Association of Cardiovascular Risk with Inhaled Long-Acting Bronchodilators in Patients with Chronic Obstructive Pulmonary Disease: a Nested Case-Control Study
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

United States Patent Office
3,092,548 United States Patent Office Patented June 4, 1963 2 are the various side effects that occur in an appreciable 3,092,548 number of patients, the foremost of which are a blurring METHOD OF TREATING PEPTICULCERS WITH PANTOTHENECACD of vision, drowsiness and a general dry condition mani Albert G. Worton, Columbus, Ohio, assignor to The fested by a retarded salivation, reduction of perspiration Warren-Teed Products Company, Columbus, Ohio, a 5 and diminished urinary output. Other side effects which corporation of Ohio occur in some cases are glaucoma, stimulation of the cen No Drawing. Filed Oct. 27, 1960, Ser. No. 65,256 trol nervous system and in severe cases cardiac and respi 5 Claims. (Ci. 67-55) ratory collapse. These side effects increase to Some de gree with the increase in dosage. Despite the side effects This invention relates to preparation adapted for use O these compounds are fairly selective and highly effective in treating disorders of the gastro-intestinal tract, more in decreasing the volume and acidity of gastric secretion particularly in treating peptic ulcer, both of the duodenal and in reducing gastrointestinal motility. and gastric type, for hyperacidity, hypertropic gastritis, Understandably, the main problem in the past has been splenic flexure syndrome, biliary dyskinesia (postchole to provide a dosage of anticholinergic drug which will cystectomy syndrome) and hiatal hernia or other condi achieve the most beneficial results possible and yet mini tions where anticholinergic effect, either spasmolytic or mize the undesired side effects. One of the objects of antisecretory is indicated or where antiuicerogenic effect this invention is to provide novel compositions containing is indicated. -
List of New Drugs Approved in India from 1991 to 2000
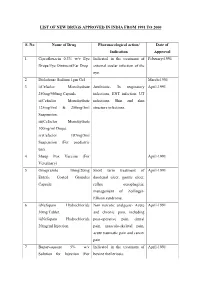
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

WITHOUTUS010307409B2 (12 ) United States Patent ( 10 ) Patent No
WITHOUTUS010307409B2 (12 ) United States Patent ( 10 ) Patent No. : US 10 , 307 ,409 B2 Chase et al. (45 ) Date of Patent: Jun . 4 , 2019 ( 54 ) MUSCARINIC COMBINATIONS AND THEIR (52 ) U . S . CI. USE FOR COMBATING CPC . .. .. A61K 31/ 4439 (2013 . 01 ) ; A61K 9 /0056 HYPOCHOLINERGIC DISORDERS OF THE (2013 . 01 ) ; A61K 9 / 7023 ( 2013 . 01 ) ; A61K CENTRAL NERVOUS SYSTEM 31 / 166 ( 2013 . 01 ) ; A61K 31 / 216 ( 2013 . 01 ) ; A61K 31 /4178 ( 2013 .01 ) ; A61K 31/ 439 (71 ) Applicant: Chase Pharmaceuticals Corporation , ( 2013 .01 ) ; A61K 31 /44 (2013 . 01 ) ; A61K Washington , DC (US ) 31/ 454 (2013 .01 ) ; A61K 31/ 4725 ( 2013 .01 ) ; A61K 31 /517 (2013 .01 ) ; A61K 45 / 06 ( 72 ) Inventors : Thomas N . Chase , Washington , DC (2013 . 01 ) (US ) ; Kathleen E . Clarence -Smith , ( 58 ) Field of Classification Search Washington , DC (US ) CPC .. A61K 31/ 167 ; A61K 31/ 216 ; A61K 31/ 439 ; A61K 31 /454 ; A61K 31 /4439 ; A61K (73 ) Assignee : Chase Pharmaceuticals Corporation , 31 /4175 ; A61K 31 /4725 Washington , DC (US ) See application file for complete search history. ( * ) Notice : Subject to any disclaimer, the term of this (56 ) References Cited patent is extended or adjusted under 35 U . S . C . 154 (b ) by 0 days . U . S . PATENT DOCUMENTS 5 ,534 ,520 A 7 / 1996 Fisher et al. ( 21) Appl . No. : 15 /260 , 996 2008 /0306103 Al 12 /2008 Fisher et al. 2011/ 0021503 A1* 1/ 2011 Chase . .. A61K 31/ 27 ( 22 ) Filed : Sep . 9 , 2016 514 / 215 2011/ 0071135 A1 * 3 / 2011 Chase . .. .. .. A61K 31/ 166 (65 ) Prior Publication Data 514 / 215 2011 /0245294 Al 10 / 2011 Paborji et al. -

