Acetonitrile (CAS #75-05-8) Information for the Public
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Use of Solvents for Pahs Extraction and Enhancement of the Pahs Bioremediation in Coal- Tar-Contaminated Soils Pak-Hing Lee Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 2000 Use of solvents for PAHs extraction and enhancement of the PAHs bioremediation in coal- tar-contaminated soils Pak-Hing Lee Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Environmental Engineering Commons Recommended Citation Lee, Pak-Hing, "Use of solvents for PAHs extraction and enhancement of the PAHs bioremediation in coal-tar-contaminated soils " (2000). Retrospective Theses and Dissertations. 13912. https://lib.dr.iastate.edu/rtd/13912 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORMATION TO USERS This manuscript has been reproduced from the microfilm master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter fece, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quaiity of the copy submitted. Broken or indistinct print colored or poor quality illustrations and photographs, print bleedthrough, substeindard margins, and improper alignment can adversely affect reproduction. In the unlilcely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. -

Acute Toxicity of Cyanide in Aerobic Respiration: Theoretical and Experimental Support for Murburn Explanation
BioMol Concepts 2020; 11: 32–56 Research Article Open Access Kelath Murali Manoj*, Surjith Ramasamy, Abhinav Parashar, Daniel Andrew Gideon, Vidhu Soman, Vivian David Jacob, Kannan Pakshirajan Acute toxicity of cyanide in aerobic respiration: Theoretical and experimental support for murburn explanation https://doi.org/10.1515/bmc-2020-0004 received January 14, 2020; accepted February 19, 2020. of diffusible radicals and proton-equilibriums, explaining analogous outcomes in mOxPhos chemistry. Further, we Abstract: The inefficiency of cyanide/HCN (CN) binding demonstrate that superoxide (diffusible reactive oxygen with heme proteins (under physiological regimes) is species, DROS) enables in vitro ATP synthesis from demonstrated with an assessment of thermodynamics, ADP+phosphate, and show that this reaction is inhibited kinetics, and inhibition constants. The acute onset of by CN. Therefore, practically instantaneous CN ion-radical toxicity and CN’s mg/Kg LD50 (μM lethal concentration) interactions with DROS in matrix catalytically disrupt suggests that the classical hemeFe binding-based mOxPhos, explaining the acute lethal effect of CN. inhibition rationale is untenable to account for the toxicity of CN. In vitro mechanistic probing of Keywords: cyanide-poisoning; murburn concept; CN-mediated inhibition of hemeFe reductionist systems aerobic respiration; ATP-synthesis; hemoglobin; was explored as a murburn model for mitochondrial cytochrome oxidase; mitochondria; diffusible reactive oxidative phosphorylation (mOxPhos). The effect of CN oxygen species (DROS). in haloperoxidase catalyzed chlorine moiety transfer to small organics was considered as an analogous probe for phosphate group transfer in mOxPhos. Similarly, Introduction inclusion of CN in peroxidase-catalase mediated one- electron oxidation of small organics was used to explore Since the discovery of the dye Prussian blue (ferric electron transfer outcomes in mOxPhos, leading to water ferrocyanide, Fe7[CN]18) and the volatile prussic acid formation. -

EPA Method 8315A (SW-846): Determination of Carbonyl Compounds by High Performance Liquid Chromatography (HPLC)
METHOD 8315A DETERMINATION OF CARBONYL COMPOUNDS BY HIGH PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC) 1.0 SCOPE AND APPLICATION 1.1 This method provides procedures for the determination of free carbonyl compounds in various matrices by derivatization with 2,4-dinitrophenylhydrazine (DNPH). The method utilizes high performance liquid chromatography (HPLC) with ultraviolet/visible (UV/vis) detection to identify and quantitate the target analytes. This method includes two procedures encompassing all aspects of the analysis (extraction to determination of concentration). Procedure 1 is appropriate for the analysis of aqueous, soil and waste samples and stack samples collected by Method 0011. Procedure 2 is appropriate for the analysis of indoor air samples collected by Method 0100. The list of target analytes differs by procedure. The appropriate procedure for each target analyte is listed in the table below. Compound CAS No. a Proc. 1b Proc. 2 b Acetaldehyde 75-07-0 X X Acetone 67-64-1 X Acrolein 107-02-8 X Benzaldehyde 100-52-7 X Butanal (Butyraldehyde) 123-72-8 X X Crotonaldehyde 123-73-9 X X Cyclohexanone 108-94-1 X Decanal 112-31-2 X 2,5-Dimethylbenzaldehyde 5779-94-2 X Formaldehyde 50-00-0 X X Heptanal 111-71-7 X Hexanal (Hexaldehyde) 66-25-1 X X Isovaleraldehyde 590-86-3 X Nonanal 124-19-6 X Octanal 124-13-0 X Pentanal (Valeraldehyde) 110-62-3 X X Propanal (Propionaldehyde) 123-38-6 X X m-Tolualdehyde 620-23-5 X X o-Tolualdehyde 529-20-4 X p-Tolualdehyde 104-87-0 X a Chemical Abstract Service Registry Number. -

