Human HIST1H2BO ORF Mammalian Expression Plasmid, C-Myc Tag
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genome-Wide Screen of Cell-Cycle Regulators in Normal and Tumor Cells
bioRxiv preprint doi: https://doi.org/10.1101/060350; this version posted June 23, 2016. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Genome-wide screen of cell-cycle regulators in normal and tumor cells identifies a differential response to nucleosome depletion Maria Sokolova1, Mikko Turunen1, Oliver Mortusewicz3, Teemu Kivioja1, Patrick Herr3, Anna Vähärautio1, Mikael Björklund1, Minna Taipale2, Thomas Helleday3 and Jussi Taipale1,2,* 1Genome-Scale Biology Program, P.O. Box 63, FI-00014 University of Helsinki, Finland. 2Science for Life laboratory, Department of Biosciences and Nutrition, Karolinska Institutet, SE- 141 83 Stockholm, Sweden. 3Science for Life laboratory, Division of Translational Medicine and Chemical Biology, Department of Medical Biochemistry and Biophysics, Karolinska Institutet, S-171 21 Stockholm, Sweden To identify cell cycle regulators that enable cancer cells to replicate DNA and divide in an unrestricted manner, we performed a parallel genome-wide RNAi screen in normal and cancer cell lines. In addition to many shared regulators, we found that tumor and normal cells are differentially sensitive to loss of the histone genes transcriptional regulator CASP8AP2. In cancer cells, loss of CASP8AP2 leads to a failure to synthesize sufficient amount of histones in the S-phase of the cell cycle, resulting in slowing of individual replication forks. Despite this, DNA replication fails to arrest, and tumor cells progress in an elongated S-phase that lasts several days, finally resulting in death of most of the affected cells. -

Product Datasheet HIST1H2BO Overexpression
Product Datasheet HIST1H2BO Overexpression Lysate NBP2-07991 Unit Size: 0.1 mg Store at -80C. Avoid freeze-thaw cycles. Protocols, Publications, Related Products, Reviews, Research Tools and Images at: www.novusbio.com/NBP2-07991 Updated 3/17/2020 v.20.1 Earn rewards for product reviews and publications. Submit a publication at www.novusbio.com/publications Submit a review at www.novusbio.com/reviews/destination/NBP2-07991 Page 1 of 2 v.20.1 Updated 3/17/2020 NBP2-07991 HIST1H2BO Overexpression Lysate Product Information Unit Size 0.1 mg Concentration The exact concentration of the protein of interest cannot be determined for overexpression lysates. Please contact technical support for more information. Storage Store at -80C. Avoid freeze-thaw cycles. Buffer RIPA buffer Target Molecular Weight 13.7 kDa Product Description Description Transient overexpression lysate of histone cluster 1, H2bo (HIST1H2BO) The lysate was created in HEK293T cells, using Plasmid ID RC219561 and based on accession number NM_003527. The protein contains a C-MYC/DDK Tag. Gene ID 8348 Gene Symbol HIST1H2BO Species Human Notes HEK293T cells in 10-cm dishes were transiently transfected with a non-lipid polymer transfection reagent specially designed and manufactured for large volume DNA transfection. Transfected cells were cultured for 48hrs before collection. The cells were lysed in modified RIPA buffer (25mM Tris-HCl pH7.6, 150mM NaCl, 1% NP-40, 1mM EDTA, 1xProteinase inhibitor cocktail mix, 1mM PMSF and 1mM Na3VO4, and then centrifuged to clarify the lysate. Protein concentration was measured by BCA protein assay kit.This product is manufactured by and sold under license from OriGene Technologies and its use is limited solely for research purposes. -

Functional Annotation of Human Long Noncoding Rnas Using Chromatin