Clinical and Laboratory Responses to Niceritrol in the Treatment of Hypercholesterolaemia
Postgrad Med J: first published as 10.1136/pgmj.64.755.672 on 1 September 1988. Downloaded from Postgraduate Medical Journal (1988) 64, 672-675 Clinical and laboratory responses to niceritrol in the treatment of hypercholesterolaemia L.D. Curtis,' J.P.D. Reckless,2 A.F. Winder3 and D.J. Betteridge1 1Department of Medicine, University College and Middlesex School of Medicine, University College London, 2Royal United Hospital, Bath and 3Leicester Royal Infirmary, Leicester, UK. Summary: Twenty-five hypercholesterolaemic patients from three centres in the UK were investigated in an open study of the efficacy and side effects of niceritrol. Five patients dropped out of the study at an early stage and had insufficient data for analysis. There were 13 males and 7 females (mean age 49.2 years, range 18-69). Fourteen patients had heterozygous familial hyperchol- esterolaemia, and six polygenic hypercholesterolaemia. Niceritrol was started at a dose of 750mg/ day and this was increased at weekly intervals over 4 weeks to the maximum tolerated dosage up to 3 g/day. This was then maintained for a further 8 weeks. There were statistically significant decreases in total plasma cholesterol, total triglyceride, LDL cholesterol and VLDL triglyceride; HDL cholesterol remained unchanged after 12 weeks of treatment (Wilcoxon matched pairs, signed ranks test). The 14 patients with familial hypercholesterolaemia showed a 13.9% fall in total cholesterol and a 19.8% fall in LDL cholesterol. All patients reported flushing and some had Protected by copyright. gastrointestinal symptoms but 19 would have been prepared to continue with the therapy at doses up to 3g/day. -

Muscarinic Acetylcholine Receptor
mAChR Muscarinic acetylcholine receptor mAChRs (muscarinic acetylcholine receptors) are acetylcholine receptors that form G protein-receptor complexes in the cell membranes of certainneurons and other cells. They play several roles, including acting as the main end-receptor stimulated by acetylcholine released from postganglionic fibersin the parasympathetic nervous system. mAChRs are named as such because they are more sensitive to muscarine than to nicotine. Their counterparts are nicotinic acetylcholine receptors (nAChRs), receptor ion channels that are also important in the autonomic nervous system. Many drugs and other substances (for example pilocarpineand scopolamine) manipulate these two distinct receptors by acting as selective agonists or antagonists. Acetylcholine (ACh) is a neurotransmitter found extensively in the brain and the autonomic ganglia. www.MedChemExpress.com 1 mAChR Inhibitors & Modulators (+)-Cevimeline hydrochloride hemihydrate (-)-Cevimeline hydrochloride hemihydrate Cat. No.: HY-76772A Cat. No.: HY-76772B Bioactivity: Cevimeline hydrochloride hemihydrate, a novel muscarinic Bioactivity: Cevimeline hydrochloride hemihydrate, a novel muscarinic receptor agonist, is a candidate therapeutic drug for receptor agonist, is a candidate therapeutic drug for xerostomia in Sjogren's syndrome. IC50 value: Target: mAChR xerostomia in Sjogren's syndrome. IC50 value: Target: mAChR The general pharmacol. properties of this drug on the The general pharmacol. properties of this drug on the gastrointestinal, urinary, and reproductive systems and other… gastrointestinal, urinary, and reproductive systems and other… Purity: >98% Purity: >98% Clinical Data: No Development Reported Clinical Data: No Development Reported Size: 10mM x 1mL in DMSO, Size: 10mM x 1mL in DMSO, 1 mg, 5 mg 1 mg, 5 mg AC260584 Aclidinium Bromide Cat. No.: HY-100336 (LAS 34273; LAS-W 330) Cat. -