Hazardous Chemicals in Secondhand Marijuana Smoke
Hazardous Chemicals in Secondhand Marijuana Smoke “The following 33 marijuana smoke constituents included in Table 1 are listed under 33 Chemicals Proposition 65 as causing cancer: acetaldehyde, acetamide, acrylonitrile, 4- aminobiphenyl, arsenic, benz[a]anthracene, benzene, benzo[a]pyrene, That Can benzo[b]fluoranthene, benzo[j]fluoranthene, benzo[k]fluoranthene, benzofuran, 1,3- butadiene, cadmium, carbazole, catechol, chromium (hexavalent compounds), Cancer chrysene, dibenz[a,h]anthracene, dibenz[a,i]pyrene, dibenzo[a,e]pyrene, “Many of the chemical diethylnitrosamine, dimethylnitrosamine, formaldehyde, indeno[1,2,3,-c,d]pyrene, constituents that have been isoprene, lead, mercury, 5-methylchrysene, naphthalene, nickel, pyridine, and identified in marijuana smoke quinoline.” are carcinogens.” 2009 OEHHA document, Evidence on the Carcinogenicity of Marijuana Smoke Hydrogen Cyanide interferes with the normal use of oxygen by nearly every organ of Hydrogen the body. Exposure to hydrogen cyanide (AC) can be rapidly fatal. It has whole-body (systemic) effects, particularly affecting those organ systems most sensitive to low Cyanide oxygen levels: the central nervous system (brain), the cardiovascular system (heart Is the same chemical used for and blood vessels), and the pulmonary system (lungs). Hydrogen cyanide (AC) is a chemical weapons. chemical warfare agent (military designation, AC). Ammonia gas is a severe respiratory tract irritant. Can cause severe irritation of the Ammonia nose and throat. Can cause life-threatening accumulation of fluid in the lungs Household cleaner used on (pulmonary edema). Symptoms may include coughing, shortness of breath, difficult floors and toilets. There is 3 breathing and tightness in the chest. Symptoms may develop hours after exposure times more in secondhand and are made worse by physical effort. -
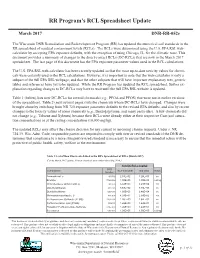
RR Program's RCL Spreadsheet Update
RR Program’s RCL Spreadsheet Update March 2017 RR Program RCL Spreadsheet Update DNR-RR-052e The Wisconsin DNR Remediation and Redevelopment Program (RR) has updated the numerical soil standards in the August 2015 DNR-RR- 052b RR spreadsheet of residual contaminant levels (RCLs). The RCLs were determined using the U.S. EPA RSL web- calculator by accepting EPA exposure defaults, with the exception of using Chicago, IL, for the climatic zone. This documentThe U.S. provides EPA updateda summary its Regionalof changes Screening to the direct-contact Level (RSL) RCLs website (DC-RCLs) in June that2015. are To now reflect in the that March 2017 spreadsheet.update, the The Wisconsin last page ofDNR this updated document the has numerical the EPA exposuresoil standards, parameter or residual values usedcontaminant in the RCL levels calculations. (RCLs), in the Remediation and Redevelopment program’s spreadsheet of RCLs. This document The providesU.S. EPA a RSL summary web-calculator of the updates has been incorporated recently updated in the Julyso that 2015 the spreadsheet.most up-to-date There toxicity were values no changes for chemi - cals madewere certainlyto the groundwater used in the RCLs,RCL calculations. but there are However, many changes it is important in the industrial to note that and the non-industrial web-calculator direct is only a subpartcontact of the (DC) full RCLsEPA RSL worksheets. webpage, Tables and that 1 andthe other 2 of thissubparts document that will summarize have important the DC-RCL explanatory changes text, generic tablesfrom and the references previous have spreadsheet yet to be (Januaryupdated. -

