Crucial Genes Associated with Diabetic Nephropathy Explored by Microarray Analysis Zhikui Wang, Zhaoxia Wang*, Zhongqi Zhou and Yueqin Ren
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

FLNC Pathogenic Variants in Patients with Cardiomyopathies
FLNC pathogenic variants in patients with cardiomyopathies: Prevalence and genotype-phenotype correlations Flavie Ader, Pascal de Groote, Patricia Réant, Caroline Rooryck-Thambo, Delphine Dupin-Deguine, Caroline Rambaud, Diala Khraiche, Claire Perret, Jean-François Pruny, Michèle Mathieu-dramard, et al. To cite this version: Flavie Ader, Pascal de Groote, Patricia Réant, Caroline Rooryck-Thambo, Delphine Dupin-Deguine, et al.. FLNC pathogenic variants in patients with cardiomyopathies: Prevalence and genotype-phenotype correlations. Clinical Genetics, Wiley, 2019, 96 (4), pp.317-329. 10.1111/cge.13594. hal-02268422 HAL Id: hal-02268422 https://hal-normandie-univ.archives-ouvertes.fr/hal-02268422 Submitted on 29 Jun 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. FLNC pathogenic variants in patients with cardiomyopathies Prevalence and genotype-phenotype correlations Running Title : FLNC variants genotype-phenotype correlation Flavie Ader1,2,3, Pascal De Groote4, Patricia Réant5, Caroline Rooryck-Thambo6, Delphine Dupin-Deguine7, Caroline Rambaud8, Diala Khraiche9, Claire Perret2, Jean Francois Pruny10, Michèle Mathieu Dramard11, Marion Gérard12, Yann Troadec12, Laurent Gouya13, Xavier Jeunemaitre14, Lionel Van Maldergem15, Albert Hagège16, Eric Villard2, Philippe Charron2, 10, Pascale Richard1, 2, 10. Conflict of interest statement: none declared for each author 1. -

Analysis of Trans Esnps Infers Regulatory Network Architecture
Analysis of trans eSNPs infers regulatory network architecture Anat Kreimer Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2014 © 2014 Anat Kreimer All rights reserved ABSTRACT Analysis of trans eSNPs infers regulatory network architecture Anat Kreimer eSNPs are genetic variants associated with transcript expression levels. The characteristics of such variants highlight their importance and present a unique opportunity for studying gene regulation. eSNPs affect most genes and their cell type specificity can shed light on different processes that are activated in each cell. They can identify functional variants by connecting SNPs that are implicated in disease to a molecular mechanism. Examining eSNPs that are associated with distal genes can provide insights regarding the inference of regulatory networks but also presents challenges due to the high statistical burden of multiple testing. Such association studies allow: simultaneous investigation of many gene expression phenotypes without assuming any prior knowledge and identification of unknown regulators of gene expression while uncovering directionality. This thesis will focus on such distal eSNPs to map regulatory interactions between different loci and expose the architecture of the regulatory network defined by such interactions. We develop novel computational approaches and apply them to genetics-genomics data in human. We go beyond pairwise interactions to define network motifs, including regulatory modules and bi-fan structures, showing them to be prevalent in real data and exposing distinct attributes of such arrangements. We project eSNP associations onto a protein-protein interaction network to expose topological properties of eSNPs and their targets and highlight different modes of distal regulation. -

Genetic Variation Screening of TNNT2 Gene in a Cohort of Patients with Hypertrophic and Dilated Cardiomyopathy
Physiol. Res. 61: 169-175, 2012 https://doi.org/10.33549/physiolres.932157 Genetic Variation Screening of TNNT2 Gene in a Cohort of Patients With Hypertrophic and Dilated Cardiomyopathy M. JÁCHYMOVÁ1, A. MURAVSKÁ1, T. PALEČEK2, P. KUCHYNKA2, H. ŘEHÁKOVÁ1, S. MAGAGE2, A. KRÁL2, T. ZIMA1, K. HORKÝ2, A. LINHART2 1Institute of Clinical Chemistry and Laboratory Diagnostics, First Faculty of Medicine and General University Hospital, Charles University, Prague, Czech Republic, 2Second Department of Internal Medicine – Clinical Department of Cardiology and Angiology, First Faculty of Medicine and General University Hospital, Charles University, Prague, Czech Republic Received February 1, 2011 Accepted October 17, 2011 On-line January 31, 2012 Summary Introduction Mutations in troponin T (TNNT2) gene represent the important part of currently identified disease-causing mutations in Cardiomyopathies are generally defined as hypertrophic (HCM) and dilated (DCM) cardiomyopathy. The aim myocardial disorders in which the heart muscle is of this study was to analyze TNNT2 gene exons in patients with structurally and functionally abnormal, in the absence of HCM and DCM diagnosis to improve diagnostic and genetic coronary artery disease, hypertension, valvular disease consultancy in affected families. All 15 exons and their flanking and congenital heart disease sufficient to cause the regions of the TNNT2 gene were analyzed by DNA sequence observed myocardial abnormality (Elliott et al. 2008). analysis in 174 patients with HCM and DCM diagnosis. We According to the morphological and functional phenotype identified genetic variations in TNNT2 exon regions in 56 patients the diagnosis of hypertrophic and dilated cardiomyopathy and genetic variations in TNNT2 intron regions in 164 patients. -

