PAPER Melanocortin-3-Receptor Gene Variants in Morbid Obesity
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Peptide, Peptidomimetic and Small Molecule Based Ligands Targeting Melanocortin Receptor System
PEPTIDE, PEPTIDOMIMETIC AND SMALL MOLECULE BASED LIGANDS TARGETING MELANOCORTIN RECEPTOR SYSTEM By ALEKSANDAR TODOROVIC A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY UNIVERSITY OF FLORIDA 2006 Copyright 2006 by Aleksandar Todorovic This document is dedicated to my family for everlasting support and selfless encouragement. ACKNOWLEDGMENTS I would like to thank and sincerely express my appreciation to all members, former and past, of Haskell-Luevano research group. First of all, I would like to express my greatest satisfaction by working with my mentor, Dr. Carrie Haskell-Luevano, whose guidance, expertise and dedication to research helped me reaching the point where I will continue the science path. Secondly, I would like to thank Dr. Ryan Holder who has taught me the principles of solid phase synthesis and initial strategies for the compounds design. I would like to thank Mr. Jim Rocca for the help and all necessary theoretical background required to perform proton 1-D NMR. In addition, I would like to thank Dr. Zalfa Abdel-Malek from the University of Cincinnati for the collaboration on the tyrosinase study project. Also, I would like to thank the American Heart Association for the Predoctoral fellowship that supported my research from 2004-2006. The special dedication and thankfulness go to my fellow graduate students within the lab and the department. iv TABLE OF CONTENTS page ACKNOWLEDGMENTS ................................................................................................ -

Functionality and Genetics of Melanocortin and Purinergic Receptors
University of Latvia Faculty of Biology Vita Ignatoviča Doctoral Thesis Functionality and genetics of melanocortin and purinergic receptors Promotion to the degree of Doctor of Biology Molecular Biology Supervisor: Dr. Biol. Jānis Kloviņš Riga, 2012 1 The doctoral thesis was carried out in University of Latvia, Faculty of Biology, Department of Molecular biology and Latvian Biomedical Reseach and Study centre. From 2007 to 2012 The research was supported by Latvian Council of Science (LZPSP10.0010.10.04), Latvian Research Program (4VPP-2010-2/2.1) and ESF funding (1DP/1.1.1.2.0/09/APIA/VIAA/150 and 1DP/1.1.2.1.2/09/IPIA/VIAA/004). The thesis contains the introduction, 9 chapters, 38 subchapters and reference list. Form of the thesis: collection of articles in biology with subdiscipline in molecular biology Supervisor: Dr. biol. Jānis Kloviņš Reviewers: 1) Dr. biol., Prof. Astrīda Krūmiņa, Latvian Biomedical Reseach and Study centre 2) Dr. biol., Prof. Ruta Muceniece, University of Latvia, Department of Medicine, Pharmacy program 3) PhD Med, Assoc.Prof.David Gloriam, University of Copenhagen, Department of Drug Design and Pharmacology The thesis will be defended at the public section of the Doctoral Commitee of Biology, University of Latvia, in the conference hall of Latvian Biomedical Research and Study centre on July 6th, 2012, at 11.00. The thesis is available at the Library of the University of Latvia, Kalpaka blvd. 4. This thesis is accepted of the commencement of the degree of Doctor of Biology on April 19th, 2012, by the Doctoral Commitee of Biology, University of Latvia. -
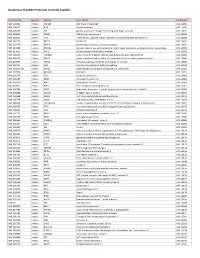
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Functional Characterization of Melanocortin-4 Receptor Mutations Associated with Childhood Obesity
0013-7227/03/$15.00/0 Endocrinology 144(10):4544–4551 Printed in U.S.A. Copyright © 2003 by The Endocrine Society doi: 10.1210/en.2003-0524 Functional Characterization of Melanocortin-4 Receptor Mutations Associated with Childhood Obesity YA-XIONG TAO AND DEBORAH L. SEGALOFF Department of Physiology and Biophysics, The University of Iowa, Iowa City, Iowa 52242 The melanocortin-4 receptor (MC4R) is a member of the rho- ulated cAMP production. Confocal microscopy confirmed that dopsin-like G protein-coupled receptor family. The binding of the observed decreases in hormone binding by these mutants ␣-MSH to the MC4R leads to increased cAMP production. Re- are associated with decreased cell surface expression due to cent pharmacological and genetic studies have provided com- intracellular retention of the mutants. The other five allelic pelling evidence that MC4R is an important regulator of food variants (D37V, P48S, V50M, I170V, N274S) were found to be intake and energy homeostasis. Allelic variants of MC4R were expressed at the cell surface and to bind agonist and respond reported in some children with early-onset severe obesity. with increased cAMP production normally. The data on these However, few studies have been performed to confirm that latter five variants raise the question as to whether they are these allelic variants result in an impairment of the receptor’s indeed causative of the obesity or not and, if so, by what mech- function. In this study, we expressed wild-type and variant anism. Our data, therefore, stress the importance of charac- MC4Rs in HEK293 cells and systematically studied ligand terizing the properties of MC4R variants associated with binding, agonist-stimulated cAMP, and cell surface expres- early-onset severe obesity. -