Qualitative Chemical Measures to Indirectly Assess Quality of Natural/Uncured, Organic Bacon Compared to Traditionally Cured Bacon
Iowa State University Capstones, Theses and Graduate Theses and Dissertations Dissertations 2009 Qualitative chemical measures to indirectly assess quality of natural/uncured, organic bacon compared to traditionally cured bacon. Charlwit Kulchaiyawat Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/etd Part of the Animal Sciences Commons Recommended Citation Kulchaiyawat, Charlwit, "Qualitative chemical measures to indirectly assess quality of natural/uncured, organic bacon compared to traditionally cured bacon." (2009). Graduate Theses and Dissertations. 10999. https://lib.dr.iastate.edu/etd/10999 This Thesis is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Graduate Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. Qualitative chemical measures to indirectly assess quality of natural/uncured, organic bacon compared to traditionally cured bacon by Charlwit Kulchaiyawat A thesis submitted to the graduate faculty in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE Major: Meat Science Program of Study Committee: Joseph Sebranek, Major Professor James Dickson Mark Honeyman Lester Wilson Iowa State University Ames, Iowa 2009 Copyright © Charlwit Kulchaiyawat 2009. All rights reserved. ii TABLE OF CONTENTS LIST OF TABLES iii ABSTRACT -

Acetonitrile
Fact Sheet Acetonitrile What is Acetonitrile Acetonitrile is a toxic, colorless liquid with an ether-like odor and a sweet, burnt taste. It is an extremely dangerous substance and must be handled with caution as it can cause severe health effects and/or death. It is also known as cyanomethane, ethyl nitrile, ethanenitrile, methanecarbonitrile, acetronitrile cluster and methyl cyanide. Acetonitrile is easily ignited by heat, sparks or flames and gives off highly toxic hydrogen cyanide fumes when heated. It dissolves easily in water. It can react with water, steam or acids to produce flammable vapors that can from explosive mixtures when exposed to air. The vapors are heavier than air and can travel to low or confined areas. Containers of the liquid can explode when heated. Acetonitrile is used to make pharmaceuticals, perfumes, rubber products, pesticides, acrylic nail removers and batteries. It is also used to extract fatty acids from animal and vegetable oils. Before working with acetonitrile, employee training must be provided on safe handling and storage procedures. Pregnant women should avoid contact with acetonitrile. Exposure Exposure usually occurs in the industries where acetonitrile is produced or used. Though unlikely, the general population may be exposed by due to chemical spills, accidents or releases. It is also found in cigarette smoke and automobile exhaust. Acetonitrile changes to cyanide with the body. Route of exposure can occur by: . Breathing ─Inhalation of acetonitrile vapors can cause adverse health effects. Eating/Drinking ─Ingestion of acetonitrile is not a likely route of exposure due to its irritating effects. Skin/Eye Contact ─Acetonitrile vapors and liquids can be absorbed through the skin or eyes. -

An Extractive Distillation System for Benzene-Acetonitrile Separation Using Dimethyl Sulfoxide As an Entrainer Bloch Sohil Y
An Extractive Distillation System for Benzene-Acetonitrile Separation Using Dimethyl Sulfoxide as an Entrainer Bloch Sohil Y. & Mehulkumar Sutariya Sardar Vallabhbhai National Institute of Technology, Surat Email: [email protected] Unit System: Pressure-Kpa; Molar Flow-kg/hr; Other-SI Background Hydrocarbons like Acetonitrile and Benzene are important raw materials in the manufacturing of polymeric products and as a solvent. They often require high-purity Acetonitrile and Benzene. Acetonitrile is widely used mainly as a solvent in the purification of butadiene in refineries it is widely used in battery applications because of its relatively high dielectric constant and ability to dissolve electrolytes. For similar reasons it is a popular solvent in cyclic voltammetry. Acetonitrile plays a significant role as the dominant solvent used in the manufacture of DNA oligonucleotides from monomers.Industrially, it is used as a solvent for the manufacture of pharmaceuticals and photographic film.The mixer of Acetonitrile and Benzene can not be seperated out by the simple distillation column because of the less difference between their boiling point (near 1.5 K) and same boiling point behaviour of azeotrope. Extractive Distillation of Close Boiling Compounds Extractive distillation is the method of separating close boiling compounds from each other by carrying out the distillation in a multiple columns in the presence of an added liquid or liquid mixture.1 This Liquid or Liquid mixture is known as extractive agent or entrainer. The presence of the entrainer alter the volatility of compounds and thus the degree of separation is increase with the same numbers of plate. This entrainer must have high boiling point than the compounds which are going to separated. -

