MAY 2021 MAY Total 86 Pages Total …….Including Broadband & IPTV …….Including Ithout Prepayment; WPP License No
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Accsiti Family Pack-220 Ne
ACCSITI FAMILY PACK-220 NE Language Sub Genre Channel Name DD Channel DD Channel ALL DD Channels Assamese Gec GYAN BRIKSHA|INDRADHANU |JONACK|PRAG CINEMA|RAMDHENU|RANG|RENGONI Assamese ASSAM TALKS|DY 365|NE NEWS|NEWS LIVE|NEWS18 ASSAM/NE|NEWS18 ASSAM/NORTH Assamese News EAST|NORTH EAST|PRAG NEWS|PROTIDIN TIME Bengali Gec AAKASH AATH|CTVN AKD PLUS|RUPASHI BANGLA Bengali Bengali Movies/Music DHOOM MUSIC|KHUSHBOO TV|SANGEET BANGLA Bengali News ABP ANANDA|BANGLA TIMES|CALCUTTA NEWS|NEWS TIME BANGLA Bhojpuri Regional Hindi B4U BHOJPURI|BHOJPURI CINEMA|DANGAL|OSCAR MOVIES Devotional GOD TV|PEACE OF MIND TV English Music / Gec HI DOSTI|VH-1 English International ARIRANG TV|CHANNEL NEWS ASIA|DW TV|NEPAL 1|NHK WORLD|RUSSIA TODAY|TV5 MONDE AASTHA|AASTHA BHAJAN|ARIHANT|BHAKTI SAGAR 2|DISHA TV|DIVYA|GRACE TV|HARE Devotional KRISHNA TV|ISHWAR BHAKTI TV|JINVANI|KAATYAYANI|LORD BHDDHA TV|PARAS TV|SANSKAR TV|SATSANG|SHUBH TV Hindi Gec COLORS|COLORS RISHTEY|DD NATIONAL|E24|MANORANJAN GRAND|ZEE ANMOL|ZOOM B4U KADAK|B4U MOVIES|BFLIX MOVIES|BOX CINEMA|COLORS CINEPLEX|ENTERR 10|HOUSE Hindi Hindi Movies FULL MOVIES|MAHA MOVIES|MANORANJAN TV|MANORANJANA MOVIES|MAX 2|MOVIE PLUS|STAR GOLD 2|SURYA CINEMA|UTV ACTION|UTV MOVIES|ZEE ACTION|ZEE BOLLYWOOD 9X JALWA|9X JHAKAAS|9X MUSIC|B4U MUSIC|INSYNC|M TUNES +|MASTIII|MTV|MTV Hindi Music BEATS|MUSIC INDIA|WOW MUSIC|ZING ANJAN TV|DABAANG|NEWS18 BIHAR/JHARKHAND|NEWS18 MADHYA Regional Hindi PRADESH/CHHATTISGARH|NEWS18 PUNJAB/HARYANA/HIMACHAL PRADESH|NEWS18 UTTAR PRADESH/UTTARAKHAND|SANGEET BHOJPURI ANIMAL PLANET|CARE -

1988 Environmental Report
Fermilab 891’63 @ Fermi National Accelerator Laboratory 1104.100 P.O. Box 500, Batavia, Illinois 60510 UC-41 Site Environmental Report For Calendar Year 1988 May 1,1989 Samuel I. Baker Operated by Universities Research As%xiation Inc. Under Contract with the United Stales Department of Energy. 7we Chlcago_’ Operations Office. Batavia Area Ottice Fermi National Accelerator Laboratory Fermilab 89/63 P. 0. Box 500, Batavia, Illinois 60510 1104.100 UC-41 SITE ENVIRONMENTAL REPORT FOR CALENDAR YEAR 1988 by Samuel I. Baker Hay 1. 1989 Laboratory Work and Measurements by R. L. Allen, S. I. Baker, S. Butala, J. H. Baldwin. V. R. Cupps. A. Elwyn. W. S. Freeman, D. W. Grobe, C. Salsbury, Y. Thomas, and P. Yurista Operated by Universities Research Association, Inc. Under Contract vith the United States Department of Energy, Chicago Operations Office, Batavia Area Office TABLE OF CONTENT8 1. Introduction ......................................................... 1 2. Summary .............................................................. 7 3. Environmental Program Information......: ............................ 10 3. 1 Environmental Program Description............... ............. 10 3. 2 Summary of Environmental Monitoring Performed in CY-1988 ...... 11 3. 3 Environmental Permits........................... ............. 13 3. 4 Assessments and Impact Statements............... ............. 16 3. 5 Summary of Significant Environmental Activities. ............. 16 3. 6 Summary of Hydrogeology......................... ............. 23 4. Environmental -

