The ESCRT-III Proteins IST1 and CHMP1B Assemble Around Nucleic Acids
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mutations in CHMP2B in Lower Motor Neuron Predominant Amyotrophic Lateral Sclerosis (ALS)
This is a repository copy of Mutations in CHMP2B in lower motor neuron predominant amyotrophic lateral sclerosis (ALS). White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/10846/ Article: Cox, L.E., Ferraiuolo, L., Goodall, E.F. et al. (13 more authors) (2010) Mutations in CHMP2B in lower motor neuron predominant amyotrophic lateral sclerosis (ALS). Plos One, 5 (3). Art no.e9872. ISSN 1932-6203 https://doi.org/10.1371/journal.pone.0009872 Reuse Unless indicated otherwise, fulltext items are protected by copyright with all rights reserved. The copyright exception in section 29 of the Copyright, Designs and Patents Act 1988 allows the making of a single copy solely for the purpose of non-commercial research or private study within the limits of fair dealing. The publisher or other rights-holder may allow further reproduction and re-use of this version - refer to the White Rose Research Online record for this item. Where records identify the publisher as the copyright holder, users can verify any specific terms of use on the publisher’s website. Takedown If you consider content in White Rose Research Online to be in breach of UK law, please notify us by emailing [email protected] including the URL of the record and the reason for the withdrawal request. [email protected] https://eprints.whiterose.ac.uk/ Mutations in CHMP2B in Lower Motor Neuron Predominant Amyotrophic Lateral Sclerosis (ALS) Laura E. Cox1, Laura Ferraiuolo1, Emily F. Goodall1, Paul R. Heath1, Adrian Higginbottom1, Heather Mortiboys1, Hannah C. Hollinger1, Judith A. Hartley1, Alice Brockington1, Christine E. -

Genetic Screen Identifies Serpin5 As a Regulator of the Toll Pathway and CHMP2B Toxicity Associated with Frontotemporal Dementia
Genetic screen identifies serpin5 as a regulator of the toll pathway and CHMP2B toxicity associated with frontotemporal dementia S. Tariq Ahmada, Sean T. Sweeneyb, Jin-A Leea, Neal T. Sweeneya,c,1, and Fen-Biao Gaoa,c,2 aGladstone Institute of Neurological Disease, San Francisco, CA 94158; bDepartment of Biology, University of York, P.O. Box 373, York YO10 5YW, United Kingdom; and cNeuroscience Graduate Program, University of California, San Francisco, CA 94158 Edited by Yuh Nung Jan, University of California School of Medicine, San Francisco, CA, and approved May 18, 2009 (received for review March 23, 2009) Frontotemporal dementia (FTD) is the most common form of demen- gain-of-function mechanism. However, animal models of FTD3 tia before 60 years of age. Rare pathogenic mutations in CHMP2B, have not been reported, and the signaling pathways that are which encodes a component of the endosomal sorting complex misregulated in vivo remain to be identified. required for transport (ESCRT-III), are associated with FTD linked to In recent years, Drosophila models have been instrumental in chromosome 3 (FTD3). Animal models of FTD3 have not yet been uncovering molecular pathways that contribute to the pathogenesis reported, and what signaling pathways are misregulated by mutant of neurodegenerative diseases (17, 18). In this study, we modeled CHMP2B in vivo is unknown. Here we report the establishment of a the effect of CHMP2B in human FTD3 using a gain-of-function Drosophila model of FTD3 and show the genetic interactions between approach, which is similar to the approach that gives effects in mutant CHMP2B and other components of ESCRT. -

Progranulin Mutations in Dutch Familial Frontotemporal Lobar Degeneration
European Journal of Human Genetics (2007) 15, 369–374 & 2007 Nature Publishing Group All rights reserved 1018-4813/07 $30.00 www.nature.com/ejhg ARTICLE Progranulin mutations in Dutch familial frontotemporal lobar degeneration Iraad F Bronner1,2,4, Patrizia Rizzu*,1,2,4, Harro Seelaar3, Saskia E van Mil1,2, Burcu Anar1,2, Asma Azmani3, Laura Donker Kaat3, Sonia Rosso3, Peter Heutink1,2 and John C van Swieten3 1Department of Human Genetics, Section Medical Genomics, VU University Medical Center and VU University, Amsterdam, The Netherlands; 2Center for Neurogenomics and Cognitive Research, VU University Medical Center and VU University, Amsterdam, The Netherlands; 3Department of Neurology, Erasmus MC University Medical Center, Rotterdam, The Netherlands Mutations in the progranulin (PGRN) gene have recently been identified in frontotemporal lobar degeneration with ubiquitin inclusions linked to chromosome 17q21. We report here the finding of two novel frameshift mutations and three possible pathogenic missense mutations in the PGRN gene. Furthermore, we determined the frequency of PGRN mutations in familial cases recruited from a large population-based study of frontotemporal lobar degeneration carried out in The Netherlands. European Journal of Human Genetics (2007) 15, 369–374. doi:10.1038/sj.ejhg.5201772; published online 17 January 2007 Keywords: PGRN; FTLD; mutations; neurodegeneration Introduction dentate gyrus of the hippocampus and in the superficial The term frontotemporal lobar degeneration (FTLD) refers layers of the frontal -