bioRxiv preprint doi: https://doi.org/10.1101/2021.01.13.426305; this version posted January 14, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 Funconal annotaon of human long noncoding RNAs using chroman conformaon data Saumya Agrawal1, Tanvir Alam2, Masaru Koido1,3, Ivan V. Kulakovskiy4,5, Jessica Severin1, Imad ABugessaisa1, Andrey Buyan5,6, Josee Dos&e7, Masayoshi Itoh1,8, Naoto Kondo9, Yunjing Li10, Mickaël Mendez11, Jordan A. Ramilowski1,12, Ken Yagi1, Kayoko Yasuzawa1, CHi Wai Yip1, Yasushi Okazaki1, MicHael M. Ho9man11,13,14,15, Lisa Strug10, CHung CHau Hon1, CHikashi Terao1, Takeya Kasukawa1, Vsevolod J. Makeev4,16, Jay W. SHin1, Piero Carninci1, MicHiel JL de Hoon1 1RIKEN Center for Integra&ve Medical Sciences, YokoHama, Japan. 2College of Science and Engineering, Hamad Bin KHalifa University, DoHa, Qatar. 3Ins&tute of Medical Science, THe University of Tokyo, Tokyo, Japan. 4Vavilov Ins&tute of General Gene&cs, Russian Academy of Sciences, Moscow, Russia. 5Ins&tute of Protein ResearcH, Russian Academy of Sciences, PusHcHino, Russia. 6Faculty of Bioengineering and Bioinforma&cs, Lomonosov Moscow State University, Moscow, Russia. 7Department of BiocHemistry, Rosalind and Morris Goodman Cancer ResearcH Center, McGill University, Montréal, QuéBec, Canada. 8RIKEN Preven&ve Medicine and Diagnosis Innova&on Program, Wako, Japan. 9RIKEN Center for Life Science TecHnologies, YokoHama, Japan. 10Division of Biosta&s&cs, Dalla Lana ScHool of PuBlic HealtH, University of Toronto, Toronto, Ontario, Canada. 11Department of Computer Science, University of Toronto, Toronto, Ontario, Canada. 12Advanced Medical ResearcH Center, YokoHama City University, YokoHama, Japan. -

Rap1-Mediated Chromatin and Gene Expression Changes at Senescence
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2019 Rap1-Mediated Chromatin And Gene Expression Changes At Senescence Shufei Song University of Pennsylvania Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Biochemistry Commons, and the Cell Biology Commons Recommended Citation Song, Shufei, "Rap1-Mediated Chromatin And Gene Expression Changes At Senescence" (2019). Publicly Accessible Penn Dissertations. 3557. https://repository.upenn.edu/edissertations/3557 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/3557 For more information, please contact [email protected]. Rap1-Mediated Chromatin And Gene Expression Changes At Senescence Abstract ABSTRACT RAP1-MEDIATED CHROMATIN AND GENE EXPRESSION CHANGES AT SENESCENCE The telomeric protein Rap1 has been extensively studied for its roles as a transcriptional activator and repressor. Indeed, in both yeast and mammals, Rap1 is known to bind throughout the genome to reorganize chromatin and regulate gene transcription. Previously, our lab published evidence that Rap1 plays important roles in cellular senescence. In telomerase-deficient S. cerevisiae, Rap1 relocalizes from telomeres and subtelomeres to new Rap1 target at senescence (NRTS). This leads to two types of histone loss: Rap1 lowers global histone levels by repressing histone gene transcription and it also results in local nucleosome displacement at the promoters of the activated NRTS. Here, I examine mechanisms of site-specific histone loss by presenting evidence that Rap1 can directly interact with histone tetramers H3/H4, and map this interaction to a three-amino-acid-patch within the DNA binding domain. Functional studies are performed in vivo using a mutant form of Rap1 with weakened histone interactions, and deficient promoter clearance as well as blunted gene activation is observed, indicating that direct Rap1-H3/H4 interactions are involved in nucleosome displacement. -

Human Tagged ORF Clone Lentiviral Particle – RC219561L2V
