Gynaecological Uses of Dienogest Alone and in Combination with Oestrogens V
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ocps) Used in the NM Family Planning Program (FPP
The following slides are intended to familiarize nurses and clinicians with oral contraceptive pills (OCPs) used in the NM Family Planning Program (FPP). The FPP does not intend for this information to supersede the Family Planning Protocol particularly on the requirement for Public Health Nurses in the Public Health Offices to consult a clinician as stated in the Protocol when in doubt or if it is necessary to switch the client’s OCP type. 1 2 Combined oral contraceptives (COCs) contain two hormones; estrogen and progestin. In general, any combined OCP is good for most women who are eligible to take estrogen according to the CDC U.S. Medical Eligibility Criteria (MEC). Once again, refer to the US MEC chart to find out if OCP is a suitable choice for clients with specific health conditions. To learn a little bit about what each hormone does, the FPP is providing the following summary: Estrogen: provides endometrial stability = menstrual cycle control. A higher estrogen dose increases the venous thromboembolism (VTE) or clot risk but OCP clot risk is still less harmful than the clot risk related to pregnancy and giving birth. Progestin: provides most of the contraceptive effect by ‐Preventing luteinizing hormone (LH) surge /ovulation ‐Thickening the cervical mucus to prevent sperm entry. Two major OCP formations are available. Monophasic: There is only one dose of estrogen and progestin in each active pill in the packet; and Multiphasic: There are varying doses of hormones, particularly progestin in the active pills. 3 Section 3 of the FPP Protocol contains the OCP Substitute Table, which groups OCPs into 6 classes according to the estrogen dosage, the type of progestin and the formulations. -

Download PDF File
Ginekologia Polska 2019, vol. 90, no. 9, 520–526 Copyright © 2019 Via Medica ORIGINAL PAPER / GYNECologY ISSN 0017–0011 DOI: 10.5603/GP.2019.0091 Anti-androgenic therapy in young patients and its impact on intensity of hirsutism, acne, menstrual pain intensity and sexuality — a preliminary study Anna Fuchs, Aleksandra Matonog, Paulina Sieradzka, Joanna Pilarska, Aleksandra Hauzer, Iwona Czech, Agnieszka Drosdzol-Cop Department of Pregnancy Pathology, Department of Woman’s Health, School of Health Sciences in Katowice, Medical University of Silesia, Katowice, Poland ABSTRACT Objectives: Using anti-androgenic contraception is one of the methods of birth control. It also has a significant, non-con- traceptive impact on women’s body. These drugs can be used in various endocrinological disorders, because of their ability to reduce the level of male hormones. The aim of our study is to establish a correlation between taking different types of anti-androgenic drugs and intensity of hirsutism, acne, menstrual pain intensity and sexuality . Material and methods: 570 women in childbearing age that had been using oral contraception for at least three months took part in our research. We examined women and asked them about quality of life, health, direct causes and effects of that treatment, intensity of acne and menstrual pain before and after. Our research group has been divided according to the type of gestagen contained in the contraceptive pill: dienogest, cyproterone, chlormadynone and drospirenone. Ad- ditionally, the control group consisted of women taking oral contraceptives without antiandrogenic component. Results: The mean age of the studied group was 23 years ± 3.23. 225 of 570 women complained of hirsutism. -

