Unravelling the Molecular Mechanism of Sorcin (Soluble
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Annexin A7 Is Required for ESCRT III-Mediated Plasma Membrane Repair
Annexin A7 is required for ESCRT III-mediated plasma membrane repair Sønder, Stine Lauritzen; Boye, Theresa Louise; Tölle, Regine; Dengjel, Jörn; Maeda, Kenji; Jäättelä, Marja; Simonsen, Adam Cohen; Jaiswal, Jyoti K.; Nylandsted, Jesper Published in: Scientific Reports DOI: 10.1038/s41598-019-43143-4 Publication date: 2019 Document version Publisher's PDF, also known as Version of record Document license: CC BY Citation for published version (APA): Sønder, S. L., Boye, T. L., Tölle, R., Dengjel, J., Maeda, K., Jäättelä, M., ... Nylandsted, J. (2019). Annexin A7 is required for ESCRT III-mediated plasma membrane repair. Scientific Reports, 9(1), [6726]. https://doi.org/10.1038/s41598-019-43143-4 Download date: 09. apr.. 2020 www.nature.com/scientificreports OPEN Annexin A7 is required for ESCRT III-mediated plasma membrane repair Received: 16 November 2018 Stine Lauritzen Sønder1, Theresa Louise Boye1, Regine Tölle2,3, Jörn Dengjel 2,3, Accepted: 15 April 2019 Kenji Maeda1, Marja Jäättelä 1,4, Adam Cohen Simonsen 5, Jyoti K. Jaiswal 6,7 & Published: xx xx xxxx Jesper Nylandsted 1,4 The plasma membrane of eukaryotic cells forms the essential barrier to the extracellular environment, and thus plasma membrane disruptions pose a fatal threat to cells. Here, using invasive breast cancer cells we show that the Ca2+ - and phospholipid-binding protein annexin A7 is part of the plasma membrane repair response by enabling assembly of the endosomal sorting complex required for transport (ESCRT) III. Following injury to the plasma membrane and Ca2+ fux into the cytoplasm, annexin A7 forms a complex with apoptosis linked gene-2 (ALG-2) to facilitate proper recruitment and binding of ALG-2 and ALG-2-interacting protein X (ALIX) to the damaged membrane. -

Farnesol-Induced Apoptosis in Human Lung Carcinoma Cells Is Coupled to the Endoplasmic Reticulum Stress Response
Research Article Farnesol-Induced Apoptosis in Human Lung Carcinoma Cells Is Coupled to the Endoplasmic Reticulum Stress Response Joung Hyuck Joo,1 Grace Liao,1 Jennifer B. Collins,2 Sherry F. Grissom,2 and Anton M. Jetten1 1Cell Biology Section, LRB, and 2Microarray Group, Division of Intramural Research, National Institute of Environmental Health Sciences, NIH, Research Triangle Park, North Carolina Abstract range of fruits and vegetables (9, 10). Each isoprenoid has been Farnesol (FOH) and other isoprenoid alcohols induce apopto- shown to inhibit proliferation and induce apoptosis in a number of sis in various carcinoma cells and inhibit tumorigenesis in neoplastic cell lines from different origins (4, 11–14). In addition, in vivo these isoprenoids have been reported to be effective in chemo- several models. However, the mechanisms by which in vivo they mediate their effects are not yet fully understood. In this prevention and chemotherapy in various cancer models study, we show that FOH is an effective inducer of apoptosis in (10, 12, 15, 16). FOH has been reported to exhibit chemopreventive several lung carcinoma cells, including H460. This induction is effects in colon and pancreas carcinogenesis in rats (9, 17) whereas associated with activation of several caspases and cleavage of phase I and II clinical trials have indicated therapeutic potential poly(ADP-ribose) polymerase (PARP). To obtain insight into for POH (16, 18). The mechanisms by which these isoprenoids induce these effects are not yet fully understood. Isoprenoids have the mechanism involved in FOH-induced apoptosis, we compared the gene expression profiles of FOH-treated and been reported to inhibit posttranslational protein prenylation (19) control H460 cells by microarray analysis. -