Assets.Kpmg › Content › Dam › Kpmg › Pdf › 2012 › 05 › Report-2012.Pdf
Digitization of theatr Digital DawnSmar Tablets tphones Online applications The metamorphosis kingSmar Mobile payments or tphones Digital monetizationbegins Smartphones Digital cable FICCI-KPMG es Indian MeNicdia anhed E nconttertainmentent Tablets Social netw Mobile advertisingTablets HighIndus tdefinitionry Report 2012 E-books Tablets Smartphones Expansion of tier 2 and 3 cities 3D exhibition Digital cable Portals Home Video Pay TV Portals Online applications Social networkingDigitization of theatres Vernacular content Mobile advertising Mobile payments Console gaming Viral Digitization of theatres Tablets Mobile gaming marketing Growing sequels Digital cable Social networking Niche content Digital Rights Management Digital cable Regionalisation Advergaming DTH Mobile gamingSmartphones High definition Advergaming Mobile payments 3D exhibition Digital cable Smartphones Tablets Home Video Expansion of tier 2 and 3 cities Vernacular content Portals Mobile advertising Social networking Mobile advertising Social networking Tablets Digital cable Online applicationsDTH Tablets Growing sequels Micropayment Pay TV Niche content Portals Mobile payments Digital cable Console gaming Digital monetization DigitizationDTH Mobile gaming Smartphones E-books Smartphones Expansion of tier 2 and 3 cities Mobile advertising Mobile gaming Pay TV Digitization of theatres Mobile gamingDTHConsole gaming E-books Mobile advertising RegionalisationTablets Online applications Digital cable E-books Regionalisation Home Video Console gaming Pay TVOnline applications -
Broadcasting Ii Aug 1
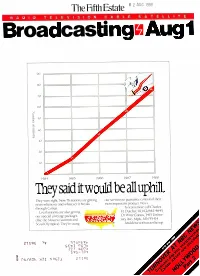
0 2 AUG 1983 The Fifth Estate B E S A T E L L I T E R A D I O T E L E V I S I O N C A L Broadcasting ii Aug 1 90 80 70 (i(1 50 40 30 20 I0 1984 1987 1988 They said it would be all uphill, u rOI their They were right. Now 76 stations arc getting our services to guarantee c of news whenever and wherever it breaks most important product. News. through Conus. To learn more call Charles -4645. Local stations are also getting H. Dutcher, Ill, 612/642 our special coverage packages ;- Or Write Conus, 3415 Univer- , 55414. (like the Moscow summit and sity Ave., Mpls,. MN to the top. Seoul Olympics). They're using e And drive with us ,5re` 2119£ 1 1lF.XVh SCbi 0 ZZT 1100b Q SdS-1ftV 06/AON )IZI Q£Z.1 Zii0£ a CtoFae ,5Q' rates Ni DUCKTALES # 1 KIDS PROGRAM MAY 88 KIDS PROGRAM HH RTG KIDS 2-11 RTG KIDS 6-11 RTG DUCKTALES 4.5 12.9 13.3 DOUBLE DARE 3.7 9.8 11.5 REAL GHOSTBUSTERS 2.8 7.2 6.9 DENNIS THE MENACE 2.7 7.4 7.3 JEM 2.2 5.9 5.7 JETSONS 2.1 5.5 6.1 SMURFS 1.9 5.1 4.2 FLINTSTONES 1.9 4.4 4.1 MY LITTLE PONY 1.8 5.1 2.9 SCOOBY D00 1.7 4.7 3.9 G.I. JOE 1.7 4.2 4.7 Source: Cassandra Tracking Report May 88 tNOON F pest afternoon time pere: The numbers tell the tale: DUCKTALES increases its lead -in in over 90% of all markets. -

Declaration Under Section 4 (4) of the Telecommunication (Broadcasting and Cable) Services Interconnection (Addressable System) Regulation, 2017 (No