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for RC219561L2V HIST1H2BO (H2BC17) (NM_003527) Human Tagged ORF Clone Lentiviral Particle Product data: Product Type: Lentiviral Particles Product Name: HIST1H2BO (H2BC17) (NM_003527) Human Tagged ORF Clone Lentiviral Particle Symbol: H2BC17 Synonyms: dJ193B12.2; H2B.2; H2B/n; H2BFN; HIST1H2BO Vector: pLenti-C-mGFP (PS100071) ACCN: NM_003527 ORF Size: 378 bp ORF Nucleotide The ORF insert of this clone is exactly the same as(RC219561). Sequence: OTI Disclaimer: The molecular sequence of this clone aligns with the gene accession number as a point of reference only. However, individual transcript sequences of the same gene can differ through naturally occurring variations (e.g. polymorphisms), each with its own valid existence. This clone is substantially in agreement with the reference, but a complete review of all prevailing variants is recommended prior to use. More info OTI Annotation: This clone was engineered to express the complete ORF with an expression tag. Expression varies depending on the nature of the gene. RefSeq: NM_003527.4, NP_003518.2 RefSeq Size: 467 bp RefSeq ORF: 381 bp Locus ID: 8348 UniProt ID: P23527 Protein Families: Druggable Genome Protein Pathways: Systemic lupus erythematosus MW: 13.9 kDa This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 2 HIST1H2BO (H2BC17) (NM_003527) Human Tagged ORF Clone Lentiviral Particle – RC219561L2V Gene Summary: Histones are basic nuclear proteins that are responsible for the nucleosome structure of the chromosomal fiber in eukaryotes. -

Quantitative Mass Spectrometry Reveals Changes in Histone H2B
University of Kentucky UKnowledge Molecular and Cellular Biochemistry Faculty Molecular and Cellular Biochemistry Publications 5-11-2016 Quantitative Mass Spectrometry Reveals Changes in Histone H2B Variants as Cells Undergo Inorganic Arsenic-Mediated Cellular Transformation Matthew Rea University of Kentucky, [email protected] Tingting Jiang Florida State University Rebekah Eleazer University of Kentucky, [email protected] Meredith Eckstein University of Kentucky, [email protected] Alan G. Marshall Florida State University FSeoe nelloxtw pa thige fors aaddndition addal aitutionhorsal works at: https://uknowledge.uky.edu/biochem_facpub Part of the Biochemistry, Biophysics, and Structural Biology Commons, Cellular and Molecular Right click to open a feedback form in a new tab to let us know how this document benefits oy u. Physiology Commons, and the Inorganic Chemicals Commons Repository Citation Rea, Matthew; Jiang, Tingting; Eleazer, Rebekah; Eckstein, Meredith; Marshall, Alan G.; and Fondufe-Mittendorf, Yvonne N., "Quantitative Mass Spectrometry Reveals Changes in Histone H2B Variants as Cells Undergo Inorganic Arsenic-Mediated Cellular Transformation" (2016). Molecular and Cellular Biochemistry Faculty Publications. 110. https://uknowledge.uky.edu/biochem_facpub/110 This Article is brought to you for free and open access by the Molecular and Cellular Biochemistry at UKnowledge. It has been accepted for inclusion in Molecular and Cellular Biochemistry Faculty Publications by an authorized administrator of UKnowledge. For more information, please contact [email protected]. Authors Matthew Rea, Tingting Jiang, Rebekah Eleazer, Meredith Eckstein, Alan G. Marshall, and Yvonne N. Fondufe- Mittendorf Quantitative Mass Spectrometry Reveals Changes in Histone H2B Variants as Cells Undergo Inorganic Arsenic- Mediated Cellular Transformation Notes/Citation Information Published in Molecular & Cellular Proteomics, v. -
![Viewer (IGV), UCSC As Described [23, 28]](https://docslib.b-cdn.net/cover/7331/viewer-igv-ucsc-as-described-23-28-1987331.webp)
Viewer (IGV), UCSC As Described [23, 28]