Multi-Targeted Mechanisms Underlying the Endothelial Protective Effects of the Diabetic-Safe Sweetener Erythritol
Multi-Targeted Mechanisms Underlying the Endothelial Protective Effects of the Diabetic-Safe Sweetener Erythritol Danie¨lle M. P. H. J. Boesten1*., Alvin Berger2.¤, Peter de Cock3, Hua Dong4, Bruce D. Hammock4, Gertjan J. M. den Hartog1, Aalt Bast1 1 Department of Toxicology, Maastricht University, Maastricht, The Netherlands, 2 Global Food Research, Cargill, Wayzata, Minnesota, United States of America, 3 Cargill RandD Center Europe, Vilvoorde, Belgium, 4 Department of Entomology and UCD Comprehensive Cancer Center, University of California Davis, Davis, California, United States of America Abstract Diabetes is characterized by hyperglycemia and development of vascular pathology. Endothelial cell dysfunction is a starting point for pathogenesis of vascular complications in diabetes. We previously showed the polyol erythritol to be a hydroxyl radical scavenger preventing endothelial cell dysfunction onset in diabetic rats. To unravel mechanisms, other than scavenging of radicals, by which erythritol mediates this protective effect, we evaluated effects of erythritol in endothelial cells exposed to normal (7 mM) and high glucose (30 mM) or diabetic stressors (e.g. SIN-1) using targeted and transcriptomic approaches. This study demonstrates that erythritol (i.e. under non-diabetic conditions) has minimal effects on endothelial cells. However, under hyperglycemic conditions erythritol protected endothelial cells against cell death induced by diabetic stressors (i.e. high glucose and peroxynitrite). Also a number of harmful effects caused by high glucose, e.g. increased nitric oxide release, are reversed. Additionally, total transcriptome analysis indicated that biological processes which are differentially regulated due to high glucose are corrected by erythritol. We conclude that erythritol protects endothelial cells during high glucose conditions via effects on multiple targets. -

1 Supporting Information for a Microrna Network Regulates
Supporting Information for A microRNA Network Regulates Expression and Biosynthesis of CFTR and CFTR-ΔF508 Shyam Ramachandrana,b, Philip H. Karpc, Peng Jiangc, Lynda S. Ostedgaardc, Amy E. Walza, John T. Fishere, Shaf Keshavjeeh, Kim A. Lennoxi, Ashley M. Jacobii, Scott D. Rosei, Mark A. Behlkei, Michael J. Welshb,c,d,g, Yi Xingb,c,f, Paul B. McCray Jr.a,b,c Author Affiliations: Department of Pediatricsa, Interdisciplinary Program in Geneticsb, Departments of Internal Medicinec, Molecular Physiology and Biophysicsd, Anatomy and Cell Biologye, Biomedical Engineeringf, Howard Hughes Medical Instituteg, Carver College of Medicine, University of Iowa, Iowa City, IA-52242 Division of Thoracic Surgeryh, Toronto General Hospital, University Health Network, University of Toronto, Toronto, Canada-M5G 2C4 Integrated DNA Technologiesi, Coralville, IA-52241 To whom correspondence should be addressed: Email: [email protected] (M.J.W.); yi- [email protected] (Y.X.); Email: [email protected] (P.B.M.) This PDF file includes: Materials and Methods References Fig. S1. miR-138 regulates SIN3A in a dose-dependent and site-specific manner. Fig. S2. miR-138 regulates endogenous SIN3A protein expression. Fig. S3. miR-138 regulates endogenous CFTR protein expression in Calu-3 cells. Fig. S4. miR-138 regulates endogenous CFTR protein expression in primary human airway epithelia. Fig. S5. miR-138 regulates CFTR expression in HeLa cells. Fig. S6. miR-138 regulates CFTR expression in HEK293T cells. Fig. S7. HeLa cells exhibit CFTR channel activity. Fig. S8. miR-138 improves CFTR processing. Fig. S9. miR-138 improves CFTR-ΔF508 processing. Fig. S10. SIN3A inhibition yields partial rescue of Cl- transport in CF epithelia. -