Interactions Medicamenteuses Index Des Classes Pharmaco
INTERACTIONS MEDICAMENTEUSES INDEX DES CLASSES PHARMACO-THERAPEUTIQUES Mise à jour avril 2006 acides biliaires (acide chenodesoxycholique, acide ursodesoxycholique) acidifiants urinaires adrénaline (voie bucco-dentaire ou sous-cutanée) (adrenaline alcalinisants urinaires (acetazolamide, sodium (bicarbonate de), trometamol) alcaloïdes de l'ergot de seigle dopaminergiques (bromocriptine, cabergoline, lisuride, pergolide) alcaloïdes de l'ergot de seigle vasoconstricteurs (dihydroergotamine, ergotamine, methylergometrine) alginates (acide alginique, sodium et de trolamine (alginate de)) alphabloquants à visée urologique (alfuzosine, doxazosine, prazosine, tamsulosine, terazosine) amidons et gélatines (gelatine, hydroxyethylamidon, polygeline) aminosides (amikacine, dibekacine, gentamicine, isepamicine, kanamycine, netilmicine, streptomycine, tobramycine) amprénavir (et, par extrapolation, fosamprénavir) (amprenavir, fosamprenavir) analgésiques morphiniques agonistes (alfentanil, codeine, dextromoramide, dextropropoxyphene, dihydrocodeine, fentanyl, hydromorphone, morphine, oxycodone, pethidine, phenoperidine, remifentanil, sufentanil, tramadol) analgésiques morphiniques de palier II (codeine, dextropropoxyphene, dihydrocodeine, tramadol) analgésiques morphiniques de palier III (alfentanil, dextromoramide, fentanyl, hydromorphone, morphine, oxycodone, pethidine, phenoperidine, remifentanil, sufentanil) analogues de la somatostatine (lanreotide, octreotide) androgènes (danazol, norethandrolone, testosterone) anesthésiques volatils halogénés -

Dorset Medicines Advisory Group
DORSET CARDIOLOGY WORKING GROUP GUIDELINE FOR CALCIUM CHANNEL BLOCKERS IN HYPERTENSION SUMMARY The pan-Dorset cardiology working group continues to recommend the use of amlodipine (a third generation dihydropyridine calcium-channel blocker) as first choice calcium channel blocker on the pan-Dorset formulary for hypertension. Lercanidipine is second choice, lacidipine third choice and felodipine is fourth choice. This is due to preferable side effect profiles in terms of ankle oedema and relative costs of the preparations. Note: where angina is the primary indication or is a co-morbidity prescribers must check against the specific product characteristics (SPC) for an individual drug to confirm this is a licensed indication. N.B. Lacidipine and lercandipine are only licensed for use in hypertension. Chapter 02.06.02 CCBs section of the Formulary has undergone an evidence-based review. A comprehensive literature search was carried out on NHS Evidence, Medline, EMBASE, Cochrane Database, and UK Duets. This was for recent reviews or meta-analyses on calcium channel blockers from 2009 onwards (comparative efficacy and side effects) and randomised controlled trials (RCTs). REVIEW BACKGROUND Very little good quality evidence exists. No reviews, meta-analyses or RCTs were found covering all calcium channel blockers currently on the formulary. Another limitation was difficulty obtaining full text original papers for some of the references therefore having to use those from more obscure journals instead. Some discrepancies exist between classification of generations of dihydropyridine CCBs, depending upon the year of publication of the reference/authors’ interpretation. Dihydropyridine (DHP) CCBs tend to be more potent vasodilators than non-dihydropyridine (non-DHP) CCBs (diltiazem, verapamil), but the latter have greater inotropic effects. -