Interaction Between the Spinal Melanocortin and Opioid Systems in a Rat Model of Neuropathic Pain Dorien H
Anesthesiology 2003; 99:449–54 © 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Interaction between the Spinal Melanocortin and Opioid Systems in a Rat Model of Neuropathic Pain Dorien H. Vrinten, M.D., Ph.D.,* Willem Hendrik Gispen, Ph.D.,† Cor J. Kalkman, M.D., Ph.D.,‡ Roger A. H. Adan, Ph.D.§ Background: The authors recently demonstrated that admin- substance P, calcitonin gene–related peptide, cholecys- istration of the melanocortin-4 receptor antagonist SHU9119 tokinin, and neuropeptide Y.6,7 We recently demon- decreased neuropathic pain symptoms in rats with a sciatic strated that such plasticity also occurs in the spinal chronic constriction injury. The authors hypothesised that there is a balance between tonic pronociceptive effects of the melanocortin system, as demonstrated by an up-regula- spinal melanocortin system and tonic antinociceptive effects of tion of melanocortin-4 (MC4) receptors in the spinal cord the spinal opioid system. Therefore, they investigated a possi- dorsal horn in a rat model for neuropathic pain, the Downloaded from http://pubs.asahq.org/anesthesiology/article-pdf/99/2/449/407606/0000542-200308000-00028.pdf by guest on 25 September 2021 ble interaction between these two systems and tested whether chronic constriction injury (CCI).8,9 Because melano- opioid effectiveness could be increased through modulation of cortins have been shown to induce hyperalgesia,10,11 the spinal melanocortin system activity. Methods: In chronic constriction injury rats, melanocortin this increase in spinal MC4 receptors might contribute to and opioid receptor ligands were administered through a lum- the increased sensitivity in neuropathic pain, through bar spinal catheter, and their effects on mechanical allodynia activation by the endogenous melanocortin receptor ag- were assessed by von Frey probing. -

