Active Components in Leaves of Rhus Chinensis Mill THERMAL SCIENCE: Year 2020, Vol
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Study on the Soil and Water Conservation Effect of Larix Kaernpferi (Sieb
12th ISCO Conference Beijing 2002 A Study on the Soil and Water Conservation Effect of Larix kaernpferi (Sieb. et Zucc.) Forest Zhao Yong Li Shuren and Wu Mingzuo Environment Department, Henan Agriculture University, Zhengzhou, Henan Province E-mail: [email protected] Abstract: The direct runoff and soil erosion was observed in the Larix kaernpferi forest that was man planted with two different site preparation methods for contrasting. The results showed that the hole or strip site preparation methods has obvious effects to decrease the direct runoff and soil erosion. The hole site preparation method has the best effect on decreasing direct runoff by 34% and soil erosion by 42%. The two different site preparation methods also had obvious effects on decreasing the washing of nutrient element by 79.75kg hm–2 a–1— 135.40kg hm–2 a–1. Keywords: soil and water conservation, site preparation, direct runoff, soil nutrient, Larix kaernpferi Forest can not only affect soil physic and chemistry traits, but also regulate rainfall’s distribution and water flow process. The study of forest ecology now put much emphasis on the forest effects on decreasing soil erosion and the change of soil characteristics. The study on these effects of man-planted Larix kaernpferi forest using two different site preparation methods was carried on in 1993—1995 in Longyuwan forestry center in Luanchuan county, Henan province. The amount of direct runoff, soil erosion and soil nutrient elements were observed in the fixed field. 1 Study site outlines The study site is lying in the Longyuwan forestry center in Luanchuan county, Henan province, locating at 111 35 E and 33 45 . -

(12) Patent Application Publication (10) Pub. No.: US 2014/0161919 A1 Thangavel Et Al
US 2014O161919A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2014/0161919 A1 Thangavel et al. (43) Pub. Date: Jun. 12, 2014 (54) PLANT PARTS AND EXTRACTS HAVING Publication Classification ANTICOCCDAL ACTIVITY (51) Int. C. (71) Applicant: Kemin Industries, Inc., Des Moines, IA A61E36/49 (2006.01) (US) A61E36/85 (2006.01) A61E36/22 (2006.01) (72) Inventors: Gokila Thangavel, Hosur (IN); (52) U.S. C. Rajalekshmi Mukkalil, Cochin (IN); CPC ................. A61K 36/49 (2013.01); A61K 36/22 Haridasan Chirakkal, Nolambur (IN); (2013.01); A61K 36/185 (2013.01) Hannah Kurian, Benson Town (IN) USPC ........................................... 424/769; 424/725 (21) Appl. No.: 13/928,504 (57) ABSTRACT (22) Filed: Jun. 27, 2013 Natural plant parts and extracts of plants selected from the Related U.S. Application Data group consisting of Quercus infectoria, Rhus chinensis and (60) Provisional application No. 61/664,795, filed on Jun. Terminalia chebula containing compounds such as gallic 27, 2012. acid, derivative of gallic acid, gallotannins and hydrolysable tannins have been found to control coccidiosis in poultry and, (30) Foreign Application Priority Data more specifically, coccidiosis caused by Eimeria spp. The plant parts and natural extracts result in a reduction of lesion Jan. 23, 2013 (IN) ............................. 177/DELA2013 score, oocysts per gram of fecal matter and mortality. Patent Application Publication Jun. 12, 2014 Sheet 1 of 14 US 2014/O161919 A1 Positive control S Negative control Positive Control Quercus infectoria Patent Application Publication Jun. 12, 2014 Sheet 2 of 14 US 2014/O161919 A1 as a 3. Q3 is niecifia FIG. 3 3 9. -