The Endocytic Membrane Trafficking Pathway Plays a Major Role
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by University of Liverpool Repository RESEARCH ARTICLE The Endocytic Membrane Trafficking Pathway Plays a Major Role in the Risk of Parkinson’s Disease Sara Bandres-Ciga, PhD,1,2 Sara Saez-Atienzar, PhD,3 Luis Bonet-Ponce, PhD,4 Kimberley Billingsley, MSc,1,5,6 Dan Vitale, MSc,7 Cornelis Blauwendraat, PhD,1 Jesse Raphael Gibbs, PhD,7 Lasse Pihlstrøm, MD, PhD,8 Ziv Gan-Or, MD, PhD,9,10 The International Parkinson’s Disease Genomics Consortium (IPDGC), Mark R. Cookson, PhD,4 Mike A. Nalls, PhD,1,11 and Andrew B. Singleton, PhD1* 1Molecular Genetics Section, Laboratory of Neurogenetics, National Institute on Aging, National Institutes of Health, Bethesda, Maryland, USA 2Instituto de Investigación Biosanitaria de Granada (ibs.GRANADA), Granada, Spain 3Transgenics Section, Laboratory of Neurogenetics, National Institute on Aging, National Institutes of Health, Bethesda, Maryland, USA 4Cell Biology and Gene Expression Section, Laboratory of Neurogenetics, National Institute on Aging, National Institutes of Health, Bethesda, Maryland, USA 5Department of Molecular and Clinical Pharmacology, Institute of Translational Medicine, University of Liverpool, Liverpool, United Kingdom 6Department of Pathophysiology, University of Tartu, Tartu, Estonia 7Computational Biology Group, Laboratory of Neurogenetics, National Institute on Aging, National Institutes of Health, Bethesda, Maryland, USA 8Department of Neurology, Oslo University Hospital, Oslo, Norway 9Department of Neurology and Neurosurgery, Department of Human Genetics, McGill University, Montréal, Quebec, Canada 10Department of Neurology and Neurosurgery, Montreal Neurological Institute, McGill University, Montréal, Quebec, Canada 11Data Tecnica International, Glen Echo, Maryland, USA ABSTRACT studies, summary-data based Mendelian randomization Background: PD is a complex polygenic disorder. -

CHMP2B Regulates TDP-43 Phosphorylation and Proteotoxicity Via Modulating
bioRxiv preprint doi: https://doi.org/10.1101/2020.06.04.133546; this version posted June 5, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Linking CHMP2B to pTDP-43 via CK1_Sun et al 1 CHMP2B regulates TDP-43 phosphorylation and proteotoxicity via modulating 2 CK1 turnover independent of the autophagy-lysosomal pathway 3 Xing Sun1,2,5, Xue Deng1,2,5, Rirong Hu1,2, Yongjia Duan1, Kai Zhang1,2, Jihong Cui1, Jiangxia Ni1,2, 4 Qiangqiang Wang1, Yelin Chen1,2, Ang Li3,4*, and Yanshan Fang1,2*. 5 1Interdisciplinary Research Center on Biology and Chemistry, Shanghai Institute of Organic 6 Chemistry, Chinese Academy of Sciences, Shanghai 201210, China 7 2University of Chinese Academy of Sciences, Beijing 100049, China 8 3Guangdong-Hong Kong-Macau Institute of CNS Regeneration, Joint International Research 9 Laboratory of CNS Regeneration Ministry of Education, Jinan University, Guangzhou 510632, 10 China 11 4Guangzhou Regenerative Medicine and Health Guangdong Laboratory, Guangzhou 510005, 12 China 13 5These authors contributed equally 14 *Correspondence to: 15 Ang Li: [email protected] 16 Yanshan Fang: [email protected] 17 Tel: +86-21-6858.2510 18 ORCID: https://orcid.org/0000-0002-4123-0174 19 Short title: Linking CHMP2B to pTDP-43 via CK1 20 Keywords (5~10): ALS, FTD, ESCRT, CHMP2B, TDP-43, phosphorylation, CK1, UPS 21 This PDF file includes: 22 Main text: ~7,500 words (excluding the title page and references) 23 Figures 1 to 6 24 Supplemental Figures S1 to S6 25 Page 1 / 34 bioRxiv preprint doi: https://doi.org/10.1101/2020.06.04.133546; this version posted June 5, 2020. -