Bayesian Hierarchical Modeling of High-Throughput Genomic Data with Applications to Cancer Bioinformatics and Stem Cell Differentiation
BAYESIAN HIERARCHICAL MODELING OF HIGH-THROUGHPUT GENOMIC DATA WITH APPLICATIONS TO CANCER BIOINFORMATICS AND STEM CELL DIFFERENTIATION by Keegan D. Korthauer A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Statistics) at the UNIVERSITY OF WISCONSIN–MADISON 2015 Date of final oral examination: 05/04/15 The dissertation is approved by the following members of the Final Oral Committee: Christina Kendziorski, Professor, Biostatistics and Medical Informatics Michael A. Newton, Professor, Statistics Sunduz Kele¸s,Professor, Biostatistics and Medical Informatics Sijian Wang, Associate Professor, Biostatistics and Medical Informatics Michael N. Gould, Professor, Oncology © Copyright by Keegan D. Korthauer 2015 All Rights Reserved i in memory of my grandparents Ma and Pa FL Grandma and John ii ACKNOWLEDGMENTS First and foremost, I am deeply grateful to my thesis advisor Christina Kendziorski for her invaluable advice, enthusiastic support, and unending patience throughout my time at UW-Madison. She has provided sound wisdom on everything from methodological principles to the intricacies of academic research. I especially appreciate that she has always encouraged me to eke out my own path and I attribute a great deal of credit to her for the successes I have achieved thus far. I also owe special thanks to my committee member Professor Michael Newton, who guided me through one of my first collaborative research experiences and has continued to provide key advice on my thesis research. I am also indebted to the other members of my thesis committee, Professor Sunduz Kele¸s,Professor Sijian Wang, and Professor Michael Gould, whose valuable comments, questions, and suggestions have greatly improved this dissertation. -

Genetic Mutations and Mechanisms in Dilated Cardiomyopathy
Genetic mutations and mechanisms in dilated cardiomyopathy Elizabeth M. McNally, … , Jessica R. Golbus, Megan J. Puckelwartz J Clin Invest. 2013;123(1):19-26. https://doi.org/10.1172/JCI62862. Review Series Genetic mutations account for a significant percentage of cardiomyopathies, which are a leading cause of congestive heart failure. In hypertrophic cardiomyopathy (HCM), cardiac output is limited by the thickened myocardium through impaired filling and outflow. Mutations in the genes encoding the thick filament components myosin heavy chain and myosin binding protein C (MYH7 and MYBPC3) together explain 75% of inherited HCMs, leading to the observation that HCM is a disease of the sarcomere. Many mutations are “private” or rare variants, often unique to families. In contrast, dilated cardiomyopathy (DCM) is far more genetically heterogeneous, with mutations in genes encoding cytoskeletal, nucleoskeletal, mitochondrial, and calcium-handling proteins. DCM is characterized by enlarged ventricular dimensions and impaired systolic and diastolic function. Private mutations account for most DCMs, with few hotspots or recurring mutations. More than 50 single genes are linked to inherited DCM, including many genes that also link to HCM. Relatively few clinical clues guide the diagnosis of inherited DCM, but emerging evidence supports the use of genetic testing to identify those patients at risk for faster disease progression, congestive heart failure, and arrhythmia. Find the latest version: https://jci.me/62862/pdf Review series Genetic mutations and mechanisms in dilated cardiomyopathy Elizabeth M. McNally, Jessica R. Golbus, and Megan J. Puckelwartz Department of Human Genetics, University of Chicago, Chicago, Illinois, USA. Genetic mutations account for a significant percentage of cardiomyopathies, which are a leading cause of conges- tive heart failure. -

