Emerging Strategies for the Bioremediation of the Phenylurea Herbicide Diuron
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

2,4-Dichlorophenoxyacetic Acid
2,4-Dichlorophenoxyacetic acid 2,4-Dichlorophenoxyacetic acid IUPAC (2,4-dichlorophenoxy)acetic acid name 2,4-D Other hedonal names trinoxol Identifiers CAS [94-75-7] number SMILES OC(COC1=CC=C(Cl)C=C1Cl)=O ChemSpider 1441 ID Properties Molecular C H Cl O formula 8 6 2 3 Molar mass 221.04 g mol−1 Appearance white to yellow powder Melting point 140.5 °C (413.5 K) Boiling 160 °C (0.4 mm Hg) point Solubility in 900 mg/L (25 °C) water Related compounds Related 2,4,5-T, Dichlorprop compounds Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) 2,4-Dichlorophenoxyacetic acid (2,4-D) is a common systemic herbicide used in the control of broadleaf weeds. It is the most widely used herbicide in the world, and the third most commonly used in North America.[1] 2,4-D is also an important synthetic auxin, often used in laboratories for plant research and as a supplement in plant cell culture media such as MS medium. History 2,4-D was developed during World War II by a British team at Rothamsted Experimental Station, under the leadership of Judah Hirsch Quastel, aiming to increase crop yields for a nation at war.[citation needed] When it was commercially released in 1946, it became the first successful selective herbicide and allowed for greatly enhanced weed control in wheat, maize (corn), rice, and similar cereal grass crop, because it only kills dicots, leaving behind monocots. Mechanism of herbicide action 2,4-D is a synthetic auxin, which is a class of plant growth regulators. -

Common and Chemical Names of Herbicides Approved by the WSSA
Weed Science 2010 58:511–518 Common and Chemical Names of Herbicides Approved by the Weed Science Society of America Below is the complete list of all common and chemical of herbicides as approved by the International Organization names of herbicides approved by the Weed Science Society of for Standardization (ISO). A sponsor may submit a proposal America (WSSA) and updated as of September 1, 2010. for a common name directly to the WSSA Terminology Beginning in 1996, it has been published yearly in the last Committee. issue of Weed Science with Directions for Contributors to A herbicide common name is not synonymous with Weed Science. This list is published in lieu of the selections a commercial formulation of the same herbicide, and in printed previously on the back cover of Weed Science. Only many instances, is not synonymous with the active ingredient common and chemical names included in this complete of a commercial formulation as identified on the product list should be used in WSSA publications. In the absence of label. If the herbicide is a salt or simple ester of a parent a WSSA-approved common name, the industry code number compound, the WSSA common name applies to the parent as compiled by the Chemical Abstracts Service (CAS) with compound only. CAS systematic chemical name or the systematic chemical The chemical name used in this list is that preferred by the name alone may be used. The current approved list is also Chemical Abstracts Service (CAS) according to their system of available at our web site (www.wssa.net). -

Risks of Linuron Use to Federally Threatened California Red-Legged Frog (Rana Aurora Draytonii)
Risks of Linuron Use to Federally Threatened California Red-legged Frog (Rana aurora draytonii) Pesticide Effects Determination Environmental Fate and Effects Division Office of Pesticide Programs Washington, D.C. 20460 June 19, 2008 Primary Authors: Michael Davy, Agronomist Wm. J. Shaughnessy, Ph.D, Environmental Scientist Environmental Risk Branch II Environmental Fate and Effects Division (7507C) Secondary Review: Donna Randall, Senior Effects Scientist Nelson Thurman, Senior Fate Scientist Environmental Risk Branch II Environmental Fate and Effects Division (7507P) Branch Chief, Environmental Risk Assessment Branch #: Arthur-Jean B. Williams, Acting Branch Chief Environmental Risk Branch II Environmental Fate and Effects Division (7507P) 2 Table of Contents 1. Executive Summary.................................................................................................8 2. Problem Formulation .............................................................................................14 2.1 Purpose...........................................................................................................................14 2.2 Scope..............................................................................................................................16 2.3 Previous Assessments ....................................................................................................18 2.4 Stressor Source and Distribution ...................................................................................19 2.4.1 Environmental Fate -