Jahan et al. Epigenetics & Chromatin (2016) 9:19 DOI 10.1186/s13072-016-0068-2 Epigenetics & Chromatin RESEARCH Open Access The chicken erythrocyte epigenome Sanzida Jahan, Wayne Xu, Shihua He, Carolina Gonzalez, Geneviève P. Delcuve and James R. Davie* Abstract Background: Transcriptional regulation is impacted by multiple layers of genome organization. A general feature of transcriptionally active chromatin is sensitivity to DNase I and association with acetylated histones. However, very few of these active DNase I-sensitive domains, such as the chicken erythrocyte β-globin domain, have been identified and characterized. In chicken polychromatic erythrocytes, dynamically acetylated histones associated with DNase I-sensi- tive, transcriptionally active chromatin prevent histone H1/H5-induced insolubility at physiological ionic strength. Results: Here, we identified and mapped out all the transcriptionally active chromosomal domains in the chicken polychromatic erythrocyte genome by combining a powerful chromatin fractionation method with next-generation DNA and RNA sequencing. Two classes of transcribed chromatin organizations were identified on the basis of the extent of solubility at physiological ionic strength. Highly transcribed genes were present in multigenic salt-soluble chromatin domains ranging in length from 30 to over 150 kb. We identified over 100 highly expressed genes that were organized in broad dynamically highly acetylated, salt-soluble chromatin domains. Highly expressed genes were associated with H3K4me3 and H3K27ac and produced discernible antisense transcripts. The moderately- and low- expressing genes had highly acetylated, salt-soluble chromatin regions confined to the ′5 end of the gene. Conclusions: Our data provide a genome-wide profile of chromatin signatures in relation to expression levels in chicken polychromatic erythrocytes. -

The Human Canonical Core Histone Catalogue David Miguel Susano Pinto*, Andrew Flaus*,†
bioRxiv preprint doi: https://doi.org/10.1101/720235; this version posted July 30, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. The Human Canonical Core Histone Catalogue David Miguel Susano Pinto*, Andrew Flaus*,† Abstract Core histone proteins H2A, H2B, H3, and H4 are encoded by a large family of genes dis- tributed across the human genome. Canonical core histones contribute the majority of proteins to bulk chromatin packaging, and are encoded in 4 clusters by 65 coding genes comprising 17 for H2A, 18 for H2B, 15 for H3, and 15 for H4, along with at least 17 total pseudogenes. The canonical core histone genes display coding variation that gives rise to 11 H2A, 15 H2B, 4 H3, and 2 H4 unique protein isoforms. Although histone proteins are highly conserved overall, these isoforms represent a surprising and seldom recognised variation with amino acid identity as low as 77 % between canonical histone proteins of the same type. The gene sequence and protein isoform diversity also exceeds com- monly used subtype designations such as H2A.1 and H3.1, and exists in parallel with the well-known specialisation of variant histone proteins. RNA sequencing of histone transcripts shows evidence for differential expression of histone genes but the functional significance of this variation has not yet been investigated. To assist understanding of the implications of histone gene and protein diversity we have catalogued the entire human canonical core histone gene and protein complement. -

Supplemental Material For: Screen Identifies Bromodomain Protein