Chromatin Dysregulation and DNA Methylation at Transcription Start Sites Associated with Transcriptional Repression in Cancers
Corrected: Publisher correction ARTICLE https://doi.org/10.1038/s41467-019-09937-w OPEN Chromatin dysregulation and DNA methylation at transcription start sites associated with transcriptional repression in cancers Mizuo Ando 1,2, Yuki Saito1,2, Guorong Xu3, Nam Q. Bui 4,5, Kate Medetgul-Ernar1, Minya Pu1, Kathleen Fisch 3, Shuling Ren1, Akihiro Sakai1, Takahito Fukusumi1, Chao Liu1, Sunny Haft1, John Pang1, Adam Mark3, Daria A. Gaykalova6, Theresa Guo6, Alexander V. Favorov 7,8, Srinivasan Yegnasubramanian7, Elana J. Fertig7, Patrick Ha 9, Pablo Tamayo 1, Tatsuya Yamasoba 2, Trey Ideker4, Karen Messer1 & Joseph A. Califano1,10 1234567890():,; Although promoter-associated CpG islands have been established as targets of DNA methylation changes in cancer, previous studies suggest that epigenetic dysregulation out- side the promoter region may be more closely associated with transcriptional changes. Here we examine DNA methylation, chromatin marks, and transcriptional alterations to define the relationship between transcriptional modulation and spatial changes in chromatin structure. Using human papillomavirus-related oropharyngeal carcinoma as a model, we show aberrant enrichment of repressive H3K9me3 at the transcriptional start site (TSS) with methylation- associated, tumor-specific gene silencing. Further analysis identifies a hypermethylated subtype which shows a functional convergence on MYC targets and association with CREBBP/EP300 mutation. The tumor-specific shift to transcriptional repression associated with DNA methylation at TSSs was confirmed in multiple tumor types. Our data may show a common underlying epigenetic dysregulation in cancer associated with broad enrichment of repressive chromatin marks and aberrant DNA hypermethylation at TSSs in combination with MYC network activation. 1 Moores Cancer Center, University of California San Diego, 3855 Health Sciences Dr, La Jolla, CA 92093, USA. -

Integrating Single-Step GWAS and Bipartite Networks Reconstruction Provides Novel Insights Into Yearling Weight and Carcass Traits in Hanwoo Beef Cattle
animals Article Integrating Single-Step GWAS and Bipartite Networks Reconstruction Provides Novel Insights into Yearling Weight and Carcass Traits in Hanwoo Beef Cattle Masoumeh Naserkheil 1 , Abolfazl Bahrami 1 , Deukhwan Lee 2,* and Hossein Mehrban 3 1 Department of Animal Science, University College of Agriculture and Natural Resources, University of Tehran, Karaj 77871-31587, Iran; [email protected] (M.N.); [email protected] (A.B.) 2 Department of Animal Life and Environment Sciences, Hankyong National University, Jungang-ro 327, Anseong-si, Gyeonggi-do 17579, Korea 3 Department of Animal Science, Shahrekord University, Shahrekord 88186-34141, Iran; [email protected] * Correspondence: [email protected]; Tel.: +82-31-670-5091 Received: 25 August 2020; Accepted: 6 October 2020; Published: 9 October 2020 Simple Summary: Hanwoo is an indigenous cattle breed in Korea and popular for meat production owing to its rapid growth and high-quality meat. Its yearling weight and carcass traits (backfat thickness, carcass weight, eye muscle area, and marbling score) are economically important for the selection of young and proven bulls. In recent decades, the advent of high throughput genotyping technologies has made it possible to perform genome-wide association studies (GWAS) for the detection of genomic regions associated with traits of economic interest in different species. In this study, we conducted a weighted single-step genome-wide association study which combines all genotypes, phenotypes and pedigree data in one step (ssGBLUP). It allows for the use of all SNPs simultaneously along with all phenotypes from genotyped and ungenotyped animals. Our results revealed 33 relevant genomic regions related to the traits of interest. -

