%(w/w initial protein) for the respective species. The flocculation of the cell debris by Azevedo. pH variations was studied to improve the centrifugations. More supernatant was recovered at acidic pH, but many proteins also precipitated at these pH values, and so the yields after centrifugation were lower than at native pH for both species. Next, it was Keywords: microalgae, discovered that by doing the centrifugations at lower biomass concentrations, for example, with 50 g/l instead of 100 g/l, the yield cell disruption, protein, increases 5.5%(w/w initial protein) on N. -

Batch and Fed Batch Cultivation and Harvesting of Nannochloropsis Gaditana for Environmental Applications
Western University Scholarship@Western Electronic Thesis and Dissertation Repository 7-11-2017 12:00 AM Batch and Fed Batch Cultivation and Harvesting of Nannochloropsis Gaditana for Environmental Applications Roopa P. Devasya The University of Western Ontario Supervisor Dr Amarjeet Bassi The University of Western Ontario Graduate Program in Chemical and Biochemical Engineering A thesis submitted in partial fulfillment of the equirr ements for the degree in Doctor of Philosophy © Roopa P. Devasya 2017 Follow this and additional works at: https://ir.lib.uwo.ca/etd Part of the Chemical Engineering Commons Recommended Citation Devasya, Roopa P., "Batch and Fed Batch Cultivation and Harvesting of Nannochloropsis Gaditana for Environmental Applications" (2017). Electronic Thesis and Dissertation Repository. 4670. https://ir.lib.uwo.ca/etd/4670 This Dissertation/Thesis is brought to you for free and open access by Scholarship@Western. It has been accepted for inclusion in Electronic Thesis and Dissertation Repository by an authorized administrator of Scholarship@Western. For more information, please contact [email protected]. Abstract The exhaustion of fossil fuels and climate change are two significant issues of our times. Microalgae are eukaryotic phototrophs or prokaryotic cyanobacteria (blue green algae), which are able to capture CO2, and thus, can mitigate these challenges. In addition, some microalgae can produce lipids suitable for biodiesel. The potential application of microalgae biomass for biofuel production is a clean and sustainable approach to replace fossil fuels. An important consideration for reducing the cost of biofuel is the economical production of algal biomass with high lipid yields. In this study, the marine microalgae Nannochloropsis gaditana were investigated for biomass and lipid production using two wastewater streams, i.e., road salt run-off and vegetable greenhouse industry effluents as the growth media using batch or fed-batch cultivation. -

2016 National Algal Biofuels Technology Review
National Algal Biofuels Technology Review Bioenergy Technologies Office June 2016 National Algal Biofuels Technology Review U.S. Department of Energy Office of Energy Efficiency and Renewable Energy Bioenergy Technologies Office June 2016 Review Editors: Amanda Barry,1,5 Alexis Wolfe,2 Christine English,3,5 Colleen Ruddick,4 and Devinn Lambert5 2010 National Algal Biofuels Technology Roadmap: eere.energy.gov/bioenergy/pdfs/algal_biofuels_roadmap.pdf A complete list of roadmap and review contributors is available in the appendix. Suggested Citation for this Review: DOE (U.S. Department of Energy). 2016. National Algal Biofuels Technology Review. U.S. Department of Energy, Office of Energy Efficiency and Renewable Energy, Bioenergy Technologies Office. Visit bioenergy.energy.gov for more information. 1 Los Alamos National Laboratory 2 Oak Ridge Institute for Science and Education 3 National Renewable Energy Laboratory 4 BCS, Incorporated 5 Bioenergy Technologies Office This report is being disseminated by the U.S. Department of Energy. As such, the document was prepared in compliance with Section 515 of the Treasury and General Government Appropriations Act for Fiscal Year 2001 (Public Law No. 106-554) and information quality guidelines issued by the Department of Energy. Further, this report could be “influential scientific information” as that term is defined in the Office of Management and Budget’s Information Quality Bulletin for Peer Review (Bulletin). This report has been peer reviewed pursuant to section II.2 of the Bulletin. Cover photo courtesy of Qualitas Health, Inc. BIOENERGY TECHNOLOGIES OFFICE Preface Thank you for your interest in the U.S. Department of Energy (DOE) Bioenergy Technologies Office’s (BETO’s) National Algal Biofuels Technology Review. -