Supplemental Material for: Screen identifies bromodomain protein ZMYND8 in chromatin recognition of transcription-associated DNA damage that promotes homologous recombination Fade Gong1,2,6, Li-Ya Chiu1,2,6, Ben Cox1,2, François Aymard3,4, Thomas Clouaire3,4, Justin W. Leung1,2, Michael Cammarata5, Mercedes Perez1,2, Poonam Agarwal1,2, Jennifer S. Brodbelt5, Gaëlle Legube3,4 & Kyle. M. Miller1,2,* 1. Supplemental Materials and Methods 2. Supplemental References 3. Supplemental Figure S1-S10 with legends 4. Supplemental Table S1-S3 with legends 1 Supplemental Materials and Methods Plasmid and siRNA transfections Mammalian expression vectors were transfected into U2OS cells by Hilymax (Dojindo) or Fugene HD (Promega) according to manufacturer’s instructions. The I-SceI expressing vector (pCAG-I-SceI) or control vector (pCAG) were transfected into the U2OS DR-GFP cells by Fugene HD (Promega). For HEK293T cells, transient transfections were carried out with pEI (Polyethylenimine, Sigma). Analyses for transient plasmid transfection were performed 24-48 h after transfection. Transfections for siRNA were carried out with lipofectamine RNAiMax (Invitrogen) following the manufacturer’s instructions. Analyses from siRNA treated cells were performed 48-72 h after transfection. The siRNAs used in this study were: siControl: non-targeting pool (Dharmacon); siZMYND8 #1: SMARTpool (Dharmacon); siZMYND8 #2: GGACUUUCCCCUUUUUAUA (targeting the 3’-UTR region of ZMYND8) (Sigma); siZMYND8 #3: GAACAUAGAUGAAUGAAA (Sigma); siCHD4: CCCAGAAGAGGAUUUGUCA (Sigma), siLSD1: GCCUAGACAUUAAACUGAAUA (Sigma); siTIP60 SMARTpool (Dharmacon); siMOF: GCAAAGACCAUAAGAUUUA (Sigma); siCtIP: GGUAAAACAGGAACGAAUC (Sigma); siLigaseIV: AGGAAGUAUUCUCAGGAAUUA (Sigma). Cloning and plasmids 2 cDNAs of human BRD-containing proteins were cloned into the Gateway entry vector pENTR11 by restriction sites, or pDONR201 by attB recombinant sites. -

Novel DNA Damage Checkpoints Mediating Cell Death Induced by the NEDD8-Activating Enzyme Inhibitor MLN4924
Published OnlineFirst October 24, 2012; DOI: 10.1158/0008-5472.CAN-12-1729 Cancer Molecular and Cellular Pathobiology Research Novel DNA Damage Checkpoints Mediating Cell Death Induced by the NEDD8-Activating Enzyme Inhibitor MLN4924 Jonathan L. Blank1, Xiaozhen J. Liu1, Katherine Cosmopoulos1, David C. Bouck1, Khristofer Garcia1, Hugues Bernard1, Olga Tayber1, Greg Hather2, Ray Liu2, Usha Narayanan1, Michael A. Milhollen1, and Eric S. Lightcap1 Abstract MLN4924 is an investigational small-molecule inhibitor of the NEDD8-activating enzyme (NAE) in phase I clinical trials. NAE inhibition prevents the ubiquitination and proteasomal degradation of substrates for cullin-RING ubiquitin E3 ligases that support cancer pathophysiology, but the genetic determinants conferring sensitivity to NAE inhibition are unknown. To address this gap in knowledge, we conducted a genome-wide siRNA screen to identify genes and pathways that affect the lethality of MLN4924 in melanoma cells. Of the 154 genes identified, approximately one-half interfered with components of the cell cycle, apoptotic machinery, ubiquitin system, and DNA damage response pathways. In particular, genes involved in DNA replication, p53, BRCA1/BRCA2, transcrip- tion-coupled repair, andbaseexcisionrepair seemedtobeimportantforMLN4924lethality. Incontrast, geneswithin the G2–M checkpoint affected sensitivity to MLN4924 in colon cancer cells. Cell-cycle analysis in melanoma cells by flow cytometry following RNAi-mediated silencing showed that MLN4924 prevented the transition of cells from S– G2 phase after induction of rereplication stress. Our analysis suggested an important role for the p21-dependent intra-S-phase checkpoint and extensive rereplication, whereas the ATR-dependent intra-S-phase checkpoint seemed to play a less dominant role. Unexpectedly, induction of the p21-dependent intra-S-phase checkpoint seemed to be independent of both Cdt1 stabilization and ATR signaling. -