Atherosclerosis-Susceptible and Atherosclerosis-Resistant Pigeon Aortic Cells Express Different Genes in Vivo
University of New Hampshire University of New Hampshire Scholars' Repository New Hampshire Agricultural Experiment Station Publications New Hampshire Agricultural Experiment Station 7-1-2013 Atherosclerosis-susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo Janet L. Anderson University of New Hampshire, [email protected] C. M. Ashwell University of New Hampshire - Main Campus S. C. Smith University of New Hampshire - Main Campus R. Shine University of New Hampshire - Main Campus E. C. Smith University of New Hampshire - Main Campus See next page for additional authors Follow this and additional works at: https://scholars.unh.edu/nhaes Part of the Poultry or Avian Science Commons Recommended Citation J. L. Anderson, C. M. Ashwell, S. C. Smith, R. Shine, E. C. Smith and R. L. Taylor, Jr. Atherosclerosis- susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo Poultry Science (2013) 92 (10): 2668-2680 doi:10.3382/ps.2013-03306 This Article is brought to you for free and open access by the New Hampshire Agricultural Experiment Station at University of New Hampshire Scholars' Repository. It has been accepted for inclusion in New Hampshire Agricultural Experiment Station Publications by an authorized administrator of University of New Hampshire Scholars' Repository. For more information, please contact [email protected]. Authors Janet L. Anderson, C. M. Ashwell, S. C. Smith, R. Shine, E. C. Smith, and Robert L. Taylor Jr. This article is available at University of New Hampshire Scholars' Repository: https://scholars.unh.edu/nhaes/207 Atherosclerosis-susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo J. -

Protein Kinase A-Mediated Septin7 Phosphorylation Disrupts Septin Filaments and Ciliogenesis
cells Article Protein Kinase A-Mediated Septin7 Phosphorylation Disrupts Septin Filaments and Ciliogenesis Han-Yu Wang 1,2, Chun-Hsiang Lin 1, Yi-Ru Shen 1, Ting-Yu Chen 2,3, Chia-Yih Wang 2,3,* and Pao-Lin Kuo 1,2,4,* 1 Department of Obstetrics and Gynecology, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan; [email protected] (H.-Y.W.); [email protected] (C.-H.L.); [email protected] (Y.-R.S.) 2 Institute of Basic Medical Sciences, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan; [email protected] 3 Department of Cell Biology and Anatomy, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan 4 Department of Obstetrics and Gynecology, National Cheng-Kung University Hospital, Tainan 704, Taiwan * Correspondence: [email protected] (C.-Y.W.); [email protected] (P.-L.K.); Tel.: +886-6-2353535 (ext. 5338); (C.-Y.W.)+886-6-2353535 (ext. 5262) (P.-L.K.) Abstract: Septins are GTP-binding proteins that form heteromeric filaments for proper cell growth and migration. Among the septins, septin7 (SEPT7) is an important component of all septin filaments. Here we show that protein kinase A (PKA) phosphorylates SEPT7 at Thr197, thus disrupting septin filament dynamics and ciliogenesis. The Thr197 residue of SEPT7, a PKA phosphorylating site, was conserved among different species. Treatment with cAMP or overexpression of PKA catalytic subunit (PKACA2) induced SEPT7 phosphorylation, followed by disruption of septin filament formation. Constitutive phosphorylation of SEPT7 at Thr197 reduced SEPT7-SEPT7 interaction, but did not affect SEPT7-SEPT6-SEPT2 or SEPT4 interaction. -

