Deep-Sea Research Part 1 (In Press)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Development of Species-Specific Edna-Based Test Systems For
REPORT SNO 7544-2020 Development of species-specific eDNA-based test systems for monitoring of non-indigenous Decapoda in Danish marine waters © Henrik Carl, Natural History Museum, Denmark History © Henrik Carl, Natural NIVA Denmark Water Research REPORT Main Office NIVA Region South NIVA Region East NIVA Region West NIVA Denmark Gaustadalléen 21 Jon Lilletuns vei 3 Sandvikaveien 59 Thormøhlensgate 53 D Njalsgade 76, 4th floor NO-0349 Oslo, Norway NO-4879 Grimstad, Norway NO-2312 Ottestad, Norway NO-5006 Bergen Norway DK 2300 Copenhagen S, Denmark Phone (47) 22 18 51 00 Phone (47) 22 18 51 00 Phone (47) 22 18 51 00 Phone (47) 22 18 51 00 Phone (45) 39 17 97 33 Internet: www.niva.no Title Serial number Date Development of species-specific eDNA-based test systems for monitoring 7544-2020 22 October 2020 of non-indigenous Decapoda in Danish marine waters Author(s) Topic group Distribution Steen W. Knudsen and Jesper H. Andersen – NIVA Denmark Environmental monitor- Public Peter Rask Møller – Natural History Museum, University of Copenhagen ing Geographical area Pages Denmark 54 Client(s) Client's reference Danish Environmental Protection Agency (Miljøstyrelsen) UCB and CEKAN Printed NIVA Project number 180280 Summary We report the development of seven eDNA-based species-specific test systems for monitoring of marine Decapoda in Danish marine waters. The seven species are 1) Callinectes sapidus (blå svømmekrabbe), 2) Eriocheir sinensis (kinesisk uldhånds- krabbe), 3) Hemigrapsus sanguineus (stribet klippekrabbe), 4) Hemigrapsus takanoi (pensel-klippekrabbe), 5) Homarus ameri- canus (amerikansk hummer), 6) Paralithodes camtschaticus (Kamchatka-krabbe) and 7) Rhithropanopeus harrisii (østameri- kansk brakvandskrabbe). -

Changes in Biomass and Chemical Composition During Lecithotrophic Larval Development of the Southern Stone Crab Paralomis Granulosa
MARINE ECOLOGY PROGRESS SERIES Vol. 257: 189–196, 2003 Published August 7 Mar Ecol Prog Ser Changes in biomass and chemical composition during lecithotrophic larval development of the southern stone crab Paralomis granulosa Javier A. Calcagno1,*, Sven Thatje2, Klaus Anger3, Gustavo A. Lovrich4, Antje Kaffenberger3 1Universidad de Buenos Aires, Facultad de Ciencias Exactas y Naturales, Intendente Güiraldes 2160, Lab 64, 4to Piso, Pab II, Cdad Universitaria C1428EHA, Buenos Aires, Argentina 2Alfred Wegener Institute for Polar and Marine Research, PO Box 120 161, 27515 Bremerhaven, Germany 3Biologische Anstalt Helgoland, Stiftung Alfred Wegener Institute for Polar and Marine Research, 27498 Helgoland, Germany 4Consejo Nacional de Investigaciones Científicas y Técnicas, Centro Austral de Investigaciones Científicas, CADIC, CC 92, V9410BFD Ushuaia, Tierra del Fuego, Argentina ABSTRACT: Changes in biomass and elemental composition (dry mass, W; carbon, C; nitrogen, N; hy- drogen, H) were studied in the laboratory during complete larval and early juvenile development of the southern stone crab Paralomis granulosa (Jacquinot). At 6 ± 0.5°C; total larval development from hatching to metamorphosis lasted ca. 56 d, comprising 2 demersal zoeal stages and a benthic mega- lopa, with mean stage durations of 5, 11 and 45 d, respectively. All larval stages of P. granulosa are lecithotrophic, and first feeding and growth were consistently observed immediately after meta- morphosis to the first juvenile crab stage. Regardless of presence or absence of food, W, C, N, and H decreased throughout larval development. Also the C:N mass ratio decreased significantly, from 7.2 at hatching to 4.2 at metamorphosis, indicating that a large initial lipid store remaining from the egg yolk was gradually utilised as an internal energy source. -

