Proteomics of Phosphorylation and Protein Dynamics During Fertilization
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Tyrosine Kinase – Role and Significance in Cancer
Int. J. Med. Sci. 2004 1(2): 101-115 101 International Journal of Medical Sciences ISSN 1449-1907 www.medsci.org 2004 1(2):101-115 ©2004 Ivyspring International Publisher. All rights reserved Review Tyrosine kinase – Role and significance in Cancer Received: 2004.3.30 Accepted: 2004.5.15 Manash K. Paul and Anup K. Mukhopadhyay Published:2004.6.01 Department of Biotechnology, National Institute of Pharmaceutical Education and Research, Sector-67, S.A.S Nagar, Mohali, Punjab, India-160062 Abstract Tyrosine kinases are important mediators of the signaling cascade, determining key roles in diverse biological processes like growth, differentiation, metabolism and apoptosis in response to external and internal stimuli. Recent advances have implicated the role of tyrosine kinases in the pathophysiology of cancer. Though their activity is tightly regulated in normal cells, they may acquire transforming functions due to mutation(s), overexpression and autocrine paracrine stimulation, leading to malignancy. Constitutive oncogenic activation in cancer cells can be blocked by selective tyrosine kinase inhibitors and thus considered as a promising approach for innovative genome based therapeutics. The modes of oncogenic activation and the different approaches for tyrosine kinase inhibition, like small molecule inhibitors, monoclonal antibodies, heat shock proteins, immunoconjugates, antisense and peptide drugs are reviewed in light of the important molecules. As angiogenesis is a major event in cancer growth and proliferation, tyrosine kinase inhibitors as a target for anti-angiogenesis can be aptly applied as a new mode of cancer therapy. The review concludes with a discussion on the application of modern techniques and knowledge of the kinome as means to gear up the tyrosine kinase drug discovery process. -

Fast Pressure-Jump All-Atom Simulations and Experiments Reveal Site-Specific Protein Dehydration-Folding Dynamics
Fast pressure-jump all-atom simulations and experiments reveal site-specific protein dehydration-folding dynamics Maxim B. Prigozhina,1,2, Yi Zhangb,1, Klaus Schultenb,c, Martin Gruebelea,b,c,3, and Taras V. Pogorelova,b,d,e,3 aDepartment of Chemistry, University of Illinois at Urbana–Champaign, Urbana, IL 61801; bBeckman Institute for Advanced Science and Technology, Center for Biophysics and Quantitative Biology, University of Illinois at Urbana–Champaign, Urbana, IL 61801; cDepartment of Physics, University of Illinois at Urbana–Champaign, Urbana, IL 61801; dNational Center for Supercomputing Applications, University of Illinois at Urbana–Champaign, Urbana, IL 61801; and eSchool of Chemical Sciences, University of Illinois at Urbana–Champaign, Urbana, IL 61801 Edited by José N. Onuchic, Rice University, Houston, TX, and approved February 8, 2019 (received for review August 30, 2018) As theory and experiment have shown, protein dehydration is a and simulated using MD (18). Desolvated cavities in the protein major contributor to protein folding. Dehydration upon folding core (15) are restored by downward pressure jumps, which fa- can be characterized directly by all-atom simulations of fast cilitate refolding without a change in solvent or thermal motion. pressure drops, which create desolvated pockets inside the In addition, pressure perturbation reduces the effects of protein nascent hydrophobic core. Here, we study pressure-drop refolding aggregation, common in temperature-jump (T-jump) experi- of three λ-repressor fragment (λ6–85) mutants computationally and ments performed near the denaturation temperature (16, 17). experimentally. The three mutants report on tertiary structure for- Finally, pressure-jump (P-jump) experiments can utilize intrinsic mation via different fluorescent helix–helix contact pairs. -