Supplemental Information
Supplemental information Dissection of the genomic structure of the miR-183/96/182 gene. Previously, we showed that the miR-183/96/182 cluster is an intergenic miRNA cluster, located in a ~60-kb interval between the genes encoding nuclear respiratory factor-1 (Nrf1) and ubiquitin-conjugating enzyme E2H (Ube2h) on mouse chr6qA3.3 (1). To start to uncover the genomic structure of the miR- 183/96/182 gene, we first studied genomic features around miR-183/96/182 in the UCSC genome browser (http://genome.UCSC.edu/), and identified two CpG islands 3.4-6.5 kb 5’ of pre-miR-183, the most 5’ miRNA of the cluster (Fig. 1A; Fig. S1 and Seq. S1). A cDNA clone, AK044220, located at 3.2-4.6 kb 5’ to pre-miR-183, encompasses the second CpG island (Fig. 1A; Fig. S1). We hypothesized that this cDNA clone was derived from 5’ exon(s) of the primary transcript of the miR-183/96/182 gene, as CpG islands are often associated with promoters (2). Supporting this hypothesis, multiple expressed sequences detected by gene-trap clones, including clone D016D06 (3, 4), were co-localized with the cDNA clone AK044220 (Fig. 1A; Fig. S1). Clone D016D06, deposited by the German GeneTrap Consortium (GGTC) (http://tikus.gsf.de) (3, 4), was derived from insertion of a retroviral construct, rFlpROSAβgeo in 129S2 ES cells (Fig. 1A and C). The rFlpROSAβgeo construct carries a promoterless reporter gene, the β−geo cassette - an in-frame fusion of the β-galactosidase and neomycin resistance (Neor) gene (5), with a splicing acceptor (SA) immediately upstream, and a polyA signal downstream of the β−geo cassette (Fig. -

Downloaded from URL: Δ Δ Nated ALG-2 GF122) and GST-ALG-2 GF122 Was Cium.Uhnres.Utoronto.Ca/Vgm
Inuzuka et al. BMC Structural Biology 2010, 10:25 http://www.biomedcentral.com/1472-6807/10/25 RESEARCH ARTICLE Open Access Molecular basis for defect in Alix-binding by alternatively spliced isoform of ALG-2 (ALG-2ΔGF122) and structural roles of F122 in target recognition Tatsutoshi Inuzuka1, Hironori Suzuki1,2, Masato Kawasaki2, Hideki Shibata1, Soichi Wakatsuki2, Masatoshi Maki1* Abstract Background: ALG-2 (a gene product of PDCD6) belongs to the penta-EF-hand (PEF) protein family and Ca2 +-dependently interacts with various intracellular proteins including mammalian Alix, an adaptor protein in the ESCRT system. Our previous X-ray crystal structural analyses revealed that binding of Ca2+ to EF3 enables the side chain of R125 to move enough to make a primary hydrophobic pocket (Pocket 1) accessible to a short fragment of Alix. The side chain of F122, facing a secondary hydrophobic pocket (Pocket 2), interacts with the Alix peptide. An alternatively spliced shorter isoform, designated ALG-2ΔGF122, lacks Gly121Phe122 and does not bind Alix, but the structural basis of the incompetence has remained to be elucidated. Results: We solved the X-ray crystal structure of the PEF domain of ALG-2ΔGF122 in the Ca2+-bound form and compared it with that of ALG-2. Deletion of the two residues shortened a-helix 5 (a5) and changed the configuration of the R125 side chain so that it partially blocked Pocket 1. A wall created by the main chain of 121- GFG-123 and facing the two pockets was destroyed. Surprisingly, however, substitution of F122 with Ala or Gly, but not with Trp, increased the Alix-binding capacity in binding assays. -

PDCD6 Antibody Order 021-34695924 [email protected] Support 400-6123-828 50Ul [email protected] 100 Ul √ √ Web
TD7978 PDCD6 Antibody Order 021-34695924 [email protected] Support 400-6123-828 50ul [email protected] 100 uL √ √ Web www.ab-mart.com.cn Description: Calcium sensor that plays a key role in processes such as endoplasmic reticulum (ER)- Golgi vesicular transport, endosomal biogenesis or membrane repair. Acts as an adapter that bridges unrelated proteins or stabilizes weak protein-protein complexes in response to calcium: calcium-binding triggers exposure of apolar surface, promoting interaction with different sets of proteins thanks to 3 different hydrophobic pockets, leading to translocation to membranes. Involved in ER-Golgi transport by promoting the association between PDCD6IP and TSG101, thereby bridging together the ESCRT-III and ESCRT-I complexes. Together with PEF1, acts as calcium-dependent adapter for the BCR(KLHL12) complex, a complex involved in ER-Golgi transport by regulating the size of COPII coats. In response to cytosolic calcium increase, the heterodimer formed with PEF1 interacts with, and bridges together the BCR(KLHL12) complex and SEC31 (SEC31A or SEC31B), promoting monoubiquitination of SEC31 and subsequent collagen export, which is required for neural crest specification. Involved in the regulation of the distribution and function of MCOLN1 in the endosomal pathway. Promotes localization and polymerization of TFG at endoplasmic reticulum exit site. Required for T-cell receptor-, Fas-, and glucocorticoid- induced apoptosis (By similarity). May mediate Ca(2+)-regulated signals along the death pathway: interaction with DAPK1 can accelerate apoptotic cell death by increasing caspase-3 activity. Its role in apoptosis may however be indirect, as suggested by knockout experiments (By similarity). May inhibit KDR/VEGFR2-dependent angiogenesis; the function involves inhibition of VEGF-induced phosphorylation of the Akt signaling pathway. -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