Delayed Fluorescence Imaging of Photosynthesis Inhibitor and Heavy Metal Induced Stress in Potato
Cent. Eur. J. Biol. • 7(3) • 2012 • 531-541 DOI: 10.2478/s11535-012-0038-z Central European Journal of Biology Delayed fluorescence imaging of photosynthesis inhibitor and heavy metal induced stress in potato Research Article Jaka Razinger1,*, Luka Drinovec2, Maja Berden-Zrimec3 1Agricultural Institute of Slovenia, 1000 Ljubljana, Slovenia 2Aerosol d.o.o., 1000 Ljubljana, Slovenia 3Institute of Physical Biology, 1000 Ljubljana, Slovenia Received 09 November 2011; Accepted 13 March 2012 Abstract: Early chemical-induced stress in Solanum tuberosum leaves was visualized using delayed fluorescence (DF) imaging. The ability to detect spatially heterogeneous responses of plant leaves exposed to several toxicants using delayed fluorescence was compared to prompt fluorescence (PF) imaging and the standard maximum fluorescence yield of PSII measurements (Fv/Fm). The toxicants used in the study were two photosynthesis inhibitors (herbicides), 100 μM methyl viologen (MV) and 140 μM diuron (DCMU), and two heavy metals, 100 μM cadmium and 100 μM copper. The exposure times were 5 and 72 h. Significant photosynthesis-inhibitor effects were already visualized after 5 h. In addition, a significant reduction in the DF/PF index was measured in DCMU- and MV-treated leaves after 5 h. In contrast, only DCMU-treated leaves exhibited a significant decrease in Fv/Fm after 5 h. All treatments resulted in a significant decrease in the DF/PF parameter after 72 h of exposure, when only MV and Cd treatment resulted in visible symptoms. Our study highlights the power of delayed fluorescence imaging. Abundant quantifiable spatial information was obtained with the instrumental setup. Delayed fluorescence imaging has been confirmed as a very responsive and useful technique for detecting stress induced by photosynthesis inhibitors or heavy metals. -

Evidence for Different Binding Sites on the 33-Kda Protein for DCMU, Atrazine and QB
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Volume 167, number 2 FEBS 1259 February 1984 Evidence for different binding sites on the 33-kDa protein for DCMU, atrazine and QB Chantal Astier, Alain Boussac and Anne-Lise Etienne Laboratoire de PhotosynthPse, CNRS, BP I, 91190 Gifsur-Yvette, France Received 21 November 1983; revised version received 4 January 1984 Two DCMU-resistant strains of the cyanobacterium Synechocystis 6714 were used to analyse the binding sites of DCMU, atrazine and QB. DCMU’-11~was DCMU and atrazine resistant; it presented an impaired electron flow and its 33-kDa protein was weakly attached to the membrane. DCMU’-IIB, derived from the former, simultaneously regained atrazine sensitivity, normal electron flow and a tight linkage of the 33-kDa protein to the membrane. This mutant shows that loss of DCMU binding does not necessarily affect the binding of either atrazine or QB. The role of the 33-kDa protein is discussed. Cyanobacteria Mutant Herbicide Photosynthesis Photosystem ZZ 1. INTRODUCTION that a specific attack of lysine residues resulted also in a partial release of inhibition by SN58132, Electron transfer on the acceptor side of a DCMU-type inhibitor [17]. Photosystem II occurs through a primary acceptor Radioactivity labelling experiments were used to QA [l] (a particular plastoquinone of the general demonstrate a competition between DCMU and pool [2] closely associated with the chlorophyll atrazine [6,18-201. A similar competition was pro- center), a secondary acceptor Qn [3] bound to its posed in [l l] between DCMU and QB. -

