Heme a • Heme a Is a Heme Protein That Is a Coordination Complex
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chapter 5 High-Pressure Stopped-Flow Kinetic Studies of Nnos
Probing the dynamics and conformational landscape of neuronal nitric oxide synthase A thesis submitted to the University of Manchester for the degree of Doctor of Philosophy in the Faculty of Life Sciences 2013 Anna Sobolewska-Stawiarz Contents Contents ...................................................................................................................... 2 List of Figures ............................................................................................................. 6 List of Tables ............................................................................................................ 10 Abstract ..................................................................................................................... 12 Declaration ................................................................................................................ 13 Copyright Statement ................................................................................................ 13 Acknowledgements ................................................................................................... 14 List of Abbreviations ............................................................................................... 15 List of Amino Acids Abbreviations ........................................................................ 17 CHAPTER 1. INTRODUCTION ........................................................................... 18 1.1 Nitric oxide synthase ......................................................................................... -

Download English-US Transcript (PDF)
MITOCW | watch?v=56vQ0S2eAjw SPEAKER 1: The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To make a donation or view additional materials from hundreds of MIT courses, visit MIT OpenCourseWare at ocw.mit.edu. PROFESSOR: Today what I want to do within the lexicon is tell you about nature's most spectacularly beautiful cofactors. And these are formed from vitamin B-12, which you find in your vitamin bottle. OK. So what is the structure of vitamin B-12, and why do I say they are spectacularly beautiful? So it's very hard to see, but if you look at the structure of this, where have you seen a molecule this complicated with five membered rings, each of which has a nitrogen in this? You've seen this when you studied hemoglobin, and you think about heme and proto protoporphyrin IX. If you look at the biosynthetic pathway of heme, a branchpoint of that pathway is to make this ring, which is found in adenosylcobalamin and methylcobalamin, which is what we're going to be focusing on today. And this ring is called the corrin ring. So what I want to do is introduce you a little bit to this corrin ring and what's unusual about it compared to protoporphyrin IX that you've seen before. So the vitamin, as in the case of all vitamins that we've talked about over the course of the semester, is not the actual cofactor used in the enzymatic transformation. -

Design and Fine-Tuning Redox Potentials of Metalloproteins Involved in Electron Transfer in Bioenergetics
1 Design and Fine-tuning Redox Potentials of Metalloproteins Involved in Electron Transfer in Bioenergetics Parisa Hosseinzadeh and Yi Lu Abstract: 1. Introduction A significant portion of biological processes are involved with providing vital energy sources such as ATP, and controlling the flow of energy through living systems. These bioenergetics processes, the most important of which are photosynthesis and respiration, require electron transfer (ET) between different redox partners. Metalloproteins are one of the most widely used ET centers in biology. They can be classified into three major classes: cupredoxins, which include type 1 copper (T1Cu) proteins and CuA centers [1- 10], cytochromes [10-17], and iron-sulfur (FeS) proteins [10,18-24]. Each class of ET proteins transfer electrons between different redox partners which possess different reduction potentials (E°) (Figure 1). Therefore, the ET centers need to adjust their E° in a way that matches those of their redox partners. No single class of protein can cover the entire range of physiological E°, which is between ~ -1V, at which protons are reduced to H2, and 1V, at which water is oxidized to O2. Cupredoxins usually function at the high end of the E°, while FeS proteins are mostly involved in ET reactions possessing relatively low E° [10]. The E°’s of FeS proteins overlap significantly with those of cytochromes that often have intermediate E° among the three classes of ET proteins. Figure 1. Reduction potential range of metal centers in electron transfer metalloprotein. Adapted from ref. [10] 2 In this review, we first describe the importance of tuning E° of ET centers, including the metalloproteins described above. -