Annexin 7 Mobilizes Calcium from Endoplasmic Reticulum Stores in Brainb
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Biochimica et Biophysica Acta 1742 (2004) 151–160 http://www.elsevier.com/locate/bba Review Annexin 7 mobilizes calcium from endoplasmic reticulum stores in brainB W.D. Watsona,1, M. Srivastavab,1, X. Leightonb, M. Glasmanb, M. Faradayc, L.H. Fossamd, H.B. Pollardb,*, A. Vermae aNeurology Department, National Naval Medical Center, Bethesda, MD 20814, United States bDepartment of Anatomy, Physiology, and Genetics, 4301 Jones Bridge Road, Bethesda, MD 20814, United States cDepartment of Medical Psychology, Bethesda, MD 20814, United States dDivision of Neuropharmacological Drug Products, CDER, FDA, Rockville, MD 20852, United States eDepartment of Neurology, Uniformed Services University of the Health Sciences, Bethesda, MD 20814, United States Received 18 September 2004; received in revised form 5 October 2004; accepted 12 October 2004 Available online 29 October 2004 Abstract Mobilization of intracellular calcium from inositol-1,4,5-triphosphate (IP3)-sensitive endoplasmic reticulum (ER) stores plays a prominent role in brain function. Mice heterozygous for the annexin A7 (Anx7) gene have a profound reduction in IP3 receptor function in pancreatic islets along with defective insulin secretion. We examined IP3-sensitive calcium pools in the brains of Anx7 (+/À) mice by utilizing ATP/Mg2+-dependent 45Ca2+ uptake into brain membrane preparations and tissue sections. Although the Anx7 (+/À) mouse brain displayed similar levels of IP3 binding sites and thapsigargin-sensitive 45Ca2+ uptake as that seen in wild-type mouse brain, the Anx7 (+/À) mouse brain Ca2+ pools showed markedly reduced sensitivity to IP3. -