How to Become a Crab: Phenotypic Constraints on a Recurring Body Plan
Preprints (www.preprints.org) | NOT PEER-REVIEWED | Posted: 25 December 2020 doi:10.20944/preprints202012.0664.v1 How to become a crab: Phenotypic constraints on a recurring body plan Joanna M. Wolfe1*, Javier Luque1,2,3, Heather D. Bracken-Grissom4 1 Museum of Comparative Zoology and Department of Organismic & Evolutionary Biology, Harvard University, 26 Oxford St, Cambridge, MA 02138, USA 2 Smithsonian Tropical Research Institute, Balboa–Ancon, 0843–03092, Panama, Panama 3 Department of Earth and Planetary Sciences, Yale University, New Haven, CT 06520-8109, USA 4 Institute of Environment and Department of Biological Sciences, Florida International University, Biscayne Bay Campus, 3000 NE 151 Street, North Miami, FL 33181, USA * E-mail: [email protected] Summary: A fundamental question in biology is whether phenotypes can be predicted by ecological or genomic rules. For over 140 years, convergent evolution of the crab-like body plan (with a wide and flattened shape, and a bent abdomen) at least five times in decapod crustaceans has been known as ‘carcinization’. The repeated loss of this body plan has been identified as ‘decarcinization’. We offer phylogenetic strategies to include poorly known groups, and direct evidence from fossils, that will resolve the pattern of crab evolution and the degree of phenotypic variation within crabs. Proposed ecological advantages of the crab body are summarized into a hypothesis of phenotypic integration suggesting correlated evolution of the carapace shape and abdomen. Our premise provides fertile ground for future studies of the genomic and developmental basis, and the predictability, of the crab-like body form. Keywords: Crustacea, Anomura, Brachyura, Carcinization, Phylogeny, Convergent evolution, Morphological integration 1 © 2020 by the author(s). -

A Metapopulation Model for Whale-Fall Specialists: the Largest Whales Are Essential to Prevent Species Extinctions
THE SEA: THE CURRENT AND FUTURE OCEAN Journal of Marine Research, 77, Supplement, 283–302, 2019 A metapopulation model for whale-fall specialists: The largest whales are essential to prevent species extinctions by Craig R. Smith,1,2 Joe Roman,3 and J. B. Nation4 ABSTRACT The sunken carcasses of great whales (i.e., whale falls) provide an important deep-sea habitat for more than 100 species that may be considered whale-fall specialists. Commercial whaling has reduced the abundance and size of whales, and thus whale-fall habitats, as great whales were hunted and removed from the oceans, often to near extinction. In this article, we use a metapopulation modeling approach to explore the consequences of whaling to the abundance and persistence of whale-fall habitats in the deep sea and to the potential for extinction of whale-fall specialists. Our modeling indicates that the persistence of metapopulations of whale-fall specialists is linearly related to the abundance of whales, and extremely sensitive (to the fourth power) to the mean size of whales. Thus, whaling-induced declines in the mean size of whales are likely to have been as important as declines in whale abundance to extinction pressure on whale-fall specialists. Our modeling also indicates that commercial whaling, even under proposed sustainable yield scenarios, has the potential to yield substantial extinction of whale-fall specialists. The loss of whale-fall habitat is likely to have had the greatest impact on the diversity of whale-fall specialists in areas where whales have been hunted for centuries, allowing extinctions to proceed to completion. -

Role of Cetaceans in Ecosystem Functioning
WORKSHOP REPORT Role of Cetaceans in Ecosystem Functioning: Defining Marine Conservation Policies in the 21st Century 28th International Congress for Conservation Biology Society for Conservation Biology Workshop Report Role of Cetaceans in Ecosystem Functioning: Defining Marine Conservation Policies in the 21st Century 28th International Congress for Conservation Biology Society for Conservation Biology 26 July 2017, Cartagena, Colombia Room Barahona 1, Cartagena Convention Center www.ccc-chile.org www.icb.org.ar www.whales.org www.oceancare.org www.hsi.org csiwhalesalive.org www.nrdc.org www.minrel.gob.cl www.belgium.be For centuries, the great whales (baleen whales and the scientists, but to ecological economists (who ascribe finan- sperm whale) and other cetaceans1 (small whales, dolphins cial values to ecological functions) and to and porpoises) were valued almost exclusively for their oil policymakers concerned with conserving biodiversity. and meat. Widespread commercial hunting reduced great These services confirm what the public, since the early whale numbers by as much as 90 percent, with some ‘Save the Whale’ movement in the 1970s, has always un- populations being hunted to extinction. derstood; cetaceans are special. In recent decades, changing attitudes toward protecting The global implications of the significant contributions of wildlife and the natural world and the growth of ecotourism cetaceans “to ecosystem functioning that are beneficial for provided new cultural and non-extractive economic values the natural environment and people” were first formally for these marine mammals. acknowledged in 2016 when the International Whaling Commission (IWC) adopted a resolution on Cetaceans and Today, whale watching is worth more than $2 billion annu- Their Contributions to Ecosystem Functioning2. -