NMR Chemical Shifts of Common Laboratory Solvents As Trace Impurities
7512 J. Org. Chem. 1997, 62, 7512-7515 NMR Chemical Shifts of Common Laboratory Solvents as Trace Impurities Hugo E. Gottlieb,* Vadim Kotlyar, and Abraham Nudelman* Department of Chemistry, Bar-Ilan University, Ramat-Gan 52900, Israel Received June 27, 1997 In the course of the routine use of NMR as an aid for organic chemistry, a day-to-day problem is the identifica- tion of signals deriving from common contaminants (water, solvents, stabilizers, oils) in less-than-analyti- cally-pure samples. This data may be available in the literature, but the time involved in searching for it may be considerable. Another issue is the concentration dependence of chemical shifts (especially 1H); results obtained two or three decades ago usually refer to much Figure 1. Chemical shift of HDO as a function of tempera- more concentrated samples, and run at lower magnetic ture. fields, than today’s practice. 1 13 We therefore decided to collect H and C chemical dependent (vide infra). Also, any potential hydrogen- shifts of what are, in our experience, the most popular bond acceptor will tend to shift the water signal down- “extra peaks” in a variety of commonly used NMR field; this is particularly true for nonpolar solvents. In solvents, in the hope that this will be of assistance to contrast, in e.g. DMSO the water is already strongly the practicing chemist. hydrogen-bonded to the solvent, and solutes have only a negligible effect on its chemical shift. This is also true Experimental Section for D2O; the chemical shift of the residual HDO is very NMR spectra were taken in a Bruker DPX-300 instrument temperature-dependent (vide infra) but, maybe counter- (300.1 and 75.5 MHz for 1H and 13C, respectively). -

Nitric Oxide.Vp
CHAPTER 5 Maple Leaf Foods, Toronto, Canada Brown Foundation Institute of Molecular Medicine, The University of Texas Health Science Center, Houston, TX 77030 ESPITE the many published reports on the beneficial properties of Dnitrite and nitrate in physiology, nitrite and nitrate in cured and pro- cessed meats continues to be perceived as harmful. The previous chapter revealed that certain foods, particularly green leafy vegetables are natu- rally enriched in nitrite and nitrate from growing in soil. However, en- riching meats with nitrite or nitrate during curing is perceived as harmful and advocated by some groups to be eliminated completely. The use of pure sodium nitrate in curing is now only a minor practice in the United States. The ingoing levels of sodium nitrite have been tightly controlled by the Food Safety and Inspection Service (FSIS) of the U.S. Depart- ment of Agriculture. To satisfy consumer demands for no added nitrite processed meats, efforts have recently been taken to creatively adjust the meat curing process by employing “nitrite free” organic vegetable pow- ders instead of directly adding sodium nitrite salts. Although the end re- sult is production of nitrite from the nitrate contained in the vegetable powders, a more “natural” or “organic” approach seems to appeal to con- sumers. This is primarily due to the public perception of nitrite and ni- trate. Reports about methemoglobinemia in infants (blue baby syndrome) caused by drinks or food prepared with nitrate-rich (and bac- terially contaminated) well water and vegetables, intentional and occu- pational intoxications in adults, increasing nitrate levels in soil and lakes as a result of fertilizer overuse, and the formation of potentially carcino- genic N-nitrosamines all contribute to the negative image that nitrite and nitrate have held in recent years. -