The Function and Mechanism of Chmp1a in Tumor Development
Marshall University Marshall Digital Scholar Theses, Dissertations and Capstones 1-1-2008 The uncF tion and Mechanism of Chmp1A in Tumor Development Jing Li [email protected] Follow this and additional works at: http://mds.marshall.edu/etd Part of the Biological Phenomena, Cell Phenomena, and Immunity Commons, and the Oncology Commons Recommended Citation Li, Jing, "The unctF ion and Mechanism of Chmp1A in Tumor Development" (2008). Theses, Dissertations and Capstones. Paper 120. This Thesis is brought to you for free and open access by Marshall Digital Scholar. It has been accepted for inclusion in Theses, Dissertations and Capstones by an authorized administrator of Marshall Digital Scholar. For more information, please contact [email protected]. THE FUNCTION AND MECHANISM OF CHMP1A IN TUMOR DEVELOPMENT Thesis submitted to the Graduate College of Marshall University In partial fulfillment of the requirements for the degree of Master of Science in Biological Sciences by Jing Li Committee Members: Dr. Simon Collier, Committee Chairperson Dr. Maiyon Park Dr. Guozhang Zhu Marshall University May 2008 ABSTRACT THE FUNCTION AND MECHANISM OF CHMP1A IN TUMOR DEVELOPMENT By Jing Li Chmp1A (Chromatin modifying protein 1A/Charged multivesicular protein 1A) is a member of the ESCRT-III (Endosomal Sorting Complex Required for Transport) family. ESCRT complexes play central roles in endosome mediated trafficking via MVB (multivesicular body) formation and sorting. Chmp1A is a potential tumor suppressor, especially in the pancreas. Knockdown of Chmp1A resulted in an increase of anchorage-independent growth of HEK 293T cells. Chmp1A shRNA expressing HEK 293T cells transformed these non-tumorigeneic cells to form tumors in xenograft assays. -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Supp Material.Pdf
Simon et al. Supplementary information: Table of contents p.1 Supplementary material and methods p.2-4 • PoIy(I)-poly(C) Treatment • Flow Cytometry and Immunohistochemistry • Western Blotting • Quantitative RT-PCR • Fluorescence In Situ Hybridization • RNA-Seq • Exome capture • Sequencing Supplementary Figures and Tables Suppl. items Description pages Figure 1 Inactivation of Ezh2 affects normal thymocyte development 5 Figure 2 Ezh2 mouse leukemias express cell surface T cell receptor 6 Figure 3 Expression of EZH2 and Hox genes in T-ALL 7 Figure 4 Additional mutation et deletion of chromatin modifiers in T-ALL 8 Figure 5 PRC2 expression and activity in human lymphoproliferative disease 9 Figure 6 PRC2 regulatory network (String analysis) 10 Table 1 Primers and probes for detection of PRC2 genes 11 Table 2 Patient and T-ALL characteristics 12 Table 3 Statistics of RNA and DNA sequencing 13 Table 4 Mutations found in human T-ALLs (see Fig. 3D and Suppl. Fig. 4) 14 Table 5 SNP populations in analyzed human T-ALL samples 15 Table 6 List of altered genes in T-ALL for DAVID analysis 20 Table 7 List of David functional clusters 31 Table 8 List of acquired SNP tested in normal non leukemic DNA 32 1 Simon et al. Supplementary Material and Methods PoIy(I)-poly(C) Treatment. pIpC (GE Healthcare Lifesciences) was dissolved in endotoxin-free D-PBS (Gibco) at a concentration of 2 mg/ml. Mice received four consecutive injections of 150 μg pIpC every other day. The day of the last pIpC injection was designated as day 0 of experiment. -