Version 1.0/2019 Declaration Under Section 4 (4) of The Telecommunication (Broadcasting and Cable) Services Interconnection (Addressable System) Regulation, 2017 (No. 1 of 2017) 4(4)a: Target Market Distribution Network Location States/Parts of State covered as "Coverage Area" Bangalore Karnataka Bhopal Madhya Pradesh Delhi Delhi; Haryana; Rajasthan and Uttar Pradesh Hyderabad Telangana Kolkata Odisha; West Bengal; Sikkim Mumbai Maharashtra 4(4)b: Total Channel carrying capacity Distribution Network Location Capacity in SD Terms Bangalore 506 Bhopal 358 Delhi 384 Hyderabad 456 Kolkata 472 Mumbai 447 Kindly Note: 1. Local Channels considered as 1 SD; 2. Consideration in SD Terms is clarified as 1 SD = 1 SD; 1 HD = 2 SD; 3. Number of channels will vary within the area serviced by a distribution network location depending upon available Bandwidth capacity. 4(4)c: List of channels available on network List attached below in Annexure I 4(4)d: Number of channels which signals of television channels have been requested by the distributor from broadcasters and the interconnection agreements signed Nil Page 1 of 37 Version 1.0/2019 4(4)e: Spare channels capacity available on the network for the purpose of carrying signals of television channels Distribution Network Location Spare Channel Capacity in SD Terms Bangalore Nil Bhopal Nil Delhi Nil Hyderabad Nil Kolkata Nil Mumbai Nil 4(4)f: List of channels, in chronological order, for which requests have been received from broadcasters for distribution of their channels, the interconnection agreements -

Genre Channel Name Channel No Hindi Entertainment Star Bharat 114 Hindi Entertainment Investigation Discovery HD 136 Hindi Enter
Genre Channel Name Channel No Hindi Entertainment Star Bharat 114 Hindi Entertainment Investigation Discovery HD 136 Hindi Entertainment Big Magic 124 Hindi Entertainment Colors Rishtey 129 Hindi Entertainment STAR UTSAV 131 Hindi Entertainment Sony Pal 132 Hindi Entertainment Epic 138 Hindi Entertainment Zee Anmol 140 Hindi Entertainment DD National 148 Hindi Entertainment DD INDIA 150 Hindi Entertainment DD BHARATI 151 Infotainment DD KISAN 152 Hindi Movies Star Gold HD 206 Hindi Movies Zee Action 216 Hindi Movies Colors Cineplex 219 Hindi Movies Sony Wah 224 Hindi Movies STAR UTSAV MOVIES 225 Hindi Zee Anmol Cinema 228 Sports Star Sports 1 Hindi HD 282 Sports DD SPORTS 298 Hindi News ZEE NEWS 311 Hindi News AAJ TAK HD 314 Hindi News AAJ TAK 313 Hindi News NDTV India 317 Hindi News News18 India 318 Hindi News Zee Hindustan 319 Hindi News Tez 326 Hindi News ZEE BUSINESS 331 Hindi News News18 Rajasthan 335 Hindi News Zee Rajasthan News 336 Hindi News News18 UP UK 337 Hindi News News18 MP Chhattisgarh 341 Hindi News Zee MPCG 343 Hindi News Zee UP UK 351 Hindi News DD UP 400 Hindi News DD NEWS 401 Hindi News DD LOK SABHA 402 Hindi News DD RAJYA SABHA 403 Hindi News DD RAJASTHAN 404 Hindi News DD MP 405 Infotainment Gyan Darshan 442 Kids CARTOON NETWORK 449 Kids Pogo 451 Music MTV Beats 482 Music ETC 487 Music SONY MIX 491 Music Zing 501 Marathi DD SAHYADRI 548 Punjabi ZEE PUNJABI 562 Hindi News News18 Punjab Haryana Himachal 566 Punjabi DD PUNJABI 572 Gujrati DD Girnar 589 Oriya DD ORIYA 617 Urdu Zee Salaam 622 Urdu News18 Urdu 625 Urdu -

Catvision Limited