Stability Studies of Selected Polycyclic Aromatic Hydrocarbons in Different Organic Solvents and Identification of Their Transformation Products
Polish J. of Environ. Stud. Vol. 17, No. 1 (2008), 17-24 Original Research Stability Studies of Selected Polycyclic Aromatic Hydrocarbons in Different Organic Solvents and Identification of Their Transformation Products D. Dąbrowska1, A. Kot-Wasik2, J. Namieśnik*2 1Polish geological institute, Central Chemical laboratory, warsaw, Poland 2Department of Analytical Chemistry, gdansk university of technology, ul. Narutowicza 11/12, 80-892 gdańsk, Poland Received: May 7, 2007 Accepted: October 1, 2007 Abstract one of the problems in an hPlC laboratory is the preservation of samples and extracts prior to analysis without any changes. It has been found that degradation processes cannot be eliminated entirely, but they can be slowed down considerably. Photodegradation kinetics of fluorene, anthracene and benzo(a)pyrene were studied in various organic solvents. Solvents known as good media to store PAhs for a long time were selected with respect to avoid degradation. in the case of fluorene, 9-fluorenone and 9-hydroxyfluorene were detected as main photoproducts. Formation of anthraquinone and 1,8-dihydroxyanthraquinone during the degradation of anthracene was observed. Benzo(a)pyrene-4,5-dihydrodiol and one of the isomers of hydroxy-BaP-dione as products of benzo(a)pyrene photodegradation have been identified. Keywords: polycyclic aromatic hydrocarbons, photodegradation, degradation products, sample stability Introduction 4 or more rings and their metabolites, have a variety of mutagenic and carcinogenic effects on microorganisms, Polycyclic aromatic hydrocarbons (PAhs) are ubiq- plants and animals, and are classified as compounds with uitous contaminants originating from natural and anthro- significant human health risks [6]. pogenic pyrolysis of organic matter such as forest fires, In the environment, primary removal processes of low automobile exhaust, coal and oil refining processes.t heir molecular weight PAhs are microbial degradation and abundance and persistence in several polluted environ- evaporation. -

Sep-Pak Xposure Aldehyde Sampler
[ CARE CARE AND AND USE USE MANUAL MANUAL ] ] Sep-Pak XPoSure Aldehyde Sampler CONTENTS I. INTRODUCTION I. INTRODUCTION Waters Sep-Pak® XPoSure™ Aldehyde Samplers are convenient, a. Sep-Pak DNPH-Silica Cartridge Description reproducible sampling devices for quantifying formaldehyde concentrations in the workplace and indoor air within a range of II. USING THE SEP-PAK XPOSURE 0.001 to 5 parts per million (ppmv). ALDEHYDE SAMPLER a. Theory of Operation b. Preventing Contamination c. Collecting the Sample d. Eluting the Derivatives from the Sampler III. ANALYZING THE DNPH DERIVATIVES a. Theory of Operation b. Preventing Contamination c. Collecting the Sample d. Calculating Results IV. APPLICATION EXAMPLES a. Formaldehyde in Laboratory Air - STEL b. Glutaraldehyde in Laboratory Air – STEL V. STORAGE AND DISPOSAL OF THE SAMPLERS a. Storing Unused Samplers b. Storing Exposed Samplers c. Disposing of used cartridges VI. TROUBLESHOOTING VII. REFERENCES AND BIBLIOGRAPHY VIII. ORDERING INFORMATION IX. APPENDICES a. Appendix A: Measuring Acetontrile Purity b. Appendix B: Synthesizing the DNPH-Derivative Standards c. Appendix C: Measuring Breakthrough d. Appendix D: Useful Conversion Factors d.1. Carbonyl to Hydrazone Conversion Factors d.2. Equation for converting µg/L to ppmv d.3. Conversion Factors: µg/L to ppmv [ CARE AND USE MANUAL ] [ CARE AND USE MANUAL ] a. Sep-Pak XPoSure Aldehyde Sampler Description II. USING THE SEP-PAK XPOSURE and Features ALDEHYDE SAMPLER ■■ Sep-Pak XPoSure Aldehyde Samplers contain acidified a. Theory of Operation 2,4-Dinitrophenylhydrazine(DNPH)-coated silica, packed Sep-Pak XPoSure Aldehyde Samplers trap aldehydes in air by in Waters Sep-Pak Plus cartridges. reacting them with acidified 2,4-dinitrophenylhydrazine(DNPH), ■■ The samplers are constructed from high-purity and high- forming stable hydrazone derivatives.