Reorganization and Corporate Structure
REORGANIZATION AND CORPORATE STRUCTURE History and Development We were established on December 22, 1999 under the name of LLMG, as a limited liability company wholly-owned by the People’s Government of Luanchuan County (which held the interest on behalf of the People’s Government of Luoyang City) following the approval of the merger of two enterprises, namely, LLMC and LCMCC on August 18, 1998 by the People’s Government of Luoyang City. Our registered capital was RMB251 million upon establishment. Immediately prior to the merger, both LLMC and LCMCC were principally engaged in mining, flotation, roasting, smelting and sale of molybdenum products. Following the establishment of LLMG, we expanded our open-pit mining operations. Our ore production reached 8,000 tpd by the end of 2001. In 2002, Luomu Group Refining commenced its operations to produce molybdenum oxide and ferromolybdenum. To accommodate the mining expansion, we doubled our daily ore processing capacity from 8.0 Kt in 2002 to approximately 15.0 Kt by the end of 2004. As part of our corporate restructuring, we disposed of some of our businesses during the years 2003 and 2004 to streamline our business operations. In September 2004, after receiving the approvals issued by the People’s Government of Luanchuan County approving the reorganization of LLMG and CFCH to subscribe shares in LLMG, we entered into an agreement with CFCH (‘‘Subscription Agreement’’) and increased our registered capital to RMB280,020,000. Luoyang SASAC reconfirmed our 2004 reorganization by ‘‘The letter on the reorganization of Luoyang Luanchuan Molybdenum Group Co., Ltd in 2004’’ in November 2006 and ‘‘The confirmation letter on the reorganization and capital increase of China Molybdenum Co., Ltd.’’ in January 2007. -

Medicinal Practices of Sacred Natural Sites: a Socio-Religious Approach for Successful Implementation of Primary
Medicinal practices of sacred natural sites: a socio-religious approach for successful implementation of primary healthcare services Rajasri Ray and Avik Ray Review Correspondence Abstract Rajasri Ray*, Avik Ray Centre for studies in Ethnobiology, Biodiversity and Background: Sacred groves are model systems that Sustainability (CEiBa), Malda - 732103, West have the potential to contribute to rural healthcare Bengal, India owing to their medicinal floral diversity and strong social acceptance. *Corresponding Author: Rajasri Ray; [email protected] Methods: We examined this idea employing ethnomedicinal plants and their application Ethnobotany Research & Applications documented from sacred groves across India. A total 20:34 (2020) of 65 published documents were shortlisted for the Key words: AYUSH; Ethnomedicine; Medicinal plant; preparation of database and statistical analysis. Sacred grove; Spatial fidelity; Tropical diseases Standard ethnobotanical indices and mapping were used to capture the current trend. Background Results: A total of 1247 species from 152 families Human-nature interaction has been long entwined in has been documented for use against eighteen the history of humanity. Apart from deriving natural categories of diseases common in tropical and sub- resources, humans have a deep rooted tradition of tropical landscapes. Though the reported species venerating nature which is extensively observed are clustered around a few widely distributed across continents (Verschuuren 2010). The tradition families, 71% of them are uniquely represented from has attracted attention of researchers and policy- any single biogeographic region. The use of multiple makers for its impact on local ecological and socio- species in treating an ailment, high use value of the economic dynamics. Ethnomedicine that emanated popular plants, and cross-community similarity in from this tradition, deals health issues with nature- disease treatment reflects rich community wisdom to derived resources. -

Gallnuts: a Potential Treasure in Anticancer Drug Discovery
Hindawi Evidence-Based Complementary and Alternative Medicine Volume 2018, Article ID 4930371, 9 pages https://doi.org/10.1155/2018/4930371 Review Article Gallnuts: A Potential Treasure in Anticancer Drug Discovery Jiayu Gao ,1 Xiao Yang ,2 Weiping Yin ,1 and Ming Li3 1 School of Chemical and Pharmaceutical Engineering, Henan University of Scientifc and Technology, Henan, China 2School of Clinical Medicine, Henan University of Scientifc and Technology, Henan, China 3Luoyang Traditional Chinese Medicine Association, Luoyang, Henan, China Correspondence should be addressed to Jiayu Gao; [email protected] Received 8 September 2017; Revised 17 February 2018; Accepted 21 February 2018; Published 29 March 2018 Academic Editor: Chris Zaslawski Copyright © 2018 Jiayu Gao et al. Tis is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Introduction. In the discovery of more potent and selective anticancer drugs, the research continually expands and explores new bioactive metabolites coming from diferent natural sources. Gallnuts are a group of very special natural products formed through parasitic interaction between plants and insects. Tough it has been traditionally used as a source of drugs for the treatment of cancerous diseases in traditional and folk medicinal systems through centuries, the anticancer properties of gallnuts are barely systematically reviewed. Objective. To evidence the traditional uses and phytochemicals and pharmacological mechanisms in anticancer aspects of gallnuts, a literature review was performed. Materials and Methods. Te systematic review approach consisted of searching web-based scientifc databases including PubMed, Web of Science, and Science Direct. -