Goat Anti-4E-T / EIF4ENIF1 Antibody Catalog No: Tcva06230
Web: www.taiclone.com Tel: +886-2-2735-9682 Email: [email protected] Goat anti-4E-T / EIF4ENIF1 Antibody Catalog No: tcva06230 Available Sizes Size: 100µg Specifications Application: Pep-ELISA, WB, IHC Research Area: RNA-sorting; mRNA decay; P-body; NIF transporter; translation Species Reactivity: Human, Mouse, Rat, Dog, Pig, Cow Host Species: Goat Immunogen / Amino acids: C-AKVISVDELEYRQ Conjugation: Unconjugated Form: Liquid Storage Buffer: Tris saline, 0.02% sodium azide, pH7.3 with 0.5% bovine serum albumin Concentration: 0.5 mg/ml in 200 µl Recommended Dilution: Western Blot: Approx 140-150kDa band observed in 293 lysates (predicted size of approx. 108kDa according to NP_062817.1 however our observation agrees with that of Dostie et al (see below) ). Recommended for use at 0.25-0.5µg/ml Copyright 2021 Taiclone Biotech Corp. Web: www.taiclone.com Tel: +886-2-2735-9682 Email: [email protected] Peptide ELISA: antibody detection limit dilution 1:32000. Amino Acid Sequence: NP_062817.2; NP_001157974.1 Storage Instruction: Aliquot store at -20C. Avoid freeze / thaw cycles. Alternative Names: EIF4ENIF1; 4E-T; Clast4; FLJ21601; 2610509L04Rik; eukaryotic translation initiation factor 4E nuclear import factor 1; eIF4E-transporter; FLJ26551; OTTHUMP00000063276 Gene ID: 56478 (human);74203 (mouse); Reference Sequence No.: NP_062817.2; NP_001157974.1 Calculated Molecular Weight: 108; 88.2 Purification: Purified from goat serum by ammonium sulphate precipitation followed by antigen affinity chromatography using the immunizing peptide DS Gene Ontology Terms: protein transporter; nucleocytoplasmic transport; cytoplasm; nucleus Positive Control: tcva06230p Notes This antibody is expected to recognise isoform 1 (NP_062817.2) and isoform b (NP_001157974.1). Reported variants represent identical protein (NP_062817.2; NP_001157973.1). -

Principles of Protein Phosphorylation Biophysical Chemistry 1, Fall 2010
Principles of protein phosphorylation Biophysical Chemistry 1, Fall 2010 Signalling “cascades” Structural biology of phosphorylation Web assignment: http://pkr.genomics.purdue.edu Reversible protein phosphorylation Enzymatic reaction Posttranslational Control ΔG~12kcal/mol Kinases and phosphatases Phosphatase Kinase dephospho- phosphorylates rylates General Examples Signalling overview Cells are way too complex! MAPK/ERK Signaling Pathway RousExample:sarcoma virus Rous (RSV) sarcoma virus (RSV) gag - encodes capsid proteins pol - encodes reverse transcriptase env - encodes envelope proteins src - encodes a tyrosine kinase that attaches phosphate groups to the amino acid tyrosine in host cell proteins MutationsExample:, viruses Rous and cancer sarcoma virus (RSV) v-src lacks the C-terminal inhibitory phosphorylation site (tyrosine-527), and is therefore constitutively active as opposed to normal src (c-src) Continuous cell profileration tumor BiophysicsStructural of signalling Effect of Phosphorylation Phosphorylation is an important regulatory mechanism Can reversible attach/detach a phosphate and therefore switch “on”/”off” the function Effect of phosphorylation is manifold • Conformational change • Ordering/disordering • Electrostatic effects • Alternate binding behavior Signalling by reorientation Reorientation: A conformational switch Rmsd = 2.5Å DHP (red) and Z-Score = 4.6 DHPs74e (blue) (>3.6 same fold) Title: A phosphorylation-induced conformation change in dematin headpiece Author(s): Jiang ZHG, McKnight CJ Source: STRUCTURE Volume: 14 Issue: 2 Pages: 379-387 Published: FEB 2006 Order/disorderDisordering:transitions NtrC, a molecular switch upon phosphorylation Orange-yellow: unphosphorylated NtrC blue-cyan: phosphorylated NtrC Volkman et al., Science 2001, 291, 2429-33 Alternate Binding: SRC SH2 Src/SH2 interactions: binding vs release domain binding ExpectedCan conformationalwe understand effects (predict) the effect of phosphorylation Electrostatics Hydrogen bonding Size Mutational analysis Experimental data Lubman, O.Y. -

