The Abnormal Liver Chemistry Profile
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Evaluation of Abnormal Liver Chemistries
ACG Clinical Guideline: Evaluation of Abnormal Liver Chemistries Paul Y. Kwo, MD, FACG, FAASLD1, Stanley M. Cohen, MD, FACG, FAASLD2, and Joseph K. Lim, MD, FACG, FAASLD3 1Division of Gastroenterology/Hepatology, Department of Medicine, Stanford University School of Medicine, Palo Alto, California, USA; 2Digestive Health Institute, University Hospitals Cleveland Medical Center and Division of Gastroenterology and Liver Disease, Department of Medicine, Case Western Reserve University School of Medicine, Cleveland, Ohio, USA; 3Yale Viral Hepatitis Program, Yale University School of Medicine, New Haven, Connecticut, USA. Am J Gastroenterol 2017; 112:18–35; doi:10.1038/ajg.2016.517; published online 20 December 2016 Abstract Clinicians are required to assess abnormal liver chemistries on a daily basis. The most common liver chemistries ordered are serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase and bilirubin. These tests should be termed liver chemistries or liver tests. Hepatocellular injury is defined as disproportionate elevation of AST and ALT levels compared with alkaline phosphatase levels. Cholestatic injury is defined as disproportionate elevation of alkaline phosphatase level as compared with AST and ALT levels. The majority of bilirubin circulates as unconjugated bilirubin and an elevated conjugated bilirubin implies hepatocellular disease or cholestasis. Multiple studies have demonstrated that the presence of an elevated ALT has been associated with increased liver-related mortality. A true healthy normal ALT level ranges from 29 to 33 IU/l for males, 19 to 25 IU/l for females and levels above this should be assessed. The degree of elevation of ALT and or AST in the clinical setting helps guide the evaluation. -

Total Bilirubin in Athletes, Determination of Reference Range
OriginalTotal bilirubin Paper in athletes DOI: 10.5114/biolsport.2017.63732 Biol. Sport 2017;34:45-48 Total bilirubin in athletes, determination of reference range AUTHORS: Witek K1, Ścisłowska J2, Turowski D1, Lerczak K1, Lewandowska-Pachecka S2, Corresponding author: Pokrywka A3 Konrad Witek Department of Biochemistry, Institute of Sport - National 1 Department of Biochemistry, Institute of Sport - National Research Institute, Warsaw, Poland Research Institute 2 01-982 Warsaw, Poland Faculty of Pharmacy with the Laboratory Medicine Division, Medical University of Warsaw, Poland 3 E-mail: Department of Biochemistry, 2nd Faculty of Medicine, Medical University of Warsaw, Poland [email protected] ABSTRACT: The purpose of this study was to determine a typical reference range for the population of athletes. Results of blood tests of 339 athletes (82 women and 257 men, aged 18-37 years) were retrospectively analysed. The subjects were representatives of different sports disciplines. The measurements of total bilirubin (BIT), iron (Fe), alkaline phosphatase (ALP), alanine aminotransferase (ALT) and gamma-glutamyltransferase (GGT) were made using a Pentra 400 biochemical analyser (Horiba, France). Red blood cell count (RBC), reticulocyte count and haemoglobin concentration measurements were made using an Advia 120 haematology analyser (Siemens, Germany). In groups of women and men the percentage of elevated results were similar at 18%. Most results of total bilirubin in both sexes were in the range 7-14 μmol ∙ L-1 (49% of women and 42% of men). The highest results of elevated levels of BIT were in the range 21-28 μmol ∙ L-1 (12% of women and 11% of men). There was a significant correlation between serum iron and BIT concentration in female and male athletes whose serum total bilirubin concentration does not exceed the upper limit of the reference range. -