Iron and Chelation in Biochemistry and Medicine: New Approaches to Controlling Iron Metabolism and Treating Related Diseases
cells Review Iron and Chelation in Biochemistry and Medicine: New Approaches to Controlling Iron Metabolism and Treating Related Diseases George J. Kontoghiorghes * and Christina N. Kontoghiorghe Postgraduate Research Institute of Science, Technology, Environment and Medicine, CY-3021 Limassol, Cyprus * Correspondence: [email protected]; Tel./Fax: +357-2627-2076 Received: 7 May 2020; Accepted: 5 June 2020; Published: 12 June 2020 Abstract: Iron is essential for all living organisms. Many iron-containing proteins and metabolic pathways play a key role in almost all cellular and physiological functions. The diversity of the activity and function of iron and its associated pathologies is based on bond formation with adjacent ligands and the overall structure of the iron complex in proteins or with other biomolecules. The control of the metabolic pathways of iron absorption, utilization, recycling and excretion by iron-containing proteins ensures normal biologic and physiological activity. Abnormalities in iron-containing proteins, iron metabolic pathways and also other associated processes can lead to an array of diseases. These include iron deficiency, which affects more than a quarter of the world’s population; hemoglobinopathies, which are the most common of the genetic disorders and idiopathic hemochromatosis. Iron is the most common catalyst of free radical production and oxidative stress which are implicated in tissue damage in most pathologic conditions, cancer initiation and progression, neurodegeneration and many other diseases. The interaction of iron and iron-containing proteins with dietary and xenobiotic molecules, including drugs, may affect iron metabolic and disease processes. Deferiprone, deferoxamine, deferasirox and other chelating drugs can offer therapeutic solutions for most diseases associated with iron metabolism including iron overload and deficiency, neurodegeneration and cancer, the detoxification of xenobiotic metals and most diseases associated with free radical pathology. -

SCHELVIS CV Profile 2010
Curriculum vitae: Johannes Schelvis 09/7/2010 PERSONAL INFORMATION Johannes P. M. Schelvis, Associate Professor Montclair State University Department of Chemistry and Biochemistry 1 Normal Avenue Montclair, NJ 07043 EDUCATION B.S., Physics, 1985, Free University, Amsterdam, Netherlands Ph.D., Biophysics, 1995, University of Leiden, Leiden, Netherlands PROFESSIONAL EXPERIENCE Associate Professor Montclair State University September 2007 – present Assistant Professor New York University September 2000 – August 2007 Postdoctoral Researcher Michigan State University March 1995 - August 2000 HONORS AND AWARDS • Institute Fellow, Margaret and Herman Sokol Institute for the Pharmaceutical Life Sciences at Montclair State University, September 2008 - present • Goddard Fellowship, New York University, 2004 • Whitehead Fellowship for Junior Faculty in Biomedical or Biological Sciences, New York University, 2003. GRANTS AWARDED ACTIVE • "Molecular Mechanisms of Photolyase and Cryptochrome" National Science Foundation, MCB-0920013, August 2009 – July 2012 , $419,453 t.c. (PI) • "Binding of ICER to Its Own Promoter as a Mode of Cooperative Regulation" Margaret and Herman Sokol Institute for Pharmaceutical Life Sciences, September 2008 – August 2011 (1-year no cost extension), $100,000 (PI with Dr. Carlos Molina) • "Light-Driven Damage and Repair of DNA", Faculty Scholarship Program, Montclair State University, 2008 – 2012 , 6 TCH (PI) COMPLETED • "Fingerprinting DNA Damage" Margaret and Herman Sokol Faculty/Student Research Grant Program, July 2008 -
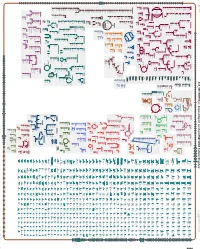
Generate Metabolic Map Poster
Authors: Pallavi Subhraveti Ron Caspi Quang Ong Peter D Karp An online version of this diagram is available at BioCyc.org. Biosynthetic pathways are positioned in the left of the cytoplasm, degradative pathways on the right, and reactions not assigned to any pathway are in the far right of the cytoplasm. Transporters and membrane proteins are shown on the membrane. Ingrid Keseler Periplasmic (where appropriate) and extracellular reactions and proteins may also be shown. Pathways are colored according to their cellular function. Gcf_900114035Cyc: Amycolatopsis sacchari DSM 44468 Cellular Overview Connections between pathways are omitted for legibility. -