Deep-Sea Biology
Deep-Sea Biology (OCN430) - Syllabus Fall 2017 Instructors: Jeff Drazen, office MSB605, [email protected], 956-6567 Craig Smith, office MSB617, [email protected], 956-7776 T TH 12:00-1:15 POST708 Syllabus schedule subject to change Course Goals – The deep sea is the largest living space on the planet. Its inhabitants are varied and its communities are often complex, adapted to the particular characteristics of their habitat. This course will cover the major topics in the field, such as bentho-pelagic coupling, depth zonation, energetics, diversity, ecosystem function, adaptations, and the ecology of major habitats. The last portion of the course will deal with anthropogenic threats such as deep-sea fisheries, mining and global climate change. Its goal is to provide you with a basic understanding of what we know (and don’t know) about the biology, ecology and biodiversity of deep-sea ecosystems, the methods used in the field, and it will create a forum for discussion of the major current questions and recent exciting discoveries. Course Structure – After each lecture (or pair of lectures) students will lead a discussion session. The lectures will present the basics of the topics. The discussions will be based on assigned readings (primarily current scientific papers), allowing the class to explore the controversies, implications of recent findings, and highlight future directions for research. Student Learning Outcomes – At the end of this course you will be able to: 1) Describe the co-varying effects of temperature, pressure, oxygen and light levels on the adaptations of deep-sea organisms. 2) Evaluate the influence of variables co-varying with depth on communities, populations, and species. -

Feeding Habits of the False Southern King Crab Paralomis Granulosa (Lithodidae) in the Beagle Channel, Tierra Del Fuego, Argentina*
SCI. MAR., 63 (Supl. 1): 361-366 SCIENTIA MARINA 1999 MAGELLAN-ANTARCTIC: ECOSYSTEMS THAT DRIFTED APART. W.E. ARNTZ and C. RÍOS (eds.) Feeding habits of the false southern king crab Paralomis granulosa (Lithodidae) in the Beagle Channel, Tierra del Fuego, Argentina* LAURA INÉS COMOGLIO and OSCAR ANTONIO AMIN Centro Austral de Investigaciones Científicas (CADIC-CONICET), CC92, 9410 Ushuaia, Tierra del Fuego, Argentina SUMMARY: Stomach contents of 282 false southern king crabs (Paralomis granulosa), between 10 to 90 mm CL from the Beagle Channel (Golondrina Bay and Roca Mora), Argentina, were examined by the frequency of occurrence method of analy- sis and by a food index. Roca Mora is an area where juveniles (<50 mm CL) dominate and in Golondrina Bay adults (>60 mm CL) are common; in this last area sexual segregation was also observed. The principal food groups for crabs of Golondrina Bay were algae, molluscs, crustaceans, bryozoans and foraminiferans; for crabs from Roca Mora the natural diet consisted of three major food groups: hydrozoans, echinoderms and foraminiferans. The relative frequency of different prey groups varied in rela- tion to the size, season and sex. There were no significant differences in the quantity of food consumed by sexes in both areas. Generally small crabs (<40 mm CL) contained more food than large crabs (>50 mm CL). Juveniles consumed a greater amount of food during winter and spring. In summer (moulting period), juveniles had the highest vacuity index. Adults consumed minor amounts of food during autumn, before the spawning-moulting-mating period when the vacuity index was higher (spring). -