Comparative Effects of Cefpirome (Hr 810) and Other Cephalosporins on Experimentally Induced Pneumonia in Mice
VOL. XXXIX NO. 7 THE JOURNAL OF ANTIBIOTICS 971 COMPARATIVE EFFECTS OF CEFPIROME (HR 810) AND OTHER CEPHALOSPORINS ON EXPERIMENTALLY INDUCED PNEUMONIA IN MICE N. KLESEL, D. ISERT, M. LIMBERT, G. SEIBERT, I. WINKLER and E. SCHRINNER Hoechst Aktiengesellschaft, Dept. of Chemotherapy, 6230 Frankfurt a. M. 80, FRG (Received for publication March 24, 1986) The chemotherapeutic efficacy of cefpirome (HR 810), a new polar aminothiazolyl- cephalosporin and that of ceftazidime, cefotaxime, cefoperazone, latamoxef and cefodizime were examined against experimental pneumonia caused by Klebsiella pneammlliae DT-S in mice. When compared in terms of MIC values against the infecting organism and the phar- macokinetic pattern, cefpirome showed equal activity and a similar pharmacokinetic behavior to ceftazidime and cefotaxime in mice. Trials to assess the bactericidial activity in vhro, however, showed that cefpirome displayed a more marked bactericidal effect in pneumonic mice than the other cephalosporins tested. Only cefodizime, a cephalosporin with extremely high and prolonged blood and tissue levels in experimental animals exerted chemotherapeutic effects similar to cefpirome. After cefpirome or cefodizime medication (50 mg/kg), the viable counts in the lungs of experimental animals fell steadily to 1/10,000 of the pretreatment level and, in contrast to the reference compounds, no regrowth of the challenge organisms could be observed with both drugs. Moreover, with ED.-,,,,sranging from 1.1 to 59.1 mg/kg in treatment studies, cefpirome as well as cefodizime were two to ten times more effective than ceftazidime and cefotaxime, whereas cefoperazone and latamoxef were considerably less effective. Cefpirome (3-((2,3-cyclopenteno-I-pyridium)methyl)-7-(2-syn-methoximino-2-(2-aminothiazol-4- yl)acetamido)ceph-3-em-4-carboxylate, HR 810) is a new semi-synthetic parenteral cephalosporin antibiotic with a broad spectrum of antibacterial activity in vitro and in rivo. -

List of Union Reference Dates A
Active substance name (INN) EU DLP BfArM / BAH DLP yearly PSUR 6-month-PSUR yearly PSUR bis DLP (List of Union PSUR Submission Reference Dates and Frequency (List of Union Frequency of Reference Dates and submission of Periodic Frequency of submission of Safety Update Reports, Periodic Safety Update 30 Nov. 2012) Reports, 30 Nov. -