Interactions of Paraquat and Nitrodiphenylether Herbicides with the Chloroplast Photosynthetic Electron Transport in the Activation of Toxic Oxygen Species
INTERACTIONS OF PARAQUAT AND NITRODIPHENYLETHER HERBICIDES WITH THE CHLOROPLAST PHOTOSYNTHETIC ELECTRON TRANSPORT IN THE ACTIVATION OF TOXIC OXYGEN SPECIES by Brad Luther Upham Dissertation submitted to the Faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Plant Physiology APPROVED: Kriton K. Hatzios, Chairman -----..------,,...-r---~---------J ohn L. Hess Joyce G. Foster Laurence D. Moore David M. Orcutt William E. Winner INTERACTIONS OF PARAQUAT AND.NITRODIPHENYLETHER HERBICIDES WITH THE PHOTOSYNTHETIC ELECTRON TRANSPORT IN THE ACTIVATION OF TOXIC OXYGEN SPECIES by Brad Luther Upham Kriton K. Hatzios, Chairman Plant Physiology (ABSTRACT) The interactions of paraquat (methylviologen) and diphenylether her- bicides with the Mehler reaction as investigated. Sera from two differ- ent rabbits (RSl & RS2) were examined for their patterns of inhibition of the photosynthetic electron transport (PET) system. Serum from RS2 was greatly hemolyzed. Fifty ul of RSI serum were required for 100% inhibi- tion of a 1120 --> methylviologen(MV)/02 reaction, whereas only 10 µl of a 1:10 dilution of RS2 were needed for 100% inhibition. The ~-globulin fraction from purified rabbit serum (RSl) did not inhibit PET, indicat- ing that the antibody fraction of the rabbit serum does not contain the inhibitor. It appears that the inhibitor is from the hemolyzed red blood cells. Rabbit sera, added to chloroplast preparations prior illu- mination, caused no inhibition of a 1120 --> MV/02 reaction while addi- tion of rabbit sera during illumination inhibited the 1120 --> MV/02 reaction within 1-3 s. Various Hill reactions were used to determine the site of inhibition. -

Literature Review of Controlling Aquatic Invasive Vegetation With
Eurasian watermilfoil in Christmas Lake, 2011 Literature Review on Controlling Aquatic Invasive Vegetation with Aquatic Herbicides Compared to Other Control Methods: Effectiveness, Impacts, and Costs Prepared for: Prepared by: Minnehaha Creek Watershed District Steve McComas Blue Water Science St. Paul, MN 55116 September 2011 1 Literature Review on Controlling Aquatic Invasive Vegetation with Aquatic Herbicides Compared to Other Control Methods: Effectiveness, Impacts, and Costs Steve McComas, Blue Water Science Table of Contents page number Introduction .................................................................................................................................................................. 1 Use of Herbicides as an Aquatic Plant Control Technique ...................................................................................... 2 How Herbicides Work and Their Mode of Action ....................................................................................................... 3 Aquatic Herbicide Impacts on Humans and the Ecosystem ....................................................................................... 8 Where to Find Sources of Specific Information on herbicide Products and Their Active Ingredients ....................... 16 Harvesting, Drawdown, and Biocontrol as Aquatic Plant Control Techniques ................................................... 17 Summary of Control Techniques for Non-Native Curlyleaf Pondweed and Eurasian Watermilfoil ................... 25 Control Techniques for Other -

Analysis of 22 Base-Neutral Pesticides and Pesticide
Analysis of 22 Base-Neutral Pesticides and Pesticide Metabolites in Raw and Treated Waters Using Liquid Chromatography and Positive Ion Electrospray-Mass Spectrometry Application Water Analysis Authors Introduction Pam Chamberlain and Toni Hall Due to the widespread use of agricultural pesti- Wessex Water plc, cides and the consequential drainage into public Bath, UK waterways and underground water reservoirs, Agilent contact these types of compounds must be monitored for health and safety reasons, and in order to comply Paul Zavitsanos with governmental regulations and standards. Agilent Technologies, Inc. There are numerous and varied methods for the 2850 Centerville Road analysis of certain classes of pesticides. Some Wilmington, DE 19808-1610 examples include: a) separate analyses of triazines USA by gas chromatography (GC) [1], b) analysis of thermally unstable urons using liquid chromatog- Abstract raphy (LC) with UV diode array detection (DAD) [1], and c) an online method using solid-phase A method was defined for the quantitative analysis of extraction (SPE) with LC and DAD, and briefly by 22 pesticides and pesticide metabolites in raw and LC/MS (LC/mass spectrometry) [2]. We needed a treated drinking water. The method used solid-phase robust, high-throughput analytical method for extraction of the water sample, liquid chromatography varied pesticides, capable of validation to UKAS and electrospray-mass spectrometry. Results were shown (United Kingdom Accreditation Service) and DWI to meet UK regulatory requirements of recovery, (Drinking Water Inspectorate) requirements. To sensitivity, accuracy, and precision. develop an appropriate compound list, we com- bined pesticide data for the Wessex region, obtained from Central Science Lab (CSL, York), with stability and leaching data to predict problem pesticides. -