G Protein-Coupled Receptors
S.P.H. Alexander et al. The Concise Guide to PHARMACOLOGY 2015/16: G protein-coupled receptors. British Journal of Pharmacology (2015) 172, 5744–5869 THE CONCISE GUIDE TO PHARMACOLOGY 2015/16: G protein-coupled receptors Stephen PH Alexander1, Anthony P Davenport2, Eamonn Kelly3, Neil Marrion3, John A Peters4, Helen E Benson5, Elena Faccenda5, Adam J Pawson5, Joanna L Sharman5, Christopher Southan5, Jamie A Davies5 and CGTP Collaborators 1School of Biomedical Sciences, University of Nottingham Medical School, Nottingham, NG7 2UH, UK, 2Clinical Pharmacology Unit, University of Cambridge, Cambridge, CB2 0QQ, UK, 3School of Physiology and Pharmacology, University of Bristol, Bristol, BS8 1TD, UK, 4Neuroscience Division, Medical Education Institute, Ninewells Hospital and Medical School, University of Dundee, Dundee, DD1 9SY, UK, 5Centre for Integrative Physiology, University of Edinburgh, Edinburgh, EH8 9XD, UK Abstract The Concise Guide to PHARMACOLOGY 2015/16 provides concise overviews of the key properties of over 1750 human drug targets with their pharmacology, plus links to an open access knowledgebase of drug targets and their ligands (www.guidetopharmacology.org), which provides more detailed views of target and ligand properties. The full contents can be found at http://onlinelibrary.wiley.com/doi/ 10.1111/bph.13348/full. G protein-coupled receptors are one of the eight major pharmacological targets into which the Guide is divided, with the others being: ligand-gated ion channels, voltage-gated ion channels, other ion channels, nuclear hormone receptors, catalytic receptors, enzymes and transporters. These are presented with nomenclature guidance and summary information on the best available pharmacological tools, alongside key references and suggestions for further reading. -

An Evolutionary Based Strategy for Predicting Rational Mutations in G Protein-Coupled Receptors
Ecology and Evolutionary Biology 2021; 6(3): 53-77 http://www.sciencepublishinggroup.com/j/eeb doi: 10.11648/j.eeb.20210603.11 ISSN: 2575-3789 (Print); ISSN: 2575-3762 (Online) An Evolutionary Based Strategy for Predicting Rational Mutations in G Protein-Coupled Receptors Miguel Angel Fuertes*, Carlos Alonso Department of Microbiology, Centre for Molecular Biology “Severo Ochoa”, Spanish National Research Council and Autonomous University, Madrid, Spain Email address: *Corresponding author To cite this article: Miguel Angel Fuertes, Carlos Alonso. An Evolutionary Based Strategy for Predicting Rational Mutations in G Protein-Coupled Receptors. Ecology and Evolutionary Biology. Vol. 6, No. 3, 2021, pp. 53-77. doi: 10.11648/j.eeb.20210603.11 Received: April 24, 2021; Accepted: May 11, 2021; Published: July 13, 2021 Abstract: Capturing conserved patterns in genes and proteins is important for inferring phenotype prediction and evolutionary analysis. The study is focused on the conserved patterns of the G protein-coupled receptors, an important superfamily of receptors. Olfactory receptors represent more than 2% of our genome and constitute the largest family of G protein-coupled receptors, a key class of drug targets. As no crystallographic structures are available, mechanistic studies rely on the use of molecular dynamic modelling combined with site-directed mutagenesis data. In this paper, we hypothesized that human-mouse orthologs coding for G protein-coupled receptors maintain, at speciation events, shared compositional structures independent, to some extent, of their percent identity as reveals a method based in the categorization of nucleotide triplets by their gross composition. The data support the consistency of the hypothesis, showing in ortholog G protein-coupled receptors the presence of emergent shared compositional structures preserved at speciation events. -

Melanocyte-Stimulating Hormones
The Journal of Neuroscience, August 15, 1996, 16(16):5182–5188 Melanocortin Antagonists Define Two Distinct Pathways of Cardiovascular Control by a- and g-Melanocyte-Stimulating Hormones Si-Jia Li,1 Ka´ roly Varga,1 Phillip Archer,1 Victor J. Hruby,2 Shubh D. Sharma,2 Robert A. Kesterson,3 Roger D. Cone,3 and George Kunos1 1Department of Pharmacology and Toxicology, Virginia Commonwealth University, Richmond, Virginia 23298-0613, 2Department of Chemistry, University of Arizona, Tucson, Arizona 85721, and 3Vollum Institute, Oregon Health Sciences University, Portland, Oregon 97210 Melanocortin peptides and at least two subtypes of melano- intracarotid than after intravenous administration. The effects cortin receptors (MC3-R and MC4-R) are present in brain of g-MSH (1.25 nmol) are not inhibited by the intracarotid regions involved in cardiovascular regulation. In urethane- injection of SHU9119 (1.25–12.5 nmol) or the novel MC3-R anesthetized rats, unilateral microinjection of a-melanocyte- antagonist SHU9005 (1.25–12.5 nmol). We conclude that the stimulating hormone (MSH) into the medullary dorsal–vagal hypotension and bradycardia elicited by the release of complex (DVC) causes dose-dependent (125–250 pmol) hy- a-MSH from arcuate neurons is mediated by neural melano- potension and bradycardia, whereas g-MSH is less effective. cortin receptors (MC4-R/MC3-R) located in the DVC, The effects of a-MSH are inhibited by microinjection to the whereas the similar effects of b-endorphin, a peptide derived same site of the novel MC4-R/MC3-R antagonist SHU9119 from the same precursor, are mediated by opiate receptors (2–100 pmol) but not naloxone (270 pmol), whereas the at the same site. -