Large-Scale Screening of 239 Traditional Chinese Medicinal Plant Extracts for Their Antibacterial Activities Against Multidrug-R
pathogens Article Large-Scale Screening of 239 Traditional Chinese Medicinal Plant Extracts for Their Antibacterial Activities against Multidrug-Resistant Staphylococcus aureus and Cytotoxic Activities Gowoon Kim 1, Ren-You Gan 1,2,* , Dan Zhang 1, Arakkaveettil Kabeer Farha 1, Olivier Habimana 3, Vuyo Mavumengwana 4 , Hua-Bin Li 5 , Xiao-Hong Wang 6 and Harold Corke 1,* 1 Department of Food Science & Technology, School of Agriculture and Biology, Shanghai Jiao Tong University, Shanghai 200240, China; [email protected] (G.K.); [email protected] (D.Z.); [email protected] (A.K.F.) 2 Research Center for Plants and Human Health, Institute of Urban Agriculture, Chinese Academy of Agricultural Sciences, Chengdu 610213, China 3 School of Biological Sciences, The University of Hong Kong, Hong Kong 999077, China; [email protected] 4 DST/NRF Centre of Excellence for Biomedical Tuberculosis Research, US/SAMRC Centre for Tuberculosis Research, Division of Molecular Biology and Human Genetics, Department of Biomedical Sciences, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town 8000, South Africa; [email protected] 5 Guangdong Provincial Key Laboratory of Food, Nutrition and Health, Department of Nutrition, School of Public Health, Sun Yat-Sen University, Guangzhou 510080, China; [email protected] 6 College of Food Science and Technology, Huazhong Agricultural University, Wuhan 430070, China; [email protected] * Correspondence: [email protected] (R.-Y.G.); [email protected] (H.C.) Received: 3 February 2020; Accepted: 29 February 2020; Published: 4 March 2020 Abstract: Novel alternative antibacterial compounds have been persistently explored from plants as natural sources to overcome antibiotic resistance leading to serious foodborne bacterial illnesses. -

Macro- and Microscopic Analyses of Anatomical Structures of Chinese
www.nature.com/scientificreports OPEN Macro- and Microscopic Analyses of Anatomical Structures of Chinese Gallnuts and Their Functional Received: 11 September 2018 Accepted: 14 March 2019 Adaptation Published: xx xx xxxx Qin Lu1, Hang Chen 1,2, Chao Wang1,3, Zi-xiang Yang1, Pin Lü1, Ming-shun Chen4 & Xiao-ming Chen1,2 The galls induced by Schlechtendaia chinensis, Schlechtendaia peitan and Nurudea shiraii on Rhus chinensis and gall induced by Kaburagia rhusicola rhusicola on Rhus potaninii Maxim. are the largest plant galls and have great economic and medical values. We examined the structures of galls and their functional adaptation using various macro- and microscopic techniques. The highly adapted structures include a stalk at the base that is specialized for mechanical support and transport of nutrients for aphids, and a network of vascular bundles which accompanying schizogenous ducts arranged in a way to best support aphid feeding and population growth. There are many circular and semicircular xylems traces in an ensiform gall in cross sectional views, which would provide more nutrition and occupy less space. We infer the evolution trail was fower-like gall, horned gall, circular gall and ensiform gall. And the possible evolutionary trend of the gall was bigger chamber, more stable mechanical supporting, easier for exchanging substance and transporting nutrients. Galls are abnormal outgrowths of plant tissues induced by gall-inducing organisms, which included various par- asitic insects and mites. It is estimated that there are ~4700 diferent species that can induce gall formation under certain conditions1. Te induction of galls is believed to be a result of plant manipulation by gall-inducing agents, and because of this, galls are generally considered as extended structures of the gallicolous organisms2–5. -