Identification and Characterization of Genes Essential for Human Brain Development
Identification and Characterization of Genes Essential for Human Brain Development The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Ganesh, Vijay S. 2012. Identification and Characterization of Genes Essential for Human Brain Development. Doctoral dissertation, Harvard University. Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:9773743 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA Copyright © 2012 by Vijay S. Ganesh All rights reserved. Dissertation Advisor: Dr. Christopher A. Walsh Author: Vijay S. Ganesh Identification and Characterization of Genes Essential for Human Brain Development Abstract The human brain is a network of ninety billion neurons that allows for many of the behavioral adaptations considered unique to our species. One-fifth of these neurons are layered in an epithelial sheet known as the cerebral cortex, which is exquisitely folded into convolutions called gyri. Defects in neuronal number clinically present with microcephaly (Greek for “small head”), and in inherited cases these defects can be linked to mutations that identify genes essential for neural progenitor proliferation. Most microcephaly genes are characterized to play a role in the centrosome, however rarer presentations of microcephaly have identified different mechanisms. Charged multivesicular body protein/Chromatin modifying protein 1A (CHMP1A) is a member of the ESCRT-III endosomal sorting complex, but is also suggested to localize to the nuclear matrix and regulate chromatin. -

Low Tolerance for Transcriptional Variation at Cohesin Genes Is Accompanied by Functional Links to Disease-Relevant Pathways
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.11.037358; this version posted April 13, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Title Low tolerance for transcriptional variation at cohesin genes is accompanied by functional links to disease-relevant pathways Authors William Schierdingǂ1, Julia Horsfieldǂ2,3, Justin O’Sullivan1,3,4 ǂTo whom correspondence should be addressed. 1 Liggins Institute, The University of Auckland, Auckland, New Zealand 2 Department of Pathology, Dunedin School of Medicine, University of Otago, Dunedin, New Zealand 3 The Maurice Wilkins Centre for Biodiscovery, The University of Auckland, Auckland, New Zealand 4 MRC Lifecourse Epidemiology Unit, University of Southampton Acknowledgements This work was supported by a Royal Society of New Zealand Marsden Grant to JH and JOS (16-UOO- 072), and WS was supported by the same grant. Contributions WS planned the study, performed analyses, and drafted the manuscript. JH and JOS revised the manuscript. Competing interests None declared. bioRxiv preprint doi: https://doi.org/10.1101/2020.04.11.037358; this version posted April 13, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract Variants in DNA regulatory elements can alter the regulation of distant genes through spatial- regulatory connections. -

Analysis of the Indacaterol-Regulated Transcriptome in Human Airway
Supplemental material to this article can be found at: http://jpet.aspetjournals.org/content/suppl/2018/04/13/jpet.118.249292.DC1 1521-0103/366/1/220–236$35.00 https://doi.org/10.1124/jpet.118.249292 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS J Pharmacol Exp Ther 366:220–236, July 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Analysis of the Indacaterol-Regulated Transcriptome in Human Airway Epithelial Cells Implicates Gene Expression Changes in the s Adverse and Therapeutic Effects of b2-Adrenoceptor Agonists Dong Yan, Omar Hamed, Taruna Joshi,1 Mahmoud M. Mostafa, Kyla C. Jamieson, Radhika Joshi, Robert Newton, and Mark A. Giembycz Departments of Physiology and Pharmacology (D.Y., O.H., T.J., K.C.J., R.J., M.A.G.) and Cell Biology and Anatomy (M.M.M., R.N.), Snyder Institute for Chronic Diseases, Cumming School of Medicine, University of Calgary, Calgary, Alberta, Canada Received March 22, 2018; accepted April 11, 2018 Downloaded from ABSTRACT The contribution of gene expression changes to the adverse and activity, and positive regulation of neutrophil chemotaxis. The therapeutic effects of b2-adrenoceptor agonists in asthma was general enriched GO term extracellular space was also associ- investigated using human airway epithelial cells as a therapeu- ated with indacaterol-induced genes, and many of those, in- tically relevant target. Operational model-fitting established that cluding CRISPLD2, DMBT1, GAS1, and SOCS3, have putative jpet.aspetjournals.org the long-acting b2-adrenoceptor agonists (LABA) indacaterol, anti-inflammatory, antibacterial, and/or antiviral activity. Numer- salmeterol, formoterol, and picumeterol were full agonists on ous indacaterol-regulated genes were also induced or repressed BEAS-2B cells transfected with a cAMP-response element in BEAS-2B cells and human primary bronchial epithelial cells by reporter but differed in efficacy (indacaterol $ formoterol . -
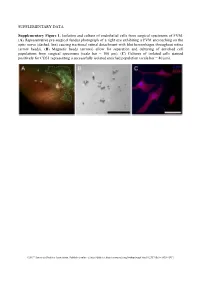
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1.