Comparing the Effects of Combined Oral Contraceptives Containing Progestins with Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis In
JMIR RESEARCH PROTOCOLS Amiri et al Review Comparing the Effects of Combined Oral Contraceptives Containing Progestins With Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis in Patients With Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis Mina Amiri1,2, PhD, Postdoc; Fahimeh Ramezani Tehrani2, MD; Fatemeh Nahidi3, PhD; Ali Kabir4, MD, MPH, PhD; Fereidoun Azizi5, MD 1Students Research Committee, School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 2Reproductive Endocrinology Research Center, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 3School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 4Minimally Invasive Surgery Research Center, Iran University of Medical Sciences, Tehran, Islamic Republic Of Iran 5Endocrine Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran Corresponding Author: Fahimeh Ramezani Tehrani, MD Reproductive Endocrinology Research Center Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences 24 Parvaneh Yaman Street, Velenjak, PO Box 19395-4763 Tehran, 1985717413 Islamic Republic Of Iran Phone: 98 21 22432500 Email: [email protected] Abstract Background: Different products of combined oral contraceptives (COCs) can improve clinical and biochemical findings in patients with polycystic ovary syndrome (PCOS) through suppression of the hypothalamic-pituitary-gonadal (HPG) axis. Objective: This systematic review and meta-analysis aimed to compare the effects of COCs containing progestins with low androgenic and antiandrogenic activities on the HPG axis in patients with PCOS. -
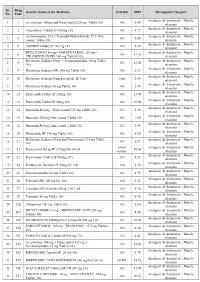
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Determination of 17 Hormone Residues in Milk by Ultra-High-Performance Liquid Chromatography and Triple Quadrupole Mass Spectrom
No. LCMSMS-065E Liquid Chromatography Mass Spectrometry Determination of 17 Hormone Residues in Milk by Ultra-High-Performance Liquid Chromatography and Triple Quadrupole No. LCMSMS-65E Mass Spectrometry This application news presents a method for the determination of 17 hormone residues in milk using Shimadzu Ultra-High-Performance Liquid Chromatograph (UHPLC) LC-30A and Triple Quadrupole Mass Spectrometer LCMS- 8040. After sample pretreatment, the compounds in the milk matrix were separated using UPLC LC-30A and analyzed via Triple Quadrupole Mass Spectrometer LCMS-8040. All 17 hormones displayed good linearity within their respective concentration range, with correlation coefficient in the range of 0.9974 and 0.9999. The RSD% of retention time and peak area of 17 hormones at the low-, mid- and high- concentrations were in the range of 0.0102-0.161% and 0.563-6.55% respectively, indicating good instrument precision. Method validation was conducted and the matrix spike recovery of milk ranged between 61.00-110.9%. The limit of quantitation was 0.14-0.975 g/kg, and it meets the requirement for detection of hormones in milk. Keywords: Hormones; Milk; Solid phase extraction; Ultra performance liquid chromatograph; Triple quadrupole mass spectrometry ■ Introduction Since 2008’s melamine-tainted milk scandal, the With reference to China’s national standard GB/T adulteration of milk powder has become a major 21981-2008 "Hormone Multi-Residue Detection food safety concern. In recent years, another case of Method for Animal-derived Food - LC-MS Method", dairy product safety is suspected to cause "infant a method utilizing solid phase extraction, ultra- sexual precocity" (also known as precocious puberty) performance liquid chromatography and triple and has become another major issue challenging the quadrupole mass spectrometry was developed for dairy industry in China. -

Desogestrel-Only Pill (Cerazette)
J Fam Plann Reprod Health Care: first published as 10.1783/147118903101197593 on 1 July 2003. Downloaded from Faculty of Family Planning and Reproductive Health Care Clinical Effectiveness Unit A unit funded by the FFPRHC and supported by the University of Aberdeen and SPCERH to provide guidance on evidence-based practice New Product Review (April 2003) Desogestrel-only Pill (Cerazette) Journal of Family Planning and Reproductive Health Care 2003; 29(3): 162–164 Evidence from a randomised trial has shown that a 75 mg (microgrammes) desogestrel pill inhibits ovulation in 97% of cycles. Thus, on theoretical grounds, we would expect the desogestrel pill to be more effective than existing progestogen- only pills (POPs). However, Pearl indices from clinical trials comparing it to a levonorgestrel POP were not significantly different. Therefore an evidence-based recommendation cannot be made that the desogestrel pill is different from other POPs in terms of efficacy, nor that it is similar to combined oral contraception (COC) in this respect. An evidence-based recommendation can be made that the desogestrel-only pill is similar to other POPs in terms of side effects and acceptability. The desogestrel-only pill is not recommended as an alternative to COC in routine practice, but provides a useful alternative for women who require oestrogen-free contraception. In clinical trials: l Ovulation was inhibited in 97% of cycles at 7 and 12 months after initiation. l The Pearl index was 0.41 per 100 woman-years, which was not significantly different from a levonorgestrel-only pill. However, the trial providing these data was too small to detect a clinically important difference. -