Toward the Enhancement of Microalgal Metabolite Production Through Microalgae–Bacteria Consortia †
biology Review Toward the Enhancement of Microalgal Metabolite Production through Microalgae–Bacteria Consortia † Lina Maria González-González 1 and Luz E. de-Bashan 1,2,3,* 1 The Bashan Institute of Science, 1730 Post Oak Ct, Auburn, AL 36830, USA; [email protected] 2 Environmental Microbiology Group, Northwestern Center for Biological Research (CIBNOR), Avenida IPN 195, La Paz, Baja California Sur 23096, Mexico 3 Department of Entomology and Plant Pathology, Auburn University, 209 Life Sciences Building, Auburn, AL 36849, USA * Correspondence: [email protected] † Dedicated to the memory of Prof. Yoav Bashan, founder of the Bashan Institute of Science and leader of the Environmental Microbiology Group at CIBNOR for 28 years. Simple Summary: Microalgae are photosynthetic microorganisms with high biotechnological po- tential. However, the sustainable production of high-value products such as lipids, proteins, carbo- hydrates, and pigments undergoes important economic challenges. In this review, we describe the mutualistic association between microalgae and bacteria and the positive effects of artificial consortia on microalgal metabolites’ production. We highlighted the potential role of growth-promoting bacte- ria in optimizing microalgal biorefineries for the integrated production of these valuable products. Besides making a significant enhancement to microalgal metabolite production, the bacterium partner might assist in the biorefinery process’s key stages, such as biomass harvesting and CO2 fixation. Citation: González-González, L.M.; de-Bashan, L.E. Toward the Abstract: Engineered mutualistic consortia of microalgae and bacteria may be a means of assembling Enhancement of Microalgal a novel combination of metabolic capabilities with potential biotechnological advantages. Microalgae Metabolite Production through Microalgae–Bacteria Consortia. -

Algal-Bacteria Consortia
DOE Bioengery Technologies Office (BETO) 2019 Project Peer Review Success through Synergy: Increasing Cultivation Yield and Stability with Rationally Designed Consortia March 6, 2019 Advanced Algal Systems Shawn Starkenburg, Los Alamos National Laboratory (LANL) Alina Corcoran, New Mexico Consortium (New Mexico Consortium) This presentation does not contain any proprietary, confidential, or otherwise restricted information. 2 of 30 Goal Statement Algae Bacteria-algae consortia consortia Project Goal: to increase productivity of open, outdoor Nannochloropsis cultures from 7 g/m2/d to HIGH-THROUGHPUT 2 >14 g/m /d (doubling of fall State of Technology SCREENING value) via development of (1) rationally-designed intrageneric Nannochloropsis consortia & (2) Nannochloropsis-bacteria consortia Complex Outputs: molecular toolkits, pipelines, consortia consortia Outcomes: increased productivity & stability BIOREACTOR TRIALS Industry Relevance: increased yield, fewer crop losses, & enhanced economic feasibility FIELD TRIALS 3 of 30 Quad Chart Timeline Barriers Addressed start date -- February 1, 2018 ➢ Biomass Genetics & Development (Aft-C); to improve the end date -- January 31, 2021 productivity and robustness of 30% complete (with verification) algae strains against perturbations FY FY 18 Total Objective 17 Costs Planned to rationally design intrageneric Costs Funding Nannochloropsis consortia & (FY 19-21) Nannochloropsis-bacteria DOE $0 LANL: LANL: consortia to increase productivity, Funded $351 K $1.19 M stability and yield of open, NMC: -

Microbial Utilization of Aqueous Co-Products from Hydrothermal Liquefaction of Microalgae Nannochloropsis Oculata
Bioresource Technology 136 (2013) 522–528 Contents lists available at SciVerse ScienceDirect Biore source Tec hnology journal homepage: www.elsevier.com/locate/biortech Microbial utilization of aqueous co-products from hydrothermal liquefaction of microalgae Nannochloropsis oculata Michael Nelson a, Lian Zhu a, Anne Thiel a, Yan Wu a, Mary Guan b, Jeremy Minty a, Henry Y. Wang a, ⇑ Xiaoxia Nina Lin a,c,d, a Department of Chemical Engineering, University of Michigan, USA b Department of Chemical Engineering, Massachusetts Institute of Technology, USA c Department of Biomedical Engineering, University of Michigan, USA d Center for Computational Medicine and Bioinformatics, University of Michigan, USA highlights Aqueous co-product from mic roalgae liquefaction was used for microbial growth. Yield, growth rate, and carbon usage data were generated for two model bacteria. A mic robial side-culture may increase efficiency of a microalgae biofuel operation . article info abstract Article history: Hydrot hermal liquefaction of algae biomass is a promising technology for the production of sustainable Received 10 November 2012 biofuels, but the non-oil, aqueous co-product of the process has only been examined to a limited extent. Received in revised form 8 March 2013 The aqueous phase from liquefaction of the alga Nannochloropsis oculata (AqAl) was used to make growth Accepted 9 March 2013 media for model heterotrophic microorganisms Escherichia coli , Pseudomonas putida , and Saccharom yces Available online 18 March 2013 cerevisiae. Growth rates, yields, and carbon/nitrogen/phosphorus uptake were measured. E. coli and P. putida could grow using AqAl as the sole C, N, and P source in media containing 10 vol.%–40 vol.% AqAl Keywords: with the best growth occurring at 20 vol.%. -