CATVISION LIMITED BASIC SERVICE TIER (BST) Sr. No. Channel Name Genre 1 Sahara One GEC Hindi 2 DD National GEC Hindi 3 DD Bharati GEC Hindi 4 Manoranjan TV GEC Hindi 5 Dabangg GEC Hindi 6 Firangi GEC Hindi 7 Filmy Hindi Movies 8 B4U Movies India Hindi Movies 9 Manoranjan Movies Hindi Movies 10 Dangal TV Hindi Movies 11 Cinema TV (India) Hindi Movies 12 Wow Cinema Hindi Movies 13 Enterr 10 Hindi Movies 14 Movie House Hindi Movies 15 House Full Movies Hindi Movies 16 House Full Action Hindi Movies 17 Oscar Movies Bhojpuri Bhojpuri Movies 18 Bhojpuri Cinema Bhojpuri Movies 19 Fight Sports Sports 20 DD Sports Sports 21 Samay National Hindi News 22 News 1 India Hindi News 23 News Nation Hindi News 24 News State MP & CHG Hindi News 25 News State UP & UK Hindi News 26 DD News Hindi News 27 India News Hindi News 28 Lok Sabha TV Hindi News 29 Rajya Sabha TV Hindi News 30 DD North-East Hindi News 31 DD Uttar Pradesh Hindi News 32 India TV (India) Hindi News 33 News State UP & UK Hindi News 34 News 24 (India) Hindi News 35 Dilli Aaj Tak Hindi News 36 ABP News India Hindi News 37 DD India Hindi News 38 Samay UP/Uttarakhand Hindi News Sr. No. Channel Name Genre 39 Zee News Hindi News 40 Zee Punjab Haryana Himachal Hindi News 41 TV 9 Telugu Regional News 42 TV 1 Telugu Regional News 43 TV 9 Kannada Regional News 44 News 9 Regional News 45 TV 9 Maharashtra Regional News 46 TV 9 Gujarat Regional News 47 Sadhna Prime News Regional News 48 ANM News Regional News 49 Super TV (India) Regional News 50 Jai Maharashtra Regional News 51 News Time Bangla Regional -

Channel List: 1 DD National SD Hindi Entertainment 114 FTA FTA FTA 2 Star Plus HD HD Hindi Entertainment 115 Pay ₹ 19.00 ₹
Channel List: - Channels, EPG numbers and prices are subject to change. - MRP: Maximum Retail Price, per month. DRP (Distributor Retail Price) of all channels is the same as the MRP. - Pack lock-in duration: 1 day Sr. Channel Name HD/SD Genre EPG No. FTA/Pay MRP MRP + No. Tax 1 DD National SD Hindi Entertainment 114 FTA FTA FTA 2 Star Plus HD HD Hindi Entertainment 115 Pay ₹ 19.00 ₹ 22.42 3 Star Plus SD Hindi Entertainment 117 Pay ₹ 19.00 ₹ 22.42 4 Star Bharat HD HD Hindi Entertainment 121 Pay ₹ 19.00 ₹ 22.42 5 Star Bharat SD Hindi Entertainment 122 Pay ₹ 10.00 ₹ 11.80 6 SET HD HD Hindi Entertainment 128 Pay ₹ 19.00 ₹ 22.42 7 SET SD Hindi Entertainment 130 Pay ₹ 19.00 ₹ 22.42 8 Sony SAB HD HD Hindi Entertainment 132 Pay ₹ 19.00 ₹ 22.42 9 Sony SAB SD Hindi Entertainment 134 Pay ₹ 19.00 ₹ 22.42 10 &TV HD HD Hindi Entertainment 137 Pay ₹ 19.00 ₹ 22.42 11 &TV SD Hindi Entertainment 139 Pay ₹ 12.00 ₹ 14.16 12 Zee TV HD HD Hindi Entertainment 141 Pay ₹ 19.00 ₹ 22.42 13 Zee TV SD Hindi Entertainment 143 Pay ₹ 19.00 ₹ 22.42 14 Colors HD HD Hindi Entertainment 147 Pay ₹ 19.00 ₹ 22.42 15 Colors SD Hindi Entertainment 149 Pay ₹ 19.00 ₹ 22.42 16 UTV Bindass SD Hindi Entertainment 153 Pay ₹ 1.00 ₹ 1.18 17 Investigation Discovery SD Hindi Entertainment 155 Pay ₹ 1.00 ₹ 1.18 18 Naaptol SD Shopping 156 FTA FTA FTA 19 Ezmall SD Others 158 FTA FTA FTA 20 Star Utsav SD Hindi Entertainment 171 Pay ₹ 1.00 ₹ 1.18 21 Zee Anmol SD Hindi Entertainment 172 Pay ₹ 0.10 ₹ 0.12 22 Colors Rishtey SD Hindi Entertainment 173 Pay ₹ 1.00 ₹ 1.18 23 Sony Pal SD Hindi Entertainment -

List of Bouquets of FTA Basic Service Tier