Etats Rapides
List of European Pharmacopoeia Reference Standards Effective from 2015/12/24 Order Reference Standard Batch n° Quantity Sale Information Monograph Leaflet Storage Price Code per vial Unit Y0001756 Exemestane for system suitability 1 10 mg 1 2766 Yes +5°C ± 3°C 79 ! Y0001561 Abacavir sulfate 1 20 mg 1 2589 Yes +5°C ± 3°C 79 ! Y0001552 Abacavir for peak identification 1 10 mg 1 2589 Yes +5°C ± 3°C 79 ! Y0001551 Abacavir for system suitability 1 10 mg 1 2589 Yes +5°C ± 3°C 79 ! Y0000055 Acamprosate calcium - reference spectrum 1 n/a 1 1585 79 ! Y0000116 Acamprosate impurity A 1 50 mg 1 3-aminopropane-1-sulphonic acid 1585 Yes +5°C ± 3°C 79 ! Y0000500 Acarbose 3 100 mg 1 See leaflet ; Batch 2 is valid until 31 August 2015 2089 Yes +5°C ± 3°C 79 ! Y0000354 Acarbose for identification 1 10 mg 1 2089 Yes +5°C ± 3°C 79 ! Y0000427 Acarbose for peak identification 3 20 mg 1 Batch 2 is valid until 31 January 2015 2089 Yes +5°C ± 3°C 79 ! A0040000 Acebutolol hydrochloride 1 50 mg 1 0871 Yes +5°C ± 3°C 79 ! Y0000359 Acebutolol impurity B 2 10 mg 1 -[3-acetyl-4-[(2RS)-2-hydroxy-3-[(1-methylethyl)amino] propoxy]phenyl] 0871 Yes +5°C ± 3°C 79 ! acetamide (diacetolol) Y0000127 Acebutolol impurity C 1 20 mg 1 N-(3-acetyl-4-hydroxyphenyl)butanamide 0871 Yes +5°C ± 3°C 79 ! Y0000128 Acebutolol impurity I 2 0.004 mg 1 N-[3-acetyl-4-[(2RS)-3-(ethylamino)-2-hydroxypropoxy]phenyl] 0871 Yes +5°C ± 3°C 79 ! butanamide Y0000056 Aceclofenac - reference spectrum 1 n/a 1 1281 79 ! Y0000085 Aceclofenac impurity F 2 15 mg 1 benzyl[[[2-[(2,6-dichlorophenyl)amino]phenyl]acetyl]oxy]acetate -

Effects of 2 Mg Chlormadinone Acetate/0.03 Mg Ethinylestradiol In
Journal für Reproduktionsmedizin und Endokrinologie – Journal of Reproductive Medicine and Endocrinology – Andrologie • Embryologie & Biologie • Endokrinologie • Ethik & Recht • Genetik Gynäkologie • Kontrazeption • Psychosomatik • Reproduktionsmedizin • Urologie Effects of 2 mg Chlormadinone Acetate/0.03 mg Ethinylestradiol in Primary Dysmenorrhoea: The BEDY (Belara(R) Evaluation on Dysmenorrhea) Study - an Open Non-Comparative, Non-Interventional Observational Study with 4,842 Women Schramm GAK, Waldmann-Rex S J. Reproduktionsmed. Endokrinol 2010; 7 (Sonderheft 1), 112-118 www.kup.at/repromedizin Online-Datenbank mit Autoren- und Stichwortsuche Offizielles Organ: AGRBM, BRZ, DVR, DGA, DGGEF, DGRM, D·I·R, EFA, OEGRM, SRBM/DGE Indexed in EMBASE/Excerpta Medica/Scopus Krause & Pachernegg GmbH, Verlag für Medizin und Wirtschaft, A-3003 Gablitz Effects of CMA/EE in Primary Dysmenorrhoea Effects of 2 mg Chlormadinone Acetate/ 0.03 mg Ethinylestradiol in Primary Dysmenorrhoea: The BEDY (Belara® Evaluation on Dysmenorrhea) Study – an Open, Non-Comparative, Non-Interventional Observational Study with 4,842 Women G. A. K. Schramm, S. Waldmann-Rex Background: This prospective, non-interventional, observational study designed to reflect the daily medical practice which is very important for the product observation liability investigated the effects of 2.0 mg chlormadinone acetate/0.03 mg ethinylestradiol (CMA/EE) on dysmenorrhoea and related problems. Study design: A total of 4,842 patients were observed during a six-cycle period (26,945.5 treatment cycles) in 608 office-based gynaecological centres throughout Germany. Results: The administration of CMA/EE significantly reduced the number of patients who suffered from menstrual pain (–50.4 %). Analgesic use and absenteeism from school or work due to dysmenorrhoea was reduced by 74.6 % in OC starters and 91.9 % in OC switchers. -