DEVELOPMENT of CRISPR/CAS9 in NANNOCHLOROPSIS and OTHER ALGAE TOWARD UNDERSTANDING and MANIPULATING ENERGY ALLOCATION by Brian V
DEVELOPMENT OF CRISPR/CAS9 IN NANNOCHLOROPSIS AND OTHER ALGAE TOWARD UNDERSTANDING AND MANIPULATING ENERGY ALLOCATION by Brian Vogler A thesis submitted to the Faculty and the Board of Trustees of the Colorado School of Mines in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Applied Chemistry). Golden, Colorado Date _______________________ Signed: ___________________________ Brian W. Vogler Signed: ___________________________ Dr. Matthew C. Posewitz Thesis Advisor Golden, Colorado Date _______________________ Signed: ___________________________ Dr. Thomas Gennett Department Head Department of Chemistry ii ABSTRACT Nannochloropsis is a genus of eukaryotic microalgae that grows well in outdoor bioreactors and produces high yields of triacylglycerols (TAGs), which can be processed into biodiesel. In this work, we chemically characterize the storage carbohydrate of Nannochloropsis for the first time, and interrupt enzymes required for its biosynthesis to (1) understand their function and (2) interrogate whether this unused biomass component can be eliminated without significant impact on viability. To generate targeted gene knockouts, we developed CRISPR/Cas9 methods for Nannochloropsis and first interrupted nitrate reductase, a common target for genetic knockout because it is both inessential and easily scored. The method and validated chassis strain was then used to interrogate the beta-glucan synthase (BGS) and transglycosylase (TGS) enzymes believed to be responsible for the backbone polymerization and branching of the storage beta glucan, respectively. We identified no significant growth defects in our laboratory culturing conditions but did confirm that both genes were fundamental to synthesis of this beta glucan storage carbohydrate. The generated knockouts of either gene do not produce the elevated carbohydrate phenotype of wild-type cells in response to nitrogen deprivation. -