The Requirement of SEPT2 and SEPT7 for Migration and Invasion in Human Breast Cancer Via MEK/ERK Activation
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 7, No. 38 Research Paper The requirement of SEPT2 and SEPT7 for migration and invasion in human breast cancer via MEK/ERK activation Nianzhu Zhang1,*, Lu Liu2,*, Ning Fan2, Qian Zhang1, Weijie Wang1, Mingnan Zheng3, Lingfei Ma4, Yan Li2, Lei Shi1,5 1Institute of Cancer Stem Cell, Cancer Center, Dalian Medical University, Dalian, 116044, Liaoning, P.R.China 2College of Basic Medical Sciences, Dalian Medical University, Dalian, 116044 Liaoning, P.R.China 3Department of Gynecology and Obstetrics, Dalian Municipal Central Hospital Affiliated to Dalian Medical University, Dalian, 116033, Liaoning, P.R.China 4The First Affiliated Hospital of Dalian Medical University, Dalian, 116011, Liaoning, P.R.China 5State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, P.R.China *These authors contributed equally to this work Correspondence to: Lei Shi, email: [email protected] Yan Li, email: [email protected] Lingfei Ma, email: [email protected] Keywords: septin, forchlorfenuron, breast cancer, invasion, MAPK Received: April 24, 2016 Accepted: July 28, 2016 Published: August 19, 2016 ABSTRACT Septins are a novel class of GTP-binding cytoskeletal proteins evolutionarily conserved from yeast to mammals and have now been found to play a contributing role in a broad range of tumor types. However, their functional importance in breast cancer remains largely unclear. Here, we demonstrated that pharmaceutical inhibition of global septin dynamics would greatly suppress proliferation, migration and invasiveness in breast cancer cell lines. We then examined the expression and subcellular distribution of the selected septins SEPT2 and SEPT7 in breast cancer cells, revealing a rather variable localization of the two proteins with cell cycle progression. -
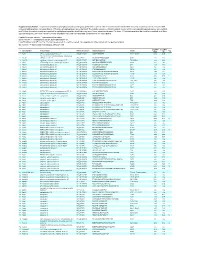
Supplementary Table 1
Supplementary Table 1. Large-scale quantitative phosphoproteomic profiling was performed on paired vehicle- and hormone-treated mTAL-enriched suspensions (n=3). A total of 654 unique phosphopeptides corresponding to 374 unique phosphoproteins were identified. The peptide sequence, phosphorylation site(s), and the corresponding protein name, gene symbol, and RefSeq Accession number are reported for each phosphopeptide identified in any one of three experimental pairs. For those 414 phosphopeptides that could be quantified in all three experimental pairs, the mean Hormone:Vehicle abundance ratio and corresponding standard error are also reported. Peptide Sequence column: * = phosphorylated residue Site(s) column: ^ = ambiguously assigned phosphorylation site Log2(H/V) Mean and SE columns: H = hormone-treated, V = vehicle-treated, n/a = peptide not observable in all 3 experimental pairs Sig. column: * = significantly changed Log 2(H/V), p<0.05 Log (H/V) Log (H/V) # Gene Symbol Protein Name Refseq Accession Peptide Sequence Site(s) 2 2 Sig. Mean SE 1 Aak1 AP2-associated protein kinase 1 NP_001166921 VGSLT*PPSS*PK T622^, S626^ 0.24 0.95 PREDICTED: ATP-binding cassette, sub-family A 2 Abca12 (ABC1), member 12 XP_237242 GLVQVLS*FFSQVQQQR S251^ 1.24 2.13 3 Abcc10 multidrug resistance-associated protein 7 NP_001101671 LMT*ELLS*GIRVLK T464, S468 -2.68 2.48 4 Abcf1 ATP-binding cassette sub-family F member 1 NP_001103353 QLSVPAS*DEEDEVPVPVPR S109 n/a n/a 5 Ablim1 actin-binding LIM protein 1 NP_001037859 PGSSIPGS*PGHTIYAK S51 -3.55 1.81 6 Ablim1 actin-binding -

Integrating Protein Copy Numbers with Interaction Networks to Quantify Stoichiometry in Mammalian Endocytosis
bioRxiv preprint doi: https://doi.org/10.1101/2020.10.29.361196; this version posted October 29, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Integrating protein copy numbers with interaction networks to quantify stoichiometry in mammalian endocytosis Daisy Duan1, Meretta Hanson1, David O. Holland2, Margaret E Johnson1* 1TC Jenkins Department of Biophysics, Johns Hopkins University, 3400 N Charles St, Baltimore, MD 21218. 2NIH, Bethesda, MD, 20892. *Corresponding Author: [email protected] bioRxiv preprint doi: https://doi.org/10.1101/2020.10.29.361196; this version posted October 29, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Abstract Proteins that drive processes like clathrin-mediated endocytosis (CME) are expressed at various copy numbers within a cell, from hundreds (e.g. auxilin) to millions (e.g. clathrin). Between cell types with identical genomes, copy numbers further vary significantly both in absolute and relative abundance. These variations contain essential information about each protein’s function, but how significant are these variations and how can they be quantified to infer useful functional behavior? Here, we address this by quantifying the stoichiometry of proteins involved in the CME network. We find robust trends across three cell types in proteins that are sub- vs super-stoichiometric in terms of protein function, network topology (e.g.