Characterisation, Classification and Conformational Variability Of
Characterisation, Classification and Conformational Variability of Organic Enzyme Cofactors Julia D. Fischer European Bioinformatics Institute Clare Hall College University of Cambridge A thesis submitted for the degree of Doctor of Philosophy 11 April 2011 This dissertation is the result of my own work and includes nothing which is the outcome of work done in collaboration except where specifically indicated in the text. This dissertation does not exceed the word limit of 60,000 words. Acknowledgements I would like to thank all the members of the Thornton research group for their constant interest in my work, their continuous willingness to answer my academic questions, and for their company during my time at the EBI. This includes Saumya Kumar, Sergio Martinez Cuesta, Matthias Ziehm, Dr. Daniela Wieser, Dr. Xun Li, Dr. Irene Pa- patheodorou, Dr. Pedro Ballester, Dr. Abdullah Kahraman, Dr. Rafael Najmanovich, Dr. Tjaart de Beer, Dr. Syed Asad Rahman, Dr. Nicholas Furnham, Dr. Roman Laskowski and Dr. Gemma Holli- day. Special thanks to Asad for allowing me to use early development versions of his SMSD software and for help and advice with the KEGG API installation, to Roman for knowing where to find all kinds of data, to Dani for help with R scripts, to Nick for letting me use his E.C. tree program, to Tjaart for python advice and especially to Gemma for her constant advice and feedback on my work in all aspects, in particular the chemistry side. Most importantly, I would like to thank Prof. Janet Thornton for giving me the chance to work on this project, for all the time she spent in meetings with me and reading my work, for sharing her seemingly limitless knowledge and enthusiasm about the fascinating world of enzymes, and for being such an experienced and motivational advisor. -

Definition of the Catalytic Site of Cytochrome C Oxidase: Specific Ligands of Heme a and the Heme A3-CUB Center JAMES P
Proc. Natl. Acad. Sci. USA Vol. 89, pp. 4786-4790, June 1992 Biochemistry Definition of the catalytic site of cytochrome c oxidase: Specific ligands of heme a and the heme a3-CUB center JAMES P. SHAPLEIGH*, JONATHAN P. HOSLERt, MARY M. J. TECKLENBURGO§, YOUNKYOO KIMt, GERALD T. BABCOCKt, ROBERT B. GENNIS*, AND SHELAGH FERGUSON-MILLERt *School of Chemical Sciences, University of Illinois, Urbana, IL 61801; and Departments of tBiochemistry and tChemistry, Michigan State University, East Lansing, MI 48824 Communicated by N. Edward Tolbert, February 6, 1992 (receivedfor review November 17, 1991) ABSTRACT The three-subunit aa3-type cytochrome c ox- and deduced amino acid sequences are very similar to those idase (EC 1.9.3.1) of Rhodobacter sphaeroides is structurally from the closely related bacterium Paracoccus denitrificans and functionally homologous to the more complex mitochon- (2, 10-12). In addition, subunit I shows 50% sequence drial oxidase. The largest subunit, subunit I, is highly con- identity to subunit I ofbovine heart cytochrome c oxidase (9). served and predicted to contain 12 transmembrane segments Hydropathy profile analysis of subunit I from Rb. sphae- that provide all the ligands for three of the four metal centers: roides is consistent with 12 transmembrane helical spans, as heme a, heme a3, and CUB. A variety of spectroscopic tech- previously proposed for bovine subunit I (11). The two- niques identify these ligands as histidines. We have used dimensional model that emerges from this analysis (Fig. 1) site-directed mutagenesis to change all the conserved histidines highlights seven histidine residues, six of which are found to within subunit I of cytochrome c oxidase from Rb. -