Towards a Molecular Understanding of Microrna-Mediated Gene Silencing
REVIEWS NON-CODING RNA Towards a molecular understanding of microRNA-mediated gene silencing Stefanie Jonas and Elisa Izaurralde Abstract | MicroRNAs (miRNAs) are a conserved class of small non-coding RNAs that assemble with Argonaute proteins into miRNA-induced silencing complexes (miRISCs) to direct post-transcriptional silencing of complementary mRNA targets. Silencing is accomplished through a combination of translational repression and mRNA destabilization, with the latter contributing to most of the steady-state repression in animal cell cultures. Degradation of the mRNA target is initiated by deadenylation, which is followed by decapping and 5ʹ‑to‑3ʹ exonucleolytic decay. Recent work has enhanced our understanding of the mechanisms of silencing, making it possible to describe in molecular terms a continuum of direct interactions from miRNA target recognition to mRNA deadenylation, decapping and 5ʹ‑to‑3ʹ degradation. Furthermore, an intricate interplay between translational repression and mRNA degradation is emerging. Deadenylation MicroRNAs (miRNAs) are conserved post-transcriptional recruit additional protein partners to mediate silenc- 5,6 Shortening of mRNA poly(A) regulators of gene expression that are integral to ing . Silencing occurs through a combination of tails. In eukaryotes, this almost all known biological processes, including translational repression, deadenylation, decapping and process is catalysed by the cell growth, proliferation and differentiation, as well 5ʹ‑to‑3ʹ mRNA degradation5,6 (FIG. 1). The GW182 pro- consecutive but partially as organismal metabolism and development1. The teins play a central part in this process and are among redundant action of two 5,6 cytoplasmic deadenylase number of miRNAs encoded within the genomes of the most extensively studied AGO partners . -

Studying Backbone Torsional Dynamics of Intrinsically Disordered Proteins Using fluorescence Depolarization Kinetics
J Biosci Vol. 43, No. 3, July 2018, pp. 455–462 Ó Indian Academy of Sciences DOI: 10.1007/s12038-018-9766-1 Studying backbone torsional dynamics of intrinsically disordered proteins using fluorescence depolarization kinetics 1,3 1,2,3 DEBAPRIYA DAS and SAMRAT MUKHOPADHYAY * 1Centre for Protein Science, Design and Engineering, Indian Institute of Science Education and Research (IISER) Mohali, Punjab, India 2Department of Biological Sciences, Indian Institute of Science Education and Research (IISER) Mohali, Punjab, India 3Department of Chemical Sciences, Indian Institute of Science Education and Research (IISER) Mohali, Punjab, India *Corresponding author (Email, [email protected]) Published online: 7 June 2018 Intrinsically disordered proteins (IDPs) do not autonomously adopt a stable unique 3D structure and exist as an ensemble of rapidly interconverting structures. They are characterized by significant conformational plasticity and are associated with several biological functions and dysfunctions. The rapid conformational fluctuation is governed by the backbone segmental dynamics arising due to the dihedral angle fluctuation on the Ramachandran /–w conformational space. We discovered that the intrinsic backbone torsional mobility can be monitored by a sensitive fluorescence readout, namely fluorescence depolarization kinetics, of tryptophan in an archetypal IDP such as a-synuclein. This methodology allows us to map the site-specific torsional mobility in the dihedral space within picosecond-nanosecond time range at a low protein concen- tration under the native condition. The characteristic timescale of * 1.4 ns, independent of residue position, represents collective torsional dynamics of dihedral angles (/ and w) of several residues from tryptophan and is independent of overall global tumbling of the protein. -