DCMU) Or Paraquat-Treated Euglena Gracilis Cells Erich F
Chlorophyll Photobleaching and Ethane Production in Dichlorophenyldimethylurea- (DCMU) or Paraquat-Treated Euglena gracilis Cells Erich F. Elstner and W. Osswald Institut für Botanik und Mikrobiologie, Technische Universität München, Arcisstr. 21, D-8000 München 2 Z. Naturforsch. 35 c, 129-135 (1980); received August 20/September 28, 1979 Chlorophyll Bleaching, Herbicides, Euglena gracilis, Ethane, Fat Oxidation Light dependent (35 Klux) chlorophyll bleaching in autotrophically grown Euglena gracilis cells at slightly acidic pH (6.5 —5.4) is stimulated by the photosystem II blockers DCMU and DBMIB (both 10~ 5 m) as well as by the autooxidizable photosystem I electron acceptor, paraquat (1 0 -3 m ). Chlorophyll photobleaching is accompanied by the formation of thiobarbituric acid — sensitive material (“malondialdehyde”) and ethane. Both chlorophyll photobleaching and light dependent ethane formation are partially prevented by higher concentrations (10~* m ) of the autooxidizable photosystem II electron acceptor DBMIB or by sodium bicarbonate (25 mM). In vitro studies with cell free extracts (homogenates) from E. gracilis suggest that a-linolenic acid oxidation by excited (reaction center II) chlorophyll represents the driving force for both ethane formation and chlorophyll bleaching. Ethane formation thus appears to be a sensitive and non-destructive “in vivo’’ marker for both restricted energy dissipation in photosystem II and, conditions yielding reactive oxygen species at the reducing side o f photosystem I. Introduction quent lipid attack and membrane destruction in the case of low potential photosystem I electron ac Chlorophyll bleaching is one of the characteristic ceptors as paraquat and other bipyridylium salts symptoms for plant diseases introduced by infec [4, 5, 9], tions, certain physical parameters or by chemicals. -
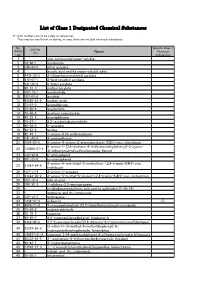
List of Class 1 Designated Chemical Substances
List of Class 1 Designated Chemical Substances *1:CAS numbers are to be solely as references. They may be insufficient or lacking, in case there are multiple chemical substances. No. Specific Class 1 CAS No. (PRTR Chemical (*1) Name Law) Substances 1 - zinc compounds(water-soluble) 2 79-06-1 acrylamide 3 140-88-5 ethyl acrylate 4 - acrylic acid and its water-soluble salts 5 2439-35-2 2-(dimethylamino)ethyl acrylate 6 818-61-1 2-hydroxyethyl acrylate 7 141-32-2 n-butyl acrylate 8 96-33-3 methyl acrylate 9 107-13-1 acrylonitrile 10 107-02-8 acrolein 11 26628-22-8 sodium azide 12 75-07-0 acetaldehyde 13 75-05-8 acetonitrile 14 75-86-5 acetone cyanohydrin 15 83-32-9 acenaphthene 16 78-67-1 2,2'-azobisisobutyronitrile 17 90-04-0 o-anisidine 18 62-53-3 aniline 19 82-45-1 1-amino-9,10-anthraquinone 20 141-43-5 2-aminoethanol 21 1698-60-8 5-amino-4-chloro-2-phenylpyridazin-3(2H)-one; chloridazon 5-amino-1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-3-cyano- 22 120068-37-3 4[(trifluoromethyl)sulfinyl]pyrazole; fipronil 23 123-30-8 p-aminophenol 24 591-27-5 m-aminophenol 4-amino-6-tert-butyl-3-methylthio-1,2,4-triazin-5(4H)-one; 25 21087-64-9 metribuzin 26 107-11-9 3-amino-1-propene 27 41394-05-2 4-amino-3-methyl-6-phenyl-1,2,4-triazin-5(4H)-one; metamitron 28 107-18-6 allyl alcohol 29 106-92-3 1-allyloxy-2,3-epoxypropane 30 - n-alkylbenzenesulfonic acid and its salts(alkyl C=10-14) 31 - antimony and its compounds 32 120-12-7 anthracene 33 1332-21-4 asbestos ○ 34 4098-71-9 3-isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate 35 78-84-2 isobutyraldehyde -