List of Bouquets of FTA Basic Service Tier BST North Channel Genre DD7 Bangla Bangla Aakash aath Bangla Dhoom Music Bangla News Time Bangla Sangeet Bangla Bangla R Plus Bangla ABP Ananda Bangla DD Bihar Bihar Sangeet Bhojpuri Bihar Dangal TV Bihar Bhojpuri Cinema Bihar Dabang Bihar Aastha Devotional Aastha Bhajan Devotional Arihant TV Devotional Divya TV Devotional GOD TV Devotional Ishwar TV Devotional Mahavira Devotional Peace of Mind Devotional Sanskar Devotional Satsang Devotional Vedic Devotional Sadhna Bhakti Devotional Shubh TV Devotional India Ahead Eng News Republic TV Eng News DD Girnar Gujarati TV9 Gujarati Gujarati GS TV Gujarati Sandesh News Gujarati ABP Asmita Gujarati DD India Hindi Gec DD Bharati Hindi Gec DD National Hindi Gec Big Magic Hindi Gec Box Cinema Hindi Movie Maha Movie Hindi Movies WOW Cinema Hindi Movies Cinema TV Hindi Movies Manoranjan Movies Hindi Movies Manoranjan TV Hindi Movies Housefull Movies Hindi Movies B4U Movies Hindi Movies Sky Star Hindi Movies Enterr10 Hindi Movies Movie House Hindi Movies DD9 Chandana (Kannada) Kannada DD Kashir Kashmir DD Loksabha Hindi News DD Rajyasabha Hindi News DD News Hindi News Aaj Tak Hindi News ABP news Hindi News Sadhana Plus Hindi News India News Hindi News India tv Hindi News News Nation Hindi News News 24 Hindi News Zee Hindustan Hindi News Zee News Hindi News Republic Bharat Hindi News DD Kisan Infotainment Digishala Infotainment CGTN International France 24 International Russia Today International Home shop 18 Lifestyle/Fashion NT 1 Lifestyle/Fashion Divyarishi -

Tracking Media Behaviour in the Time of a Pandemic
A JOURNAL OF THE PRESS INSTITUTE OF INDIA ISSN 0042-5303 October-December 2020 Volume 12 Issue 4 Rs 60 COVID-19 Tracking media behaviour in the time of a pandemic A pandemic is a public health emergency and much more. It CONTENTS not only galvanizes the health institutions but several other • Domestic workers bear the brunt, parts of public life, says Pradeep Krishnatray seek return to good times / Sudha Umashankar ass media is an integral part of public life. It has to necessarily • How the innovative Indian is report about and react to the pandemic. How it does and whose fighting a deadly pandemic / Rina Mukherji perspective it adopts, is of critical importance. There are at least M • Learning in the new normal four ways of examining this in its entirety. Put together, they help explain comes with a set of challenges / media behaviour. Afsana Rashid The first, of course is content. That is, what the media chooses to tell or • Anxiety, job losses, financial woes not tell is of importance. The second aspect is its role vis-à-vis the state and driving more people to suicide / Shoma A. Chatterji Central Government of the day --- the agency tasked to deal with the pan- • A different Durga Puja, with poor demic. Does it adopt a watchdog role, an adversarial role, or a supportive craftsperson the hardest hit / role? The third is its public function, its relationship with the society from Manjira Mazumdar which it derives its credibility and sustainability. Whose side it is on or • Of contributors, editors and what version of the truth does it prefer to bet on? Does it speak up for the journalistic ethics / Sakuntala Narasimhan people? Finally, a lot depends on the media’s ability to manage the tension • Wanted: objectivity and and contradictions that invariably emerge from its editorial stance. -

Origin of a Folded Repeat Protein from an Intrinsically Disordered Ancestor