Assessment of Nutrient Composition and Antioxidant Activity of Some Popular Underutilized Edible Crops of Nagaland, India
Natural Resources, 2021, 12, 44-58 https://www.scirp.org/journal/nr ISSN Online: 2158-7086 ISSN Print: 2158-706X Assessment of Nutrient Composition and Antioxidant Activity of Some Popular Underutilized Edible Crops of Nagaland, India Chitta Ranjan Deb* , Neilazonuo Khruomo Department of Botany, Nagaland University, Lumami, India How to cite this paper: Deb, C.R. and Abstract Khruomo, N. (2021) Assessment of Nu- trient Composition and Antioxidant Activ- In Nagaland ~70% of population lives in rural areas and depends on forest ity of Some Popular Underutilized Edible products for livelihood. Being part of the biodiversity hotspot, state is rich in Crops of Nagaland, India. Natural Resources, biodiversity. The present study was an attempt made to understand the nutri- 12, 44-58. https://doi.org/10.4236/nr.2021.122005 tional properties of 22 popular underutilized edible plants (UEP) Kohima, Phek, Tuensang districts. Results revealed moisture content of 22 studied Received: December 25, 2020 plants ranged between 4.8 to 88.15 g/100g, while protein content varied be- Accepted: February 23, 2021 tween 0.00269 - 0.773 g/100g with highest in Terminalia chebula (0.773 g/100g) Published: February 26, 2021 fruit while lowest protein content was in Setaria italica (0.00269 g/100g). To- Copyright © 2021 by author(s) and tal carbohydrate content was between 0.198 - 5.212 g/100g with highest in Scientific Research Publishing Inc. Setaria italica (5.212 g/100g) and lowest in Juglans regia (0.198 g/100g). Of This work is licensed under the Creative the 22 samples, maximum antioxidant activity was in Terminalia chebula fruits Commons Attribution International License (CC BY 4.0). -

Here Discharged Water Is Treated with Limewater, Purification Agents, and Five-Stage Sedimentation, Bringing It up to Chinese National Standards
希尔威金属矿业有限公司 2 0 2 0 可持续发展报告 SILVERCORP METALS INC. Fiscal 2020 Sustainability Report About This Report About This Report Time Period This report mainly focuses on our Fiscal Year 2020 (April 1, 2019 to March 31, 2020), and may refer to information of other years in order to strengthen the comparison of statistics. Entities Covered in this Report This report covers the headquarters and subsidiaries of Silvercorp Metals Inc. For convenience of expression and simplicity, Silvercorp Metals Inc. is also referred to as Silvercorp, the Company, or we. Its subsidiaries, Henan Found Mining Co. Ltd. and Guangdong Found Mining Co. Ltd., are also referred to as Henan Found and Guangdong Found respectively. Data This annual report is the first such report issued by the Company, and the information provided has not yet been verified by an external auditor. This report aims at reflecting the economic, environmental and social performance of the Company. Reference Standards This report is prepared based on the Global Reporting Initiatives (GRI) Standards: Core option, the Guide on Preparation of Corporate Social Responsibility Reports of Chinese Enterprises (CASS-CSR 4.0) and the Guide on Preparation of Corporate Social Responsibility Reports of Chinese Enterprises (CASS-CSR 4.0) - Mining Industry, both published by the Chinese Academy of Social Sciences, and the Guide for Business Action on SDGs by the UN Global Compact. Availability This report is available in both printed copies and an electronic version available on our official website. Requests for printed copies of this report should be addressed to Silvercorp Metals Inc. Address: Suite 1750-1066 W. -
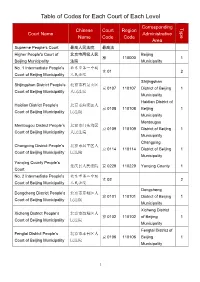
Table of Codes for Each Court of Each Level
Table of Codes for Each Court of Each Level Corresponding Type Chinese Court Region Court Name Administrative Name Code Code Area Supreme People’s Court 最高人民法院 最高法 Higher People's Court of 北京市高级人民 Beijing 京 110000 1 Beijing Municipality 法院 Municipality No. 1 Intermediate People's 北京市第一中级 京 01 2 Court of Beijing Municipality 人民法院 Shijingshan Shijingshan District People’s 北京市石景山区 京 0107 110107 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Haidian District of Haidian District People’s 北京市海淀区人 京 0108 110108 Beijing 1 Court of Beijing Municipality 民法院 Municipality Mentougou Mentougou District People’s 北京市门头沟区 京 0109 110109 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Changping Changping District People’s 北京市昌平区人 京 0114 110114 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Yanqing County People’s 延庆县人民法院 京 0229 110229 Yanqing County 1 Court No. 2 Intermediate People's 北京市第二中级 京 02 2 Court of Beijing Municipality 人民法院 Dongcheng Dongcheng District People’s 北京市东城区人 京 0101 110101 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Xicheng District Xicheng District People’s 北京市西城区人 京 0102 110102 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Fengtai District of Fengtai District People’s 北京市丰台区人 京 0106 110106 Beijing 1 Court of Beijing Municipality 民法院 Municipality 1 Fangshan District Fangshan District People’s 北京市房山区人 京 0111 110111 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Daxing District of Daxing District People’s 北京市大兴区人 京 0115 -