A Review of Worldwide Fisheries for Lithodid Crabs (Decapoda: Anomura: Lithodidae) and Their Fluctuations
Bi ology ofAnomura II (A .Asakura ,e d.),Cr ustaceanR esearc h,Sp ec ialN umber6: 167-185,2006 A review of worldwide fisheries for lithodid crabs (Decapoda: Anomura: Lithodidae) and their fluctuations R obert S. Otto Abstra ct.-Lithodid crab fi sheries began species within these genera,trends in landings before1900 in Japan and spread across the and cu汀 ent status of major stocks contributing North Pacific Ocean by1940. Fisheries targeted to the world lithodid landings. lncidental or red kin gcrab (Paralithodes ca mtsc hati cus) with experimental fisheries for Neolithodes spp. and lesser amounts of blue king crab (P. platypus) for Lopholithodes spp. are omitt ed,because and brown king crab (P. brevip es) .Paralithod es documented landings are sporadic,trends spp.,es pec iall yred kin gcrabs ,h ave always are not evident and magnitudes negligible. dominated lithodid fisherie s. Golden king crab Likewise,1 have excluded recreational or (Lithodes aequispinus) becam eimportant in personal use fisheries,because statistics are North Pacific Ocean waters after major decline s frequently incomplete and known landings are in red king crab fisheries in the early 1980's. Southern kin gcrab (Lithodes sa ntolla) are fished generall ysm a ll relative to commercial白sheries. in so uthern South America along with softshell red crab (Paralomis granulosa). These five species DATASOURCES accounted for more than 89% of lithodid landings for 1984・2003. World lithodid landings pea ked at 1use United Nations Food and Agricultural 150,100 metric tons (t) in 1966 after development Organization (FAO) landing statistic sas a in pre-World War 11 Asia and rapid post-1950 starting point and more detai led publications ex pansion in A laskaand Asia. -
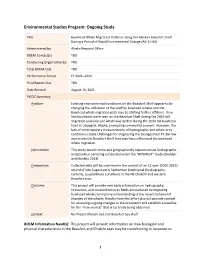
SDP PICOC Template
Environmental Studies Program: Ongoing Study Title Bowhead Whale Migration Patterns along the Alaskan Beaufort Shelf During a Period of Rapid Environmental Change (AK-21-04) Administered by Alaska Regional Office BOEM Contact(s) TBD Conducting Organization(s) TBD Total BOEM Cost TBD Performance Period FY 2021–2024 Final Report Due TBD Date Revised August 19, 2021 PICOC Summary Problem Evolving environmental conditions on the Beaufort Shelf appear to be changing the utilization of the shelf by bowhead whales and the bowhead whale migration path may be shifting farther offshore. Very few bowheads were seen on the Beaufort Shelf during the 2019 fall migration and only one whale was landed during the 2019 fall bowhead hunt at Utqiaġvik, Alaska, prompting community concern. However, the lack of contemporary measurements of hydrographic and whale prey conditions create challenges for diagnosing the changes near Pt. Barrow and across the Beaufort Shelf that may have influenced the bowhead whale migration. Intervention This study would renew and geographically expand annual hydrographic and plankton sampling conducted under the “BOWFEST” study (Shelden and Mocklin 2013). Comparison Collected data will be examined in the context of an 11-year (2005-2015) record of late August-early September biophysical (hydrography, currents, zooplankton) conditions in the NE Chukchi and western Beaufort seas. Outcome This project will provide new basic information on hydrography, circulation, and zooplankton prey fields encountered by migrating bowhead whales to improve understanding of the recent behavioral changes of the whales. Results from this effort also will provide context for assessing ongoing changes to the ecosystem and establish a baseline for the “new normal” that is currently being observed. -

Articles and Detrital Matter
Biogeosciences, 7, 2851–2899, 2010 www.biogeosciences.net/7/2851/2010/ Biogeosciences doi:10.5194/bg-7-2851-2010 © Author(s) 2010. CC Attribution 3.0 License. Deep, diverse and definitely different: unique attributes of the world’s largest ecosystem E. Ramirez-Llodra1, A. Brandt2, R. Danovaro3, B. De Mol4, E. Escobar5, C. R. German6, L. A. Levin7, P. Martinez Arbizu8, L. Menot9, P. Buhl-Mortensen10, B. E. Narayanaswamy11, C. R. Smith12, D. P. Tittensor13, P. A. Tyler14, A. Vanreusel15, and M. Vecchione16 1Institut de Ciencies` del Mar, CSIC. Passeig Mar´ıtim de la Barceloneta 37-49, 08003 Barcelona, Spain 2Biocentrum Grindel and Zoological Museum, Martin-Luther-King-Platz 3, 20146 Hamburg, Germany 3Department of Marine Sciences, Polytechnic University of Marche, Via Brecce Bianche, 60131 Ancona, Italy 4GRC Geociencies` Marines, Parc Cient´ıfic de Barcelona, Universitat de Barcelona, Adolf Florensa 8, 08028 Barcelona, Spain 5Universidad Nacional Autonoma´ de Mexico,´ Instituto de Ciencias del Mar y Limnolog´ıa, A.P. 70-305 Ciudad Universitaria, 04510 Mexico,` Mexico´ 6Woods Hole Oceanographic Institution, MS #24, Woods Hole, MA 02543, USA 7Integrative Oceanography Division, Scripps Institution of Oceanography, La Jolla, CA 92093-0218, USA 8Deutsches Zentrum fur¨ Marine Biodiversitatsforschung,¨ Sudstrand¨ 44, 26382 Wilhelmshaven, Germany 9Ifremer Brest, DEEP/LEP, BP 70, 29280 Plouzane, France 10Institute of Marine Research, P.O. Box 1870, Nordnes, 5817 Bergen, Norway 11Scottish Association for Marine Science, Scottish Marine Institute, Oban, -