PDCD6IP (Human) Recombinant Protein (P01)
PDCD6IP (Human) Recombinant Preparation Method: in vitro wheat germ expression Protein (P01) system Purification: Glutathione Sepharose 4 Fast Flow Catalog Number: H00010015-P01 Storage Buffer: 50 mM Tris-HCI, 10 mM reduced Regulation Status: For research use only (RUO) Glutathione, pH=8.0 in the elution buffer. Product Description: Human PDCD6IP full-length ORF Storage Instruction: Store at -80°C. Aliquot to avoid ( AAH20066, 1 a.a. - 868 a.a.) recombinant protein with repeated freezing and thawing. GST-tag at N-terminal. Entrez GeneID: 10015 Sequence: MATFISVQLKKTSEVDLAKPLVKFIQQTYPSGGEEQAQ Gene Symbol: PDCD6IP YCRAAEELSKLRRAAVGRPLDKHEGALETLLRYYDQIC SIEPKFPFSENQICLTFTWKDAFDKGSLFGGSVKLALA Gene Alias: AIP1, Alix, DRIP4, HP95, MGC17003 SLGYEKSCVLFNCAALASQIAAEQNLDNDEGLKIAAKH YQFASGAFLHIKETVLSALSREPTVDISPDTVGTLSLIM Gene Summary: This gene encodes a protein thought LAQAQEVFFLKATRDKMKDAIIAKLANQAADYFGDAFK to participate in programmed cell death. Studies using QCQYKDTLPKEVFPVLAAKHCIMQANAEYHQSILAKQ mouse cells have shown that overexpression of this QKKFGEEIARLQHAAELIKTVASRYDEYVNVKDFSDKI protein can block apoptosis. In addition, the product of NRALAAAKKDNDFIYHDRVPDLKDLDPIGKATLVKSTP this gene binds to the product of the PDCD6 gene, a VNVPISQKFTDLFEKMVPVSVQQSLAAYNQRKADLVN protein required for apoptosis, in a calcium-dependent RSIAQMREATTLANGVLASLNLPAAIEDVSGDTVPQSIL manner. This gene product also binds to endophilins, TKSRSVIEQGGIQTVDQLIKELPELLQRNREILDESLRLL proteins that regulate membrane shape during DEEEATDNDLRAKFKERWQRTPSNELYKPLRAEGTNF endocytosis. -
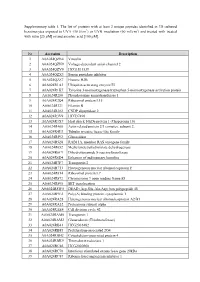
Supplementary Table 1. the List of Proteins with at Least 2 Unique
Supplementary table 1. The list of proteins with at least 2 unique peptides identified in 3D cultured keratinocytes exposed to UVA (30 J/cm2) or UVB irradiation (60 mJ/cm2) and treated with treated with rutin [25 µM] or/and ascorbic acid [100 µM]. Nr Accession Description 1 A0A024QZN4 Vinculin 2 A0A024QZN9 Voltage-dependent anion channel 2 3 A0A024QZV0 HCG1811539 4 A0A024QZX3 Serpin peptidase inhibitor 5 A0A024QZZ7 Histone H2B 6 A0A024R1A3 Ubiquitin-activating enzyme E1 7 A0A024R1K7 Tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein 8 A0A024R280 Phosphoserine aminotransferase 1 9 A0A024R2Q4 Ribosomal protein L15 10 A0A024R321 Filamin B 11 A0A024R382 CNDP dipeptidase 2 12 A0A024R3V9 HCG37498 13 A0A024R3X7 Heat shock 10kDa protein 1 (Chaperonin 10) 14 A0A024R408 Actin related protein 2/3 complex, subunit 2, 15 A0A024R4U3 Tubulin tyrosine ligase-like family 16 A0A024R592 Glucosidase 17 A0A024R5Z8 RAB11A, member RAS oncogene family 18 A0A024R652 Methylenetetrahydrofolate dehydrogenase 19 A0A024R6C9 Dihydrolipoamide S-succinyltransferase 20 A0A024R6D4 Enhancer of rudimentary homolog 21 A0A024R7F7 Transportin 2 22 A0A024R7T3 Heterogeneous nuclear ribonucleoprotein F 23 A0A024R814 Ribosomal protein L7 24 A0A024R872 Chromosome 9 open reading frame 88 25 A0A024R895 SET translocation 26 A0A024R8W0 DEAD (Asp-Glu-Ala-Asp) box polypeptide 48 27 A0A024R9E2 Poly(A) binding protein, cytoplasmic 1 28 A0A024RA28 Heterogeneous nuclear ribonucleoprotein A2/B1 29 A0A024RA52 Proteasome subunit alpha 30 A0A024RAE4 Cell division cycle 42 31 -

Directional Exosome Proteomes Reflect Polarity-Specific Functions in Retinal Pigmented Epithelium Monolayers
www.nature.com/scientificreports Correction: Author Correction OPEN Directional Exosome Proteomes Refect Polarity-Specifc Functions in Retinal Pigmented Epithelium Received: 9 February 2017 Accepted: 30 May 2017 Monolayers Published online: 07 July 2017 Mikael Klingeborn1, W. Michael Dismuke1, Nikolai P. Skiba1, Una Kelly1, W. Daniel Stamer1,2 & Catherine Bowes Rickman1,3 The retinal pigmented epithelium (RPE) forms the outer blood-retinal barrier in the eye and its polarity is responsible for directional secretion and uptake of proteins, lipoprotein particles and extracellular vesicles (EVs). Such a secretional division dictates directed interactions between the systemic circulation (basolateral) and the retina (apical). Our goal is to defne the polarized proteomes and physical characteristics of EVs released from the RPE. Primary cultures of porcine RPE cells were diferentiated into polarized RPE monolayers on permeable supports. EVs were isolated from media bathing either apical or basolateral RPE surfaces, and two subpopulations of small EVs including exosomes, and dense EVs, were purifed and processed for proteomic profling. In parallel, EV size distribution and concentration were determined. Using protein correlation profling mass spectrometry, a total of 631 proteins were identifed in exosome preparations, 299 of which were uniquely released apically, and 94 uniquely released basolaterally. Selected proteins were validated by Western blot. The proteomes of these exosome and dense EVs preparations suggest that epithelial polarity impacts directional release. These data serve as a foundation for comparative studies aimed at elucidating the role of exosomes in the molecular pathophysiology of retinal diseases and help identify potential therapeutic targets and biomarkers. Te retinal pigmented epithelium (RPE) is a cell monolayer that is situated between the photoreceptors and the systemic circulation of the choroid.