RESEARCH ARTICLE Origin of a folded repeat protein from an intrinsically disordered ancestor Hongbo Zhu, Edgardo Sepulveda, Marcus D Hartmann, Manjunatha Kogenaru†, Astrid Ursinus, Eva Sulz, Reinhard Albrecht, Murray Coles, Jo¨ rg Martin, Andrei N Lupas* Department of Protein Evolution, Max Planck Institute for Developmental Biology, Tu¨ bingen, Germany Abstract Repetitive proteins are thought to have arisen through the amplification of subdomain- sized peptides. Many of these originated in a non-repetitive context as cofactors of RNA-based replication and catalysis, and required the RNA to assume their active conformation. In search of the origins of one of the most widespread repeat protein families, the tetratricopeptide repeat (TPR), we identified several potential homologs of its repeated helical hairpin in non-repetitive proteins, including the putatively ancient ribosomal protein S20 (RPS20), which only becomes structured in the context of the ribosome. We evaluated the ability of the RPS20 hairpin to form a TPR fold by amplification and obtained structures identical to natural TPRs for variants with 2–5 point mutations per repeat. The mutations were neutral in the parent organism, suggesting that they could have been sampled in the course of evolution. TPRs could thus have plausibly arisen by amplification from an ancestral helical hairpin. DOI: 10.7554/eLife.16761.001 *For correspondence: andrei. [email protected] Introduction † Present address: Department Most present-day proteins arose through the combinatorial shuffling and differentiation of a set of of Life Sciences, Imperial College domain prototypes. In many cases, these prototypes can be traced back to the root of cellular life London, London, United and have since acted as the primary unit of protein evolution (Anantharaman et al., 2001; Kingdom Apic et al., 2001; Koonin, 2003; Kyrpides et al., 1999; Orengo and Thornton, 2005; Ponting and Competing interests: The Russell, 2002; Ranea et al., 2006). -

Triple Play Ultra 252 Channels & Services*
Triple Play Ultra 252 channels & services* HINDI ENTERTAINMENT &TV Big Magic Colors Colors +1 ID Rishtey SAB Sony Sony +1 Sony Pal Star Bharat STAR Plus STAR Plus +1 STAR Utsav UTV Bindass Zee Anmol Zee TV Zee TV +1 HINDI MOVIES &Pictures B4U Movies Bflix Movies Cinema TV Enterr10 Movies Movies Ok Multiplex Rishtey Cineplex Sky Star Sony MAX Sony MAX +1 Sony Max2 Sony Wah STAR Gold STAR Gold +1 Star Gold Select Star Utsav Movies UTV Action UTV Movies WOW Cinema Z Action Zee Anmol Cinema Zee Cinema Zee Cinema +1 Zee Classic HINDI NEWS Aaj Tak Aaj Tak Tez ABP News APN Bharat Samachar CNBC Awaaz Hindi Khabar HNN 24x7 India News India News Haryana India TV Janta TV JK 24x7 News K News Kashish News Khabarain Abhi Tak LTV MH One News NDTV India News 11 News 24 News India News Live News Nation News World India News1 India News18 India News18 Punjab Patrika TV Rajasthan Sadhna Plus Haryana Himachal Sadhna Prime News Samachar Plus Samay Samay Samay MP/CG Bihar/Jharkhand Samay Rajasthan Samay UP/UK Sudarshan News Swaraj Express Total TV SMBC Yo TV Zee Business Zee Hindustan Zee News SPORTS Neo Prime Neo Sports Sony ESPN Sony Six Sony Ten 1 Sony Ten 2 Sony Ten 3 Star Sports 1 Star Sports 2 Star Sports Hindi 1 Star Sports Select 1 Star Sports Select 2 Star Sports Tamil 1 MUSIC 9X Jalwa 9XM B4U Music Channel V E24 Mastiii MTV MTV Beats Music India Nat Geo Music Sony MIX VH1 Z ETC Bollywood Zing Zoom KIDS Baby TV Cartoon Network Discovery Kids Disney Channel Disney Junior Disney XD Hungama Nick Nick Jr.