Sustained Wash-Resistant Receptor Activation Responses of GPR119 Agonists
European Journal of Pharmacology 762 (2015) 430–442 Contents lists available at ScienceDirect European Journal of Pharmacology journal homepage: www.elsevier.com/locate/ejphar Molecular and cellular pharmacology Sustained wash-resistant receptor activation responses of GPR119 agonists J. Daniel Hothersall a,n, Charlotte E. Bussey a, Alastair J. Brown a,b, James S. Scott a, Ian Dale c, Philip Rawlins c a AstraZeneca, Alderley Park, Macclesfield SK10 4TG, UK b Heptares Therapeutics Limited, Welwyn Garden City AL7 3AX, UK c AstraZeneca, Cambridge Science Park, Cambridge CB4 0WG, UK article info abstract Article history: G protein-coupled receptor 119 (GPR119) is involved in regulating metabolic homoeostasis, with GPR119 Received 24 February 2015 agonists targeted for the treatment of type-2 diabetes and obesity. Using the endogenous agonist Received in revised form oleoylethanolamide and a number of small molecule synthetic agonists we have investigated the tem- 15 June 2015 poral dynamics of receptor signalling. Using both a dynamic luminescence biosensor-based assay and an Accepted 16 June 2015 endpoint cAMP accumulation assay we show that agonist-driven desensitization is not a major reg- Available online 20 June 2015 ulatory mechanism for GPR119 despite robust activation responses, regardless of the agonist used. Keywords: Temporal analysis of the cAMP responses demonstrated sustained signalling resistant to washout for GPR119 some, but not all of the agonists tested. Further analysis indicated that the sustained effects of one Agonist synthetic agonist AR-231,453 were consistent with a role for slow dissociation kinetics. In contrast, the Desensitization sustained responses to MBX-2982 and AZ1 appeared to involve membrane deposition. -

Melanocortin-4 Receptor: a Novel Signalling Pathway Involved in Body Weight Regulation
International Journal of Obesity (1999) 23, Suppl 1, 54±58 ß 1999 Stockton Press All rights reserved 0307±0565/99 $12.00 http://www.stockton-press.co.uk/ijo Melanocortin-4 receptor: A novel signalling pathway involved in body weight regulation SL Fisher1, KA Yagaloff1 and P Burn1* 1Department of Metabolic Diseases, Hoffmann LaRoche, Nutley, NJ 07110, USA For many years, genetically obese mouse strains have provided models for human obesity. The Avy=-agouti mouse, one of the oldest obese mouse models, is characterized by maturity-onset obesity and diabetes as a result of ectopic expression of the secreted protein hormone, agouti protein. Agouti protein is normally expressed in hair follicles to regulate pigmentation through antagonism of the melanocortin-1 receptor, but in-vitro studies have demonstrated that the hormone also has potent antagonist activity for the melanocortin-4 receptor (MC4-R). Subsequent develop- ment of the MC4-R knockout mouse model demonstrated that MC4-R plays a role in weight homeostasis as these mice recapitulated the metabolic defects of the agouti mouse. Further evidence for this hypothesis was obtained from pharmacological studies utilizing peptides with MC4-R agonist activity, that inhibitied food intake (when administered intracerebrally). Additional studies with peptide antagonists have now implicated the MC4-R in the leptin signalling pathway. Finally, evidence that the MC4-R may play a role in human obesity has been obtained from the identi®cation of a dis-functional variant of the receptor in genetically obese subjects. Keywords: obesity; diabetes; agouti; melanocortin; POMC; leptin; ob Introduction There has been an explosion in obesity research and with this has come an understanding of the molecular mechanisms that underly the disease. -