Progestogens and Venous Thromboembolism Among Postmenopausal Women Using Hormone Therapy. Marianne Canonico, Geneviève Plu-Bureau, Pierre-Yves Scarabin
Progestogens and venous thromboembolism among postmenopausal women using hormone therapy. Marianne Canonico, Geneviève Plu-Bureau, Pierre-Yves Scarabin To cite this version: Marianne Canonico, Geneviève Plu-Bureau, Pierre-Yves Scarabin. Progestogens and venous throm- boembolism among postmenopausal women using hormone therapy.. Maturitas, Elsevier, 2011, 70 (4), pp.354-60. 10.1016/j.maturitas.2011.10.002. inserm-01148705 HAL Id: inserm-01148705 https://www.hal.inserm.fr/inserm-01148705 Submitted on 5 May 2015 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Progestogens and VTE Finale version Progestogens and venous thromboembolism among postmenopausal women using hormone therapy Marianne Canonico1,2, Geneviève Plu-Bureau1,3 and Pierre-Yves Scarabin1,2 1 Centre for Research in Epidemiology and Population Health, U1018, Hormones and Cardiovascular Disease 2 University Paris-Sud, UMR-S 1018, Villejuif, France 3 University Paris Descartes and Hôtel-Dieu Hospital, Paris, France Adresse: 16 av. Paul Vaillant Couturier 94807 Villejuif Cedex Tel: +33 1 45 59 51 66 Fax: +33 1 45 59 51 70 Corresponding author: Marianne Canonico ([email protected]) 1/21 Progestogens and VTE Finale version Abstract Hormone therapy (HT) is the most effective treatment for correcting menopausal symptoms after menopause. -

Contraceptive Choices for Women with Inflammatory Bowel Disease
J Fam Plann Reprod Health Care: first published as 10.1783/147118903101197782 on 1 July 2003. Downloaded from Faculty of Family Planning and Reproductive Health Care Clinical Effectiveness Unit A unit funded by the FFPRHC and supported by the University of Aberdeen and SPCERH to provide guidance on evidence-based practice FFPRHC Guidance (July 2003) Contraceptive Choices for Women with Inflammatory Bowel Disease Journal of Family Planning and Reproductive Health Care 2003; 29(3): 127–135 This Guidance provides information on the effects of inflammatory bowel disease (IBD) in women of reproductive age with particular reference to contraceptive choices, fertility and pregnancy. For comprehensive advice on the management of patients with IBD, readers should refer to guidelines from the British Society for Gastroenterology.1 A key to the grades of recommendations, based on levels of evidence, is given at the end of this document. Details of the methods used by the Clinical Effectiveness Unit (CEU) in developing this Guidance, and evidence tables summarising the research basis of the recommendations, are available on the Faculty website (www.ffprhc.org.uk). Abbreviations used include: 5-aminosalicylic acid (5-ASA), body mass index (BMI), bone mineral density (BMD), combined oral contraception (COC), confidence interval (CI), copper-bearing intrauterine contraceptive device (IUD), Crohn’s disease (CD), depot medroxyprogesterone acetate (DMPA), dual X-ray absorptiometry (DEXA), emergency contraception (EC), gastrointestinal (GI), incidence rate ratio (IRR), inflammatory bowel disease (IBD), levonorgestrel-releasing intrauterine system (IUS), odds ratio (OR), progestogen-only emergency contraception (POEC), progestogen-only pill (POP), anti-tumour necrosis factor-alpha (anti- TNF-a), ulcerative colitis (UC), venous thromboembolism (VTE), World Health Organization (WHO). -