Medicinal Plants Used in the Treatment of Human Immunodeficiency Virus
International Journal of Molecular Sciences Review Medicinal Plants Used in the Treatment of Human Immunodeficiency Virus Bahare Salehi 1,2 ID , Nanjangud V. Anil Kumar 3 ID , Bilge ¸Sener 4, Mehdi Sharifi-Rad 5,*, Mehtap Kılıç 4, Gail B. Mahady 6, Sanja Vlaisavljevic 7, Marcello Iriti 8,* ID , Farzad Kobarfard 9,10, William N. Setzer 11,*, Seyed Abdulmajid Ayatollahi 9,12,13, Athar Ata 13 and Javad Sharifi-Rad 9,13,* ID 1 Medical Ethics and Law Research Center, Shahid Beheshti University of Medical Sciences, 88777539 Tehran, Iran; [email protected] 2 Student Research Committee, Shahid Beheshti University of Medical Sciences, 22439789 Tehran, Iran 3 Department of Chemistry, Manipal Institute of Technology, Manipal University, Manipal 576104, India; [email protected] 4 Department of Pharmacognosy, Gazi University, Faculty of Pharmacy, 06330 Ankara, Turkey; [email protected] (B.¸S.);[email protected] (M.K.) 5 Department of Medical Parasitology, Zabol University of Medical Sciences, 61663-335 Zabol, Iran 6 PAHO/WHO Collaborating Centre for Traditional Medicine, College of Pharmacy, University of Illinois, 833 S. Wood St., Chicago, IL 60612, USA; [email protected] 7 Department of Chemistry, Biochemistry and Environmental Protection, Faculty of Sciences, University of Novi Sad, Trg Dositeja Obradovica 3, 21000 Novi Sad, Serbia; [email protected] 8 Department of Agricultural and Environmental Sciences, Milan State University, 20133 Milan, Italy 9 Phytochemistry Research Center, Shahid Beheshti University of -

Study on Evaluation of Ecological Environment Quality and Temporal-Spatial Evolution of Danjiang River Basin (Henan Section)
Pol. J. Environ. Stud. Vol. 30, No. 3 (2021), 2353-2367 DOI: 10.15244/pjoes/128110 ONLINE PUBLICATION DATE: 2021-02-10 Original Research Study on Evaluation of Ecological Environment Quality and Temporal-Spatial Evolution of Danjiang River Basin (Henan Section) Shi-Dong Wang, Jin-Jin Si*, Yong Wang School of Surveying and Land Information Engineering, Henan Polytechnic University, Jiaozuo, Henan 454000, P.R. China Received: 28 April 2020 Accepted: 3 October 2020 Abstract The Danjiang River Basin (Henan Section) is the water source of the Middle Route of the South-to- North Water Transfer Project. Its ecological environment quality has received increasing attention. In this study, on the basis of the ecological environment of the study area, we constructed the evaluation index system from four aspects: ecological basis, ecological structure, ecological stress and ecological benefits. The variable fuzzy set model was improved by using the central point triangle whitening weight function and the relative difference function. The ecological environment quality of the Danjiang River Basin (Henan Section) in the past 20 years was evaluated, and the evolution patterns and causes of ecological environment quality were analyzed by combination with land use changing. And the results showed that the area with better ecological environment quality was significantly larger than the deterioration area. The area with poorer grade was decreased from 441.22 km2 in 1998 to 3.93 km2 in 2017, and its proportion was correspondingly decreased from 2.23% to 0.05%. The area with the ordinary grade was decreased from 4528.79 km2 in 1998 to 2636.47 km2 in 2017, and its proportion was correspondingly decreased from 53.67% to 31.24%; The moderately graded area was increased from 3132.7 km2 in 1998 to 5206.88 km2 in 2017, and its proportion was increased from 37.12% to 61.71%; the area with good grade was increased from 319.58 km2 in 1998 to 579.01 km2 in 2017, the area with the excellent grade of an evaluation rating was almost unchanged.