Cheminformatics for Genome-Scale Metabolic Reconstructions
CHEMINFORMATICS FOR GENOME-SCALE METABOLIC RECONSTRUCTIONS John W. May European Molecular Biology Laboratory European Bioinformatics Institute University of Cambridge Homerton College A thesis submitted for the degree of Doctor of Philosophy June 2014 Declaration This thesis is the result of my own work and includes nothing which is the outcome of work done in collaboration except where specifically indicated in the text. This dissertation is not substantially the same as any I have submitted for a degree, diploma or other qualification at any other university, and no part has already been, or is currently being submitted for any degree, diploma or other qualification. This dissertation does not exceed the specified length limit of 60,000 words as defined by the Biology Degree Committee. This dissertation has been typeset using LATEX in 11 pt Palatino, one and half spaced, according to the specifications defined by the Board of Graduate Studies and the Biology Degree Committee. June 2014 John W. May to Róisín Acknowledgements This work was carried out in the Cheminformatics and Metabolism Group at the European Bioinformatics Institute (EMBL-EBI). The project was fund- ed by Unilever, the Biotechnology and Biological Sciences Research Coun- cil [BB/I532153/1], and the European Molecular Biology Laboratory. I would like to thank my supervisor, Christoph Steinbeck for his guidance and providing intellectual freedom. I am also thankful to each member of my thesis advisory committee: Gordon James, Julio Saez-Rodriguez, Kiran Patil, and Gos Micklem who gave their time, advice, and guidance. I am thankful to all members of the Cheminformatics and Metabolism Group. -

Biological Chemistry I, Lexicon
Chemistry 5.07, Fall 2013 Lexicon of biochemical reactions Topic Description Page # # 1 Review of electrophiles and nucleophiles found in Biochemistry 2 2 C-C bond formation via carbonyl chem (aldol and Claisen reactions): Aldehydes, ketones, and Coenzyme 3-5 A and thioesters 3 Prenyl transfer reactions (will not be covered in 5.07) 6 4 Redox cofactors – Flavins (FAD, FMN, riboflavin) and NAD+ (NADP+) 7-12 5 ATP and phosphoryl transfer reactions 13-15 6 Thiamine pyrophosphate 16-18 7 Lipoic acid 19 8 S-Adenosylmethionine (SAM, Adomet), methylations 20 9 Pyridoxal phosphate 21-24 10 Biotin 25 11 Hemes 26-27 12 Metal cofactors: FeS clusters, Cu clusters 28 13 Coenzyme Q 29 14 ET theory 30-32 15 Folates 33-34 16 Methylcobalamin and adenosylcobalamin 35-37 1. Review of electrophiles and nucleophiles in biology Electrophiles Nucleophiles Nucleophilic form H+ Protons + Hydroxyl group Mn+ Metal ions + Sulfhydryl group + Carbonyl carbon Amino group Imine (protonated imine) carbon + Imidazole group (Schiff base) 2 2. Carbonyl Chemistry: Mechanisms of C-C Bond Formation • There are three general ways to form C-C bonds: 1. Aldol reactions where an aldehyde is condensed in a reversible reaction with another aldehyde or a ketone; 2. Claisen reaction in which a Coenzyme A thioester is condensed with an aldehyde or a ketone; 3. Prenyl transfer reactions where two 5 carbon units are condensed to form isoprenoids. • The aldol and claisen reactions involve carbonyl chemistry. These reactions are prevalent in glycolysis, fatty acid biosynthesis and degradation and the pentose phosphate pathway. 1. Aldol reaction generalizations: a. -