20210311 IAEG AD-DSL V5.0 for Pdf.Xlsx
IAEGTM AD-DSL Release Version 4.1 12-30-2020 Authority: IAEG Identity: AD-DSL Version number: 4.1 Issue Date: 2020-12-30 Key Yellow shading indicates AD-DSL family group entries, which can be expanded to display a non-exhaustive list of secondary CAS numbers belonging to the family group Substance Identification Change Log IAEG Regulatory Date First Parent Group IAEG ID CAS EC Name Synonyms Revision Date ECHA ID Entry Type Criteria Added IAEG ID IAEG000001 1327-53-3 215-481-4 Diarsenic trioxide Arsenic trioxide R1;R2;D1 2015-03-17 2015-03-17 100.014.075 Substance Direct Entry IAEG000002 1303-28-2 215-116-9 Diarsenic pentaoxide Arsenic pentoxide; Arsenic oxide R1;R2;D1 2015-03-17 2015-03-17 100.013.743 Substance Direct Entry IAEG000003 15606-95-8 427-700-2 Triethyl arsenate R1;R2;D1 2015-03-17 2017-08-14 100.102.611 Substance Direct Entry IAEG000004 7778-39-4 231-901-9 Arsenic acid R1;R2;D1 2015-03-17 2015-03-17 100.029.001 Substance Direct Entry IAEG000005 3687-31-8 222-979-5 Trilead diarsenate R1;R2;D1 2015-03-17 2017-08-14 100.020.890 Substance Direct Entry IAEG000006 7778-44-1 231-904-5 Calcium arsenate R1;R2;D1 2015-03-17 2017-08-14 100.029.003 Substance Direct Entry IAEG000009 12006-15-4 234-484-1 Cadmium arsenide Tricadmium diarsenide R1;R2;D1 2017-08-14 2017-08-14 Substance Direct Entry IAEG000021 7440-41-7 231-150-7 Beryllium (Be) R2 2015-03-17 2019-01-24 Substance Direct Entry IAEG000022 1306-19-0 215-146-2 Cadmium oxide R1;R2;D1 2015-03-17 2017-08-14 100.013.770 Substance Direct Entry IAEG000023 10108-64-2 233-296-7 Cadmium -

Determination of Atrazine and Degradation Products in Luxembourgish Drinking Water: Origin and Fate of Potential Endocrine-Disrupting Pesticides T
This article was downloaded by: [CRP Gabriel Lippmann (Centre de Recherch] On: 05 January 2012, At: 04:44 Publisher: Taylor & Francis Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer House, 37-41 Mortimer Street, London W1T 3JH, UK Food Additives & Contaminants: Part A Publication details, including instructions for authors and subscription information: http://www.tandfonline.com/loi/tfac20 Determination of atrazine and degradation products in Luxembourgish drinking water: origin and fate of potential endocrine-disrupting pesticides T. Bohn a , E. Cocco a , L. Gourdol a , C. Guignard a & L. Hoffmann a a Centre de Recherche Public – Gabriel Lippmann, Department of Environment and Agro- Biotechnologies, 41, rue du Brill, L-4422 Belvaux, Luxembourg Available online: 27 Jun 2011 To cite this article: T. Bohn, E. Cocco, L. Gourdol, C. Guignard & L. Hoffmann (2011): Determination of atrazine and degradation products in Luxembourgish drinking water: origin and fate of potential endocrine-disrupting pesticides, Food Additives & Contaminants: Part A, 28:8, 1041-1054 To link to this article: http://dx.doi.org/10.1080/19440049.2011.580012 PLEASE SCROLL DOWN FOR ARTICLE Full terms and conditions of use: http://www.tandfonline.com/page/terms-and-conditions This article may be used for research, teaching, and private study purposes. Any substantial or systematic reproduction, redistribution, reselling, loan, sub-licensing, systematic supply, or distribution in any form to anyone is expressly forbidden. The publisher does not give any warranty express or implied or make any representation that the contents will be complete or accurate or up to date. The accuracy of any instructions, formulae, and drug doses should be independently verified with primary sources.