About Seafood Watch®
Southern king crab Lithodes santolla ©Monterey Bay Aquarium Argentine waters Traps January 2, 2013 Kelsey James, Consulting Researcher Disclaimer Seafood Watch® strives to ensure all our Seafood Reports and the recommendations contained therein are accurate and reflect the most up-to-date evidence available at time of publication. All our reports are peer- reviewed for accuracy and completeness by external scientists with expertise in ecology, fisheries science or aquaculture. Scientific review, however, does not constitute an endorsement of the Seafood Watch program or its recommendations on the part of the reviewing scientists. Seafood Watch is solely responsible for the conclusions reached in this report. We always welcome additional or updated data that can be used for the next revision. Seafood Watch and Seafood Reports are made possible through a grant from the David and Lucile Packard Foundation. 2 Final Seafood Recommendation Southern king crab (Lithodes santolla) from trap fisheries within Argentine waters is assessed as a Good Alternative. Stock Fishery Impacts Impacts on Manage- Habitat Overall on the other Species ment and Recommendation Stock Ecosystem Rank (Lowest scoring Rank Rank (Score) (Score) species (Score) (Score) Rank*, Subscore, Score) Southern king Trap No other main GOOD crab Yellow Red Yellow species caught ALTERNATIVE (2.64) (2) (3.12) Green, (5,4.5) (2.93) Scoring note – scores range from zero to five where zero indicates very poor performance and five indicates the fishing operations have no significant impact. -

An Illustrated Key to the Malacostraca (Crustacea) of the Northern Arabian Sea. Part VI: Decapoda Anomura
An illustrated key to the Malacostraca (Crustacea) of the northern Arabian Sea. Part 6: Decapoda anomura Item Type article Authors Kazmi, Q.B.; Siddiqui, F.A. Download date 04/10/2021 12:44:02 Link to Item http://hdl.handle.net/1834/34318 Pakistan Journal of Marine Sciences, Vol. 15(1), 11-79, 2006. AN ILLUSTRATED KEY TO THE MALACOSTRACA (CRUSTACEA) OF THE NORTHERN ARABIAN SEA PART VI: DECAPODA ANOMURA Quddusi B. Kazmi and Feroz A. Siddiqui Marine Reference Collection and Resource Centre, University of Karachi, Karachi-75270, Pakistan. E-mails: [email protected] (QBK); safianadeem200 [email protected] .in (FAS). ABSTRACT: The key deals with the Decapoda, Anomura of the northern Arabian Sea, belonging to 3 superfamilies, 10 families, 32 genera and 104 species. With few exceptions, each species is accompanied by illustrations of taxonomic importance; its first reporter is referenced, supplemented by a subsequent record from the area. Necessary schematic diagrams explaining terminologies are also included. KEY WORDS: Malacostraca, Decapoda, Anomura, Arabian Sea - key. INTRODUCTION The Infraorder Anomura is well represented in Northern Arabian Sea (Paldstan) (see Tirmizi and Kazmi, 1993). Some important investigations and documentations on the diversity of anomurans belonging to families Hippidae, Albuneidae, Lithodidae, Coenobitidae, Paguridae, Parapaguridae, Diogenidae, Porcellanidae, Chirostylidae and Galatheidae are as follows: Alcock, 1905; Henderson, 1893; Miyake, 1953, 1978; Tirmizi, 1964, 1966; Lewinsohn, 1969; Mustaquim, 1972; Haig, 1966, 1974; Tirmizi and Siddiqui, 1981, 1982; Tirmizi, et al., 1982, 1989; Hogarth, 1988; Tirmizi and Javed, 1993; and Siddiqui and Kazmi, 2003, however these informations are scattered and fragmentary. In 1983 McLaughlin suppressed the old superfamily Coenobitoidea and combined it with the superfamily Paguroidea and placed all hermit crab families under the superfamily Paguroidea.