Variation in Sodic Soil Bacterial Communities Associated with Different Alkali Vegetation Types
microorganisms Article Variation in Sodic Soil Bacterial Communities Associated with Different Alkali Vegetation Types Andrea K. Borsodi 1,2,*, Márton Mucsi 1,3, Gergely Krett 1, Attila Szabó 2, Tamás Felföldi 1,2 and Tibor Szili-Kovács 3,* 1 Department of Microbiology, ELTE Eötvös Loránd University, Pázmány P. Sétány 1/C, H-1117 Budapest, Hungary; [email protected] (M.M.); [email protected] (G.K.); [email protected] (T.F.) 2 Institute of Aquatic Ecology, Centre for Ecological Research, Karolina út 29, H-1113 Budapest, Hungary; [email protected] 3 Institute for Soil Sciences, Centre for Agricultural Research, Herman Ottó út 15, H-1022 Budapest, Hungary * Correspondence: [email protected] (A.K.B.); [email protected] (T.S.-K.); Tel.: +36-13812177 (A.K.B.); +36-309617452 (T.S.-K.) Abstract: In this study, we examined the effect of salinity and alkalinity on the metabolic potential and taxonomic composition of microbiota inhabiting the sodic soils in different plant communities. The soil samples were collected in the Pannonian steppe (Hungary, Central Europe) under extreme dry and wet weather conditions. The metabolic profiles of microorganisms were analyzed using the MicroResp method, the bacterial diversity was assessed by cultivation and next-generation amplicon sequencing based on the 16S rRNA gene. Catabolic profiles of microbial communities varied primarily according to the alkali vegetation types. Most members of the strain collection were identified as plant associated and halophilic/alkaliphilic species of Micrococcus, Nesterenkonia, Citation: Borsodi, A.K.; Mucsi, M.; Nocardiopsis, Streptomyces (Actinobacteria) and Bacillus, Paenibacillus (Firmicutes) genera. -
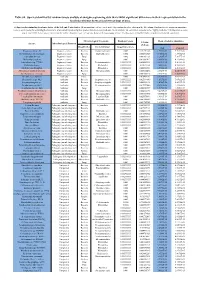
Table S8. Species Identified by Random Forests Analysis of Shotgun Sequencing Data That Exhibit Significant Differences In
Table S8. Species identified by random forests analysis of shotgun sequencing data that exhibit significant differences in their representation in the fecal microbiomes between each two groups of mice. (a) Species discriminating fecal microbiota of the Soil and Control mice. Mean importance of species identified by random forest are shown in the 5th column. Random forests assigns an importance score to each species by estimating the increase in error caused by removing that species from the set of predictors. In our analysis, we considered a species to be “highly predictive” if its importance score was at least 0.001. T-test was performed for the relative abundances of each species between the two groups of mice. P-values were at least 0.05 to be considered statistically significant. Microbiological Taxonomy Random Forests Mean of relative abundance P-Value Species Microbiological Function (T-Test) Classification Bacterial Order Importance Score Soil Control Rhodococcus sp. 2G Engineered strain Bacteria Corynebacteriales 0.002 5.73791E-05 1.9325E-05 9.3737E-06 Herminiimonas arsenitoxidans Engineered strain Bacteria Burkholderiales 0.002 0.005112829 7.1580E-05 1.3995E-05 Aspergillus ibericus Engineered strain Fungi 0.002 0.001061181 9.2368E-05 7.3057E-05 Dichomitus squalens Engineered strain Fungi 0.002 0.018887472 8.0887E-05 4.1254E-05 Acinetobacter sp. TTH0-4 Engineered strain Bacteria Pseudomonadales 0.001333333 0.025523638 2.2311E-05 8.2612E-06 Rhizobium tropici Engineered strain Bacteria Rhizobiales 0.001333333 0.02079554 7.0081E-05 4.2000E-05 Methylocystis bryophila Engineered strain Bacteria Rhizobiales 0.001333333 0.006513543 3.5401E-05 2.2044E-05 Alteromonas naphthalenivorans Engineered strain Bacteria Alteromonadales 0.001 0.000660472 2.0747E-05 4.6463E-05 Saccharomyces cerevisiae Engineered strain Fungi 0.001 0.002980726 3.9901E-05 7.3043E-05 Bacillus phage Belinda Antibiotic Phage 0.002 0.016409765 6.8789E-07 6.0681E-08 Streptomyces sp. -

Thermophysical Properties of the Marine Microalgae Nannochloropsis Salina☆
Fuel Processing Technology 152 (2016) 390–398 Contents lists available at ScienceDirect Fuel Processing Technology journal homepage: www.elsevier.com/locate/fuproc Thermophysical properties of the marine microalgae Nannochloropsis salina☆ Nico Schneider a,⁎,TaraJ.Fortinb, Roland Span a,MandyGerberc a Ruhr-Universität Bochum, Lehrstuhl für Thermodynamik, Universitätsstr. 150, 44801 Bochum, Germany b National Institute of Standards and Technology, Applied Chemicals and Materials Division, 325 Broadway MS 647.07, Boulder, CO 80305, USA c Hochschule Bochum, Institut für Thermo- und Fluiddynamik, Lennershofstr. 140, 44801 Bochum, Germany article info abstract Article history: Algae have a variety of uses, including pharmaceutical and cosmetic products, and as a potential energy source as Received 24 March 2016 a feedstock for biodiesel, bioethanol and biogas. To optimize the commercial production and use of algal biomass, Received in revised form 24 June 2016 production facilities have to be specifically designed to handle the source materials and subsequent products. Accepted 28 June 2016 This requires a knowledge of their thermophysical properties. To that end, density, viscosity, and heat capacity Available online 15 July 2016 ofthemarinemicroalgaeNannochloropsis salina were determined experimentally at atmospheric pressure and as a function of temperature and total solid (TS) content. Correlation analyses show a relationship between ex- Keywords: Microalgae perimental data, temperature, and total solid content for each of the investigated