Linking Predictions of Protein Structure and Disorder Through Molecular Simulation
The Order-Disorder Continuum: Linking Predictions of Protein Structure and Disorder through Molecular Simulation The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Hsu, Claire C., Markus J. Buehler, and Anna Tarakanova. "The Order-Disorder Continuum: Linking Predictions of Protein Structure and Disorder through Molecular Simulation." Scientific Reports, 10 (February 2020): 2068. © 2020, The Author(s). As Published http://dx.doi.org/10.1038/s41598-020-58868-w Publisher Springer Science and Business Media LLC Citable link https://hdl.handle.net/1721.1/125166 Terms of Use Creative Commons Attribution 4.0 International license Detailed Terms https://creativecommons.org/licenses/by/4.0/ www.nature.com/scientificreports OPEN The Order-Disorder Continuum: Linking Predictions of Protein Structure and Disorder through Molecular Simulation Claire C. Hsu1, Markus J. Buehler2 & Anna Tarakanova3,4* Intrinsically disordered proteins (IDPs) and intrinsically disordered regions within proteins (IDRs) serve an increasingly expansive list of biological functions, including regulation of transcription and translation, protein phosphorylation, cellular signal transduction, as well as mechanical roles. The strong link between protein function and disorder motivates a deeper fundamental characterization of IDPs and IDRs for discovering new functions and relevant mechanisms. We review recent advances in experimental techniques that have improved identifcation of disordered regions -

The Conformation of Serum Albumin in Solution: a Combined Phosphorescence Depolarization-Hydrodynamic Modeling Study
2422 Biophysical Journal Volume 80 May 2001 2422–2430 The Conformation of Serum Albumin in Solution: A Combined Phosphorescence Depolarization-Hydrodynamic Modeling Study M. Luisa Ferrer,* Ricardo Duchowicz,* Beatriz Carrasco,† Jose´ Garcı´a de la Torre,† and A. Ulises Acun˜a* *Instituto Quı´mica-Fı´sica “Rocasolano”, Consejo Superior de Investigaciones Cientificas, 28006 Madrid, and †Departamento de Quı´mica-Fı´sica, Universidad de Murcia, 30071 Murcia, Spain ABSTRACT There is a striking disparity between the heart-shaped structure of human serum albumin (HSA) observed in single crystals and the elongated ellipsoid model used for decades to interpret the protein solution hydrodynamics at neutral pH. These two contrasting views could be reconciled if the protein were flexible enough to change its conformation in solution from that found in the crystal. To investigate this possibility we recorded the rotational motions in real time of an erythrosin- bovine serum albumin complex (Er-BSA) over an extended time range, using phosphorescence depolarization techniques. These measurements are consistent with the absence of independent motions of large protein segments in solution, in the time range from nanoseconds to fractions of milliseconds, and give a single rotational correlation time (BSA, 1 cP, 20°C) ϭ 40 Ϯ 2 ns. In addition, we report a detailed analysis of the protein hydrodynamics based on two bead-modeling methods. In the first, BSA was modeled as a triangular prismatic shell with optimized dimensions of 84 ϫ 84 ϫ 84 ϫ 31.5 Å, whereas in the second, the atomic-level structure of HSA obtained from crystallographic data was used to build a much more refined rough-shell model. -

Numerical Evaluation of Protein Global Vibrations at Terahertz Frequencies by Means of Elastic Lattice Models †
Proceedings Numerical Evaluation of Protein Global Vibrations at Terahertz Frequencies by Means of Elastic † Lattice Models Domenico Scaramozzino *, Giuseppe Lacidogna, Gianfranco Piana and Alberto Carpinteri Department of Structural, Geotechnical and Building Engineering, Politecnico di Torino, Corso Duca degli Abruzzi 24, 10129 Torino, Italy; [email protected] (G.L.); [email protected] (G.P.); [email protected] (A.C.) * Correspondence: [email protected] † Presented at 1st International Electronic Conference on Applied Sciences, 10–30 November 2020; Available online: https://asec2020.sciforum.net/. Published: 9 November 2020 Abstract: Proteins represent one of the most important building blocks for most biological processes. Their biological mechanisms have been found to correlate significantly with their dynamics, which is commonly investigated through molecular dynamics (MD) simulations. However, important insights on protein dynamics and biological mechanisms have also been obtained via much simpler and computationally efficient calculations based on elastic lattice models (ELMs). The application of structural mechanics approaches, such as modal analysis, to the protein ELMs has allowed to find impressive results in terms of protein dynamics and vibrations. The low-frequency vibrations extracted from the protein ELM are usually found to occur within the terahertz (THz) frequency range and correlate fairly accurately with the observed functional motions. In this contribution, the global vibrations of lysozyme will be investigated by means of a finite element (FE) truss model, and we will show that there exists complete consistency between the proposed FE approach and one of the more well-known ELMs for protein dynamics, the anisotropic network model (ANM). The proposed truss model can consequently be seen as a simple method, easily accessible to the structural mechanics community members, to analyze protein vibrations and their connections with the biological activity. -