Heme Deficiency May Be a Factor in the Mitochondrial and Neuronal Decay of Aging
Heme deficiency may be a factor in the mitochondrial and neuronal decay of aging Hani Atamna*, David W. Killilea, Alison Nisbet Killilea, and Bruce N. Ames* Children’s Hospital Oakland Research Institute, Oakland, CA 94609 Contributed by Bruce N. Ames, September 26, 2002 Heme, a major functional form of iron in the cell, is synthesized in other environmental toxins (15, 16). Ferrochelatase is inacti- the mitochondria by ferrochelatase inserting ferrous iron into vated when its iron-sulfur cluster disassembles because intracel- protoporphyrin IX. Heme deficiency was induced with N-methyl- lular iron levels are low (17) or nitric oxide (NO) is present (18). protoporphyrin IX, a selective inhibitor of ferrochelatase, in two Iron deficiency is by far the most studied; it impairs cognitive human brain cell lines, SHSY5Y (neuroblastoma) and U373 (astro- function in children, impacts the morphology and the physiology cytoma), as well as in rat primary hippocampal neurons. Heme of brain even before changes in hemoglobin appear (19), dam- deficiency in brain cells decreases mitochondrial complex IV, acti- ages mitochondria (20), and causes oxidative stress (20). Iron vates nitric oxide synthase, alters amyloid precursor protein, and deficiency in children is associated with difficulties in performing corrupts iron and zinc homeostasis. The metabolic consequences cognitive tasks and retardation in the development of the CNS resulting from heme deficiency seem similar to dysfunctional (21, 22). Prenatal iron deficiency causes loss of cytochrome c neurons in patients with Alzheimer’s disease. Heme-deficient oxidase (complex IV) in selected regions in the brain of rats (23) SHSY5Y or U373 cells die when induced to differentiate or to and causes a decrease in ferrochelatase (17). -

Structural Basis of Inter-Domain Electron Transfer in Ncb5or, a Redox Enzyme Implicated in Diabetes and Lipid Metabolism
STRUCTURAL BASIS OF INTER-DOMAIN ELECTRON TRANSFER IN NCB5OR, A REDOX ENZYME IMPLICATED IN DIABETES AND LIPID METABOLISM By Bin Deng Submitted to the graduate degree program in Rehabilitation Science and the Graduate Faculty of the University of Kansas in partial fulfillment of the requirements for the degree of Doctor of Philosophy Hao Zhu, Ph.D. (co-chair, advisor) Irina Smirnova, Ph.D. (co-chair) David Benson, Ph.D. (co-advisor) WenFang Wang, Ph.D. Aron Fenton, Ph.D. Date defended: August 17, 2011 The Dissertation Committee for Bin Deng certifies that this is the approved version of the following dissertation STRUCTURAL BASIS OF INTER-DOMAIN ELECTRON TRANSFER IN NCB5OR, A REDOX ENZYME IMPLICATED IN DIABETES AND LIPID METABOLISM Hao Zhu, Ph.D. (co-chair, advisor) Irina Smirnova, Ph.D. (co-chair) Date approved: August 22, 2011 ii ABSTRACT NADH cytochrome b5 oxidoreductase (Ncb5or) is a multi-domain redox enzyme found in all animal tissues and associated with the endoplasmic reticulum (ER). Ncb5or contains (from N-terminus to C terminus) a novel N-terminal region, the b5 domain (Ncb5or-b5), the CS domain, and the b5R domain (Ncb5or-b5R). Ncb5or-b5, the heme binding domain, is homologous to microsomal cytochrome b5 (Cyb5A) and belongs to cytochrome b5 superfamily. Ncb5or-b5R, the FAD (flavin adenine dinucleotide) binding domain, is homologous to cytochrome b5 reductase (Cyb5R3) and belongs to ferredoxin NADP+ reductase superfamily. Both superfamilies are of great biological significance whose members have important functions. The CS domain can be assigned into the heat shock protein 20 (HSP20, or p23) family, whose members are known to mediate protein-protein interactions.