( 12 ) Patent Application Publication ( 10 ) Pub . No .: US 2021/0005284 A1 Nuzhdina Et Al
US 20210005284A1 IN ( 19 ) United States ( 12 ) Patent Application Publication ( 10 ) Pub . No .: US 2021/0005284 A1 Nuzhdina et al . ( 43 ) Pub . Date : Jan. 7 , 2021 ( 54 ) TECHNIQUES FOR NUCLEIC ACID DATA G16B 25/10 ( 2006.01 ) QUALITY CONTROL G16B 35/10 ( 2006.01 ) ( 52 ) U.S. CI . ( 71 ) Applicant: BostonGene Corporation , Waltham , CPC G16B 30/10 ( 2019.02 ) ; G16B 35/10 MA ( US ) ( 2019.02 ) ; G16B 25/10 ( 2019.02 ) ; G16B ( 72 ) Inventors : Ekaterina Nuzhdina , Moscow ( RU ); 20/20 ( 2019.02 ) Alexander Bagaev , Moscow ( RU ); Maksim Chelushkin , Moscow (RU ) ; Yaroslav Lozinsky, Moscow ( RU ); ( 57 ) ABSTRACT Natalia Miheecheva , Moscow ( RU ) ; Alexander Zaitsev , Leninskiy R - N (RU ) Described herein are various methods of collecting and processing of tumor and /or healthy tissue samples to extract ( 21 ) Appl . No .: 16 / 920,641 nucleic acid and perform nucleic acid sequencing . Also described herein are various methods of processing nucleic ( 22 ) Filed : Jul . 3 , 2020 acid sequencing data to remove bias from the nucleic acid sequencing data . Also described herein are various methods Related U.S. Application Data of evaluating the quality of nucleic acid sequence informa ( 60 ) Provisional application No. 62 / 991,570 , filed on Mar. tion . The identity and / or integrity of nucleic acid sequence 18 , 2020 , provisional application No. 62 /870,622 , data is evaluated prior to using the sequence information for filed on Jul. 3 , 2019 . subsequent analysis ( for example for diagnostic , prognostic , or clinical purposes ). The methods enable a subject, doctor, Publication Classification or user to characterize or classify various types of cancer ( 51 ) Int . Cl . precisely , and thereby determine a therapy or combination of G16B 30/10 ( 2006.01 ) therapies that may be effective to treat a cancer in a subject G16B 20/20 ( 2006.01 ) based on the precise characterization . -
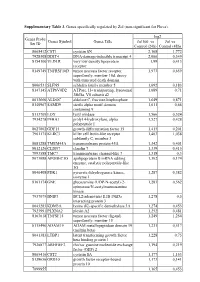
Supplementary Table 3. Genes Specifically Regulated by Zol (Non-Significant for Fluva)
Supplementary Table 3. Genes specifically regulated by Zol (non-significant for Fluva). log2 Genes Probe Genes Symbol Genes Title Zol100 vs Zol vs Set ID Control (24h) Control (48h) 8065412 CST1 cystatin SN 2,168 1,772 7928308 DDIT4 DNA-damage-inducible transcript 4 2,066 0,349 8154100 VLDLR very low density lipoprotein 1,99 0,413 receptor 8149749 TNFRSF10D tumor necrosis factor receptor 1,973 0,659 superfamily, member 10d, decoy with truncated death domain 8006531 SLFN5 schlafen family member 5 1,692 0,183 8147145 ATP6V0D2 ATPase, H+ transporting, lysosomal 1,689 0,71 38kDa, V0 subunit d2 8013660 ALDOC aldolase C, fructose-bisphosphate 1,649 0,871 8140967 SAMD9 sterile alpha motif domain 1,611 0,66 containing 9 8113709 LOX lysyl oxidase 1,566 0,524 7934278 P4HA1 prolyl 4-hydroxylase, alpha 1,527 0,428 polypeptide I 8027002 GDF15 growth differentiation factor 15 1,415 0,201 7961175 KLRC3 killer cell lectin-like receptor 1,403 1,038 subfamily C, member 3 8081288 TMEM45A transmembrane protein 45A 1,342 0,401 8012126 CLDN7 claudin 7 1,339 0,415 7993588 TMC7 transmembrane channel-like 7 1,318 0,3 8073088 APOBEC3G apolipoprotein B mRNA editing 1,302 0,174 enzyme, catalytic polypeptide-like 3G 8046408 PDK1 pyruvate dehydrogenase kinase, 1,287 0,382 isozyme 1 8161174 GNE glucosamine (UDP-N-acetyl)-2- 1,283 0,562 epimerase/N-acetylmannosamine kinase 7937079 BNIP3 BCL2/adenovirus E1B 19kDa 1,278 0,5 interacting protein 3 8043283 KDM3A lysine (K)-specific demethylase 3A 1,274 0,453 7923991 PLXNA2 plexin A2 1,252 0,481 8163618 TNFSF15 tumor necrosis