Health Reports for Mutual Recognition of Medical Prescriptions: State of Play
The information and views set out in this report are those of the author(s) and do not necessarily reflect the official opinion of the European Union. Neither the European Union institutions and bodies nor any person acting on their behalf may be held responsible for the use which may be made of the information contained therein. Executive Agency for Health and Consumers Health Reports for Mutual Recognition of Medical Prescriptions: State of Play 24 January 2012 Final Report Health Reports for Mutual Recognition of Medical Prescriptions: State of Play Acknowledgements Matrix Insight Ltd would like to thank everyone who has contributed to this research. We are especially grateful to the following institutions for their support throughout the study: the Pharmaceutical Group of the European Union (PGEU) including their national member associations in Denmark, France, Germany, Greece, the Netherlands, Poland and the United Kingdom; the European Medical Association (EMANET); the Observatoire Social Européen (OSE); and The Netherlands Institute for Health Service Research (NIVEL). For questions about the report, please contact Dr Gabriele Birnberg ([email protected] ). Matrix Insight | 24 January 2012 2 Health Reports for Mutual Recognition of Medical Prescriptions: State of Play Executive Summary This study has been carried out in the context of Directive 2011/24/EU of the European Parliament and of the Council of 9 March 2011 on the application of patients’ rights in cross- border healthcare (CBHC). The CBHC Directive stipulates that the European Commission shall adopt measures to facilitate the recognition of prescriptions issued in another Member State (Article 11). At the time of submission of this report, the European Commission was preparing an impact assessment with regards to these measures, designed to help implement Article 11. -

NINDS Custom Collection II
ACACETIN ACEBUTOLOL HYDROCHLORIDE ACECLIDINE HYDROCHLORIDE ACEMETACIN ACETAMINOPHEN ACETAMINOSALOL ACETANILIDE ACETARSOL ACETAZOLAMIDE ACETOHYDROXAMIC ACID ACETRIAZOIC ACID ACETYL TYROSINE ETHYL ESTER ACETYLCARNITINE ACETYLCHOLINE ACETYLCYSTEINE ACETYLGLUCOSAMINE ACETYLGLUTAMIC ACID ACETYL-L-LEUCINE ACETYLPHENYLALANINE ACETYLSEROTONIN ACETYLTRYPTOPHAN ACEXAMIC ACID ACIVICIN ACLACINOMYCIN A1 ACONITINE ACRIFLAVINIUM HYDROCHLORIDE ACRISORCIN ACTINONIN ACYCLOVIR ADENOSINE PHOSPHATE ADENOSINE ADRENALINE BITARTRATE AESCULIN AJMALINE AKLAVINE HYDROCHLORIDE ALANYL-dl-LEUCINE ALANYL-dl-PHENYLALANINE ALAPROCLATE ALBENDAZOLE ALBUTEROL ALEXIDINE HYDROCHLORIDE ALLANTOIN ALLOPURINOL ALMOTRIPTAN ALOIN ALPRENOLOL ALTRETAMINE ALVERINE CITRATE AMANTADINE HYDROCHLORIDE AMBROXOL HYDROCHLORIDE AMCINONIDE AMIKACIN SULFATE AMILORIDE HYDROCHLORIDE 3-AMINOBENZAMIDE gamma-AMINOBUTYRIC ACID AMINOCAPROIC ACID N- (2-AMINOETHYL)-4-CHLOROBENZAMIDE (RO-16-6491) AMINOGLUTETHIMIDE AMINOHIPPURIC ACID AMINOHYDROXYBUTYRIC ACID AMINOLEVULINIC ACID HYDROCHLORIDE AMINOPHENAZONE 3-AMINOPROPANESULPHONIC ACID AMINOPYRIDINE 9-AMINO-1,2,3,4-TETRAHYDROACRIDINE HYDROCHLORIDE AMINOTHIAZOLE AMIODARONE HYDROCHLORIDE AMIPRILOSE AMITRIPTYLINE HYDROCHLORIDE AMLODIPINE BESYLATE AMODIAQUINE DIHYDROCHLORIDE AMOXEPINE AMOXICILLIN AMPICILLIN SODIUM AMPROLIUM AMRINONE AMYGDALIN ANABASAMINE HYDROCHLORIDE ANABASINE HYDROCHLORIDE ANCITABINE HYDROCHLORIDE ANDROSTERONE SODIUM SULFATE ANIRACETAM ANISINDIONE ANISODAMINE ANISOMYCIN ANTAZOLINE PHOSPHATE ANTHRALIN ANTIMYCIN A (A1 shown) ANTIPYRINE APHYLLIC