Noelia Díaz Blanco
Effects of environmental factors on the gonadal transcriptome of European sea bass (Dicentrarchus labrax), juvenile growth and sex ratios Noelia Díaz Blanco Ph.D. thesis 2014 Submitted in partial fulfillment of the requirements for the Ph.D. degree from the Universitat Pompeu Fabra (UPF). This work has been carried out at the Group of Biology of Reproduction (GBR), at the Department of Renewable Marine Resources of the Institute of Marine Sciences (ICM-CSIC). Thesis supervisor: Dr. Francesc Piferrer Professor d’Investigació Institut de Ciències del Mar (ICM-CSIC) i ii A mis padres A Xavi iii iv Acknowledgements This thesis has been made possible by the support of many people who in one way or another, many times unknowingly, gave me the strength to overcome this "long and winding road". First of all, I would like to thank my supervisor, Dr. Francesc Piferrer, for his patience, guidance and wise advice throughout all this Ph.D. experience. But above all, for the trust he placed on me almost seven years ago when he offered me the opportunity to be part of his team. Thanks also for teaching me how to question always everything, for sharing with me your enthusiasm for science and for giving me the opportunity of learning from you by participating in many projects, collaborations and scientific meetings. I am also thankful to my colleagues (former and present Group of Biology of Reproduction members) for your support and encouragement throughout this journey. To the “exGBRs”, thanks for helping me with my first steps into this world. Working as an undergrad with you Dr. -
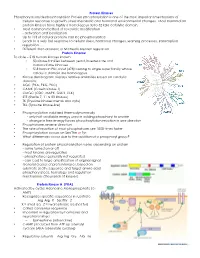
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Generation of H11-Albumin-Rtta Transgenic Mice: a Tool for Inducible Gene Expression in the Liver
INVESTIGATION Generation of H11-albumin-rtTA Transgenic Mice: A Tool for Inducible Gene Expression in the Liver Yu-Shan Li,*,1 Ran-Ran Meng,†,1 Xiu Chen,‡ Cui-Ling Shang,§ Hong-Bin Li,* Tao-Jun Zhang,† Hua-Yang Long,† Hui-Qi Li,† Yi-Jing Wang,† and Feng-Chao Wang†,2 *The School of Public Health, Xinxiang Medical University, Xinxiang, Henan, China 453003, †Transgenic Animal Center, National Institute of Biological Sciences, Beijing, China 102206, ‡Department of Pharmacy, Heze University, Heze, § Shandong, China, 274015, and Department of Reproductive Medicine, The Third Affiliated Hospital of Xinxiang Medical University, Xinxiang, Henan, China 453003 ABSTRACT The modification of the mouse genome by site-specific gene insertion of transgenes and other KEYWORDS genetic elements allows the study of gene function in different developmental stages and in the pathogenesis Hipp11 of diseases. Here, we generated a “genomic safe harbor” Hipp11 (H11)locus-specific knock-in transgenic albumin mouse line in which the albumin promoter is used to drive the expression of the reverse tetracycline promoter transactivator (rtTA) in the liver. The newly generated H11-albumin-rtTA transgenic mice were bred with doxycycline tetracycline-operator-Histone-2B-green fluorescent protein (TetO-H2BGFP) mice to assess inducibility and Tet-on system tissue-specificity. Expression of the H2BGFP fusion protein was observed exclusively upon doxycycline (Dox) induction in the liver of H11-albumin-rtTA/TetO-H2BGFP double transgenic mice. To further analyze the ability of the Dox-inducible H11-albumin-rtTA mice to implement conditional DNA recombination, H11- albumin-rtTA transgenic mice were crossed with TetO-Cre and Ai14 mice to generate H11-albumin-rtTA/ TetO-Cre/Ai14 triple transgenic mice.