Normal Rangesа–А
Effective August 2007 5361 NW 33 rd Avenue Fort Lauderdale, FL 33309 954-777-0018 NORMAL RANGES – fax: 954-777-0211 ADULT MALE FEMALE CBC WBC 3.6 – 11.0 K/UL 3.6 – 11.0 RBC 4.5 – 5.9 M/UL 4.5 – 5.9 HEMOGLOBIN 13.0 – 18.0 G/DL 12.0 – 15.6 HEMATOCRIT 40 – 52 % 36.0 – 46.0 MCV 81 – 97 FL 81 93 MCH 26 – 34 PG 26 34 MCHC 31 – 37 gm/dl 31 37 RDW 11.5 – 15 % 11.5 – 15.0 PLATELET CT 150 – 400 K/UL 140 400 PLT VOL 7.4 – 10.4 FL 7.4 – 10.4 NEUTROPHIL % 36 – 66 36 75 LYMPHOCYTE % 23 – 43 27 47 MONOCYTE % 0.0 – 10 0.0 10 EOSINOPHIL % 0 – 5.0 0.0 – 5.0 BASOPHIL % 0 – 1.0 0.0 – 1.0 RETIC 0.4 – 1.9 0.4 – 1.9 COMPREHENSIVE METABOLIC PROFILE /LIVER PROFILE SODIUM 135 – 145 MMOL/L 135 145 POTASSIUM 3.5 – 5.5 MMOL/L 3.5 – 5.5 (OUTREACH) CHLORIDE 95 – 110 MMOL/L 95 110 CO2 19 – 34.MMOL/L 19 34 GLUCOSE 70 – 110 MG/DL 70 110 BUN 6 – 22 MG/DL 6 22 CREATININE 0.6 – 1.3 MG/DL 0.6 – 1.3 MG/DL CALCIUM 8.4 – 10.2 MG/DL 8.4 – 10.2 TOTAL PROTEIN 5.5 – 8.7 G/DL 5.5 – 8.7 ALBUMIN 3.2 – 5.0 g/dl 3.2 – 5.0 BILI TOTAL 0.1 – 1.2 MG/DL 0.1 – 1.2 BILI DIRECT 0.0 – 0.3 MG/DL 0.0 – 0.3 BILI INDIRECT 0.2 – 1.0 MG/DL 0.2 – 1.0 ALKALINE PHOS 20 – 130 U/L 20 130 AST/SGOT 10 – 40 U/L 10 40 ALT/SGPT 7 55 U/L 7 55 AST/ALT RATIO 0.5 – 0.9 RATIO 0.5 – 0.9 AMYLASE 25 – 115 U/L 25 – 115 U/L LIPASE 114 – 286 U/L 114 – 286 U/L CRP 0 – 0.9 MG/DL 0 – 0.9 MG/DL PREALBUMIN 18.0 – 35.7 mg/dL 18.0 – 35.7 mg/dL Revised 807 MAGNESIUM 1.8 – 2.4 mg/dL 1.8 – 2.4 mg/Dl LDH 100 – 200 Units /L 100 – 200 Units/L PHOSPHOROUS 2.3 – 5.0 mg/dL 2.3 – 5.0 mg/dL LACTIC ACID 0.4 -
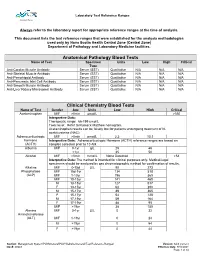
Laboratory Test Reference Ranges
Laboratory Test Reference Ranges Central Zone Always refer to the laboratory report for appropriate reference ranges at the time of analysis. This document lists the test reference ranges that were established for the analysis methodologies used only by Nova Scotia Health Central Zone (Central Zone) Department of Pathology and Laboratory Medicine facilities. Anatomical Pathology Blood Tests Name of Test Specimen Units Low High Critical Type Anti-Cardiac Muscle Antibody Serum (SST) Qualitative N/A N/A N/A Anti-Skeletal Muscle Antibody Serum (SST) Qualitative N/A N/A N/A Anti-Pemphigoid Antibody Serum (SST) Qualitative N/A N/A N/A Anti-Pancreatic Islet Cell Antibody Serum (SST) Qualitative N/A N/A N/A Anti-Smooth Muscle Antibody Serum (SST) Qualitative N/A N/A N/A Anti-Liver Kidney Microsomal Antibody Serum (SST) Qualitative N/A N/A N/A Clinical Chemistry Blood Tests Name of Test Gender Age Units Low High Critical Acetaminophen M/F >0min µmol/L >350 Interpretive Data: Therapeutic range: 66-199 umol/L Toxic level: Refer to Rumack Matthew nomogram. Acetaminophen results can be falsely low for patients undergoing treatment of N- acetylcysteine (NAC). Adrenocorticotropic M/F >0min pmol/L 2.3 10.1 hormone Interpretive Data: Adrenocorticotropic Hormone (ACTH) reference ranges are based on (ACTH) samples collected prior to 10 AM. Albumin M/F 0-1yr g/L 25 46 >1yr 35 50 Alcohol M/F >0min mmol/L None Detected >54 Interpretive Data: The method is intended for clinical purposes only. Medical-legal specimens should be analyzed by gas chromatographic method for confirmation of results. -