Distinct Actions of Ancestral Vinclozolin and Juvenile Stress on Neural Gene Expression in the Male Rat
ORIGINAL RESEARCH ARTICLE published: 02 March 2015 doi: 10.3389/fgene.2015.00056 Distinct actions of ancestral vinclozolin and juvenile stress on neural gene expression in the male rat Ross Gillette1, Isaac Miller-Crews1, Michael K. Skinner 2 and David Crews1,3 * 1 Institute for Cellular and Molecular Biology, The University of Texas at Austin, Austin, TX, USA 2 Center for Reproductive Biology, School of Biological Sciences, Washington State University, Pullman, WA, USA 3 Department of Integrative Biology, The University of Texas at Austin, Austin, TX, USA Edited by: Exposure to the endocrine disrupting chemical vinclozolin during gestation of an F0 Douglas Mark Ruden, Wayne State generation and/or chronic restraint stress during adolescence of the F3 descendants affects University, USA behavior, physiology, and gene expression in the brain. Genes related to the networks Reviewed by: of growth factors, signaling peptides, and receptors, steroid hormone receptors and Eberhard Weihe, University of Marburg, Germany enzymes, and epigenetic related factors were measured using quantitative polymerase Alice Hudder, Lake Erie College of chain reaction via Taqman low density arrays targeting 48 genes in the central amygdaloid Osteopathic Medicine, USA nucleus, medial amygdaloid nucleus, medial preoptic area (mPOA), lateral hypothalamus *Correspondence: (LH), and the ventromedial nucleus of the hypothalamus. We found that growth factors are David Crews, Department of particularly vulnerable to ancestral exposure in the central and medial amygdala; restraint Integrative Biology, The University of Texas at Austin, 2405 Speedway, stress during adolescence affected neural growth factors in the medial amygdala. Signaling Austin, TX 78712, USA peptides were affected by both ancestral exposure and stress during adolescence primarily e-mail: [email protected] in hypothalamic nuclei. -

New Insights Into Metabolic Homeostasis Keith Tan
Rockefeller University Digital Commons @ RU Student Theses and Dissertations 2015 Activity Based Profiling: New Insights into Metabolic Homeostasis Keith Tan Follow this and additional works at: http://digitalcommons.rockefeller.edu/ student_theses_and_dissertations Part of the Life Sciences Commons Recommended Citation Tan, Keith, "Activity Based Profiling: New Insights into Metabolic Homeostasis" (2015). Student Theses and Dissertations. Paper 285. This Thesis is brought to you for free and open access by Digital Commons @ RU. It has been accepted for inclusion in Student Theses and Dissertations by an authorized administrator of Digital Commons @ RU. For more information, please contact [email protected]. ACTIVITY BASED PROFILING: NEW INSIGHTS INTO METABOLIC HOMEOSTASIS A Thesis Presented to the Faculty of The Rockefeller University in Partial Fulfillment of the Requirements for the degree of Doctor of Philosophy by Keith Tan June 2015 © Copyright by Keith Tan 2015 ACTIVITY BASED PROFILING: NEW INSIGHTS INTO METABOLIC HOMEOSTASIS Keith Tan, Ph.D. The Rockefeller University 2015 There is mounting evidence that demonstrates that body weight and energy homeostasis is tightly regulated by a physiological system. This system consists of sensing and effector components that primarily reside in the central nervous system and disruption to these components can lead to obesity and metabolic disorders. Although many neural substrates have been identified in the past decades, there is reason to believe that there are numerous unidentified neural populations that play a role in energy balance. Besides regulating caloric consumption and energy expenditure, neural components that control energy homeostasis are also tightly intertwined with circadian rhythmicity but this aspect has received less attention.