Minutes of PRAC Meeting on 09-12 July 2018
6 September 2018 EMA/PRAC/576790/2018 Inspections, Human Medicines Pharmacovigilance and Committees Division Pharmacovigilance Risk Assessment Committee (PRAC) Minutes of the meeting on 09-12 July 2018 Chair: June Raine – Vice-Chair: Almath Spooner Health and safety information In accordance with the Agency’s health and safety policy, delegates were briefed on health, safety and emergency information and procedures prior to the start of the meeting. Disclaimers Some of the information contained in the minutes is considered commercially confidential or sensitive and therefore not disclosed. With regard to intended therapeutic indications or procedure scope listed against products, it must be noted that these may not reflect the full wording proposed by applicants and may also change during the course of the review. Additional details on some of these procedures will be published in the PRAC meeting highlights once the procedures are finalised. Of note, the minutes are a working document primarily designed for PRAC members and the work the Committee undertakes. Note on access to documents Some documents mentioned in the minutes cannot be released at present following a request for access to documents within the framework of Regulation (EC) No 1049/2001 as they are subject to on- going procedures for which a final decision has not yet been adopted. They will become public when adopted or considered public according to the principles stated in the Agency policy on access to documents (EMA/127362/2006, Rev. 1). 30 Churchill Place ● Canary Wharf ● London E14 5EU ● United Kingdom Telephone +44 (0)20 3660 6000 Facsimile +44 (0)20 3660 5555 Send a question via our website www.ema.europa.eu/contact An agency of the European Union © European Medicines Agency, 2018. -

Malta Medicines List April 08
Defined Daily Doses Pharmacological Dispensing Active Ingredients Trade Name Dosage strength Dosage form ATC Code Comments (WHO) Classification Class Glucobay 50 50mg Alpha Glucosidase Inhibitor - Blood Acarbose Tablet 300mg A10BF01 PoM Glucose Lowering Glucobay 100 100mg Medicine Rantudil® Forte 60mg Capsule hard Anti-inflammatory and Acemetacine 0.12g anti rheumatic, non M01AB11 PoM steroidal Rantudil® Retard 90mg Slow release capsule Carbonic Anhydrase Inhibitor - Acetazolamide Diamox 250mg Tablet 750mg S01EC01 PoM Antiglaucoma Preparation Parasympatho- Powder and solvent for solution for mimetic - Acetylcholine Chloride Miovisin® 10mg/ml Refer to PIL S01EB09 PoM eye irrigation Antiglaucoma Preparation Acetylcysteine 200mg/ml Concentrate for solution for Acetylcysteine 200mg/ml Refer to PIL Antidote PoM Injection injection V03AB23 Zovirax™ Suspension 200mg/5ml Oral suspension Aciclovir Medovir 200 200mg Tablet Virucid 200 Zovirax® 200mg Dispersible film-coated tablets 4g Antiviral J05AB01 PoM Zovirax® 800mg Aciclovir Medovir 800 800mg Tablet Aciclovir Virucid 800 Virucid 400 400mg Tablet Aciclovir Merck 250mg Powder for solution for inj Immunovir® Zovirax® Cream PoM PoM Numark Cold Sore Cream 5% w/w (5g/100g)Cream Refer to PIL Antiviral D06BB03 Vitasorb Cold Sore OTC Cream Medovir PoM Neotigason® 10mg Acitretin Capsule 35mg Retinoid - Antipsoriatic D05BB02 PoM Neotigason® 25mg Acrivastine Benadryl® Allergy Relief 8mg Capsule 24mg Antihistamine R06AX18 OTC Carbomix 81.3%w/w Granules for oral suspension Antidiarrhoeal and Activated Charcoal