Journal of Blood Group Serology and Molecular Genetics Volume 34, Number 1, 2018 CONTENTS
Journal of Blood Group Serology and Molecular Genetics VOLUME 34, N UMBER 1, 2018 This issue of Immunohematology is supported by a contribution from Grifols Diagnostics Solutions, Inc. Dedicated to advancement and education in molecular and serologic immunohematology Immunohematology Journal of Blood Group Serology and Molecular Genetics Volume 34, Number 1, 2018 CONTENTS S EROLOGIC M ETHOD R EVIEW 1 Warm autoadsorption using ZZAP F.M. Tsimba-Chitsva, A. Caballero, and B. Svatora R EVIEW 4 Proceedings from the International Society of Blood Transfusion Working Party on Immunohaematology Workshop on the Clinical Significance of Red Blood Cell Alloantibodies, Friday, September 2, 2016, Dubai A brief overview of clinical significance of blood group antibodies M.J. Gandhi, D.M. Strong, B.I. Whitaker, and E. Petrisli C A S E R EPORT 7 Management of pregnancy sensitized with anti-Inb with monocyte monolayer assay and maternal blood donation R. Shree, K.K. Ma, L.S. Er and M. Delaney R EVIEW 11 Proceedings from the International Society of Blood Transfusion Working Party on Immunohaematology Workshop on the Clinical Significance of Red Blood Cell Alloantibodies, Friday, September 2, 2016, Dubai A review of in vitro methods to predict the clinical significance of red blood cell alloantibodies S.J. Nance S EROLOGIC M ETHOD R EVIEW 16 Recovery of autologous sickle cells by hypotonic wash E. Wilson, K. Kezeor, and M. Crosby TO THE E DITOR 19 The devil is in the details: retention of recipient group A type 5 years after a successful allogeneic bone marrow transplant from a group O donor L.L.W. -

Determination of Reference Ranges for Selected Clinical Laboratory Tests for a Medical Laboratory in Namibia Using Pre-Tested Data
DETERMINATION OF REFERENCE RANGES FOR SELECTED CLINICAL LABORATORY TESTS FOR A MEDICAL LABORATORY IN NAMIBIA USING PRE-TESTED DATA by CORNELIA DE WAAL-MILLER Student no: 197084516 Thesis submitted in fulfillment of the requirements of the degree Master of Technology: Biomedical Technology in the Faculty of Health and Wellness Sciences at the Cape Peninsula University of Technology Supervisor: Professor A.J. Esterhuyse Co-Supervisor: Professor B. Noden Bellville March 2015 CPUT copyright information The thesis may not be published either in part (in scholarly, scientific or technical journals), or as a whole (as a monograph), unless permission has been obtained from the University. (i) Declaration I, Cornelia de Waal-Miller, declare that the content of this thesis represents my own unaided work, and that this thesis has not previously been submitted for academic examination towards any qualification. Furthermore, this thesis represents my own opinions and not necessarily those of the Cape Peninsula University of Technology. 12th March 2015 Signed Date 2 | of 84 Pages (ii) Abstract Aim: The aim of the study was to compile pre-tested laboratory results stored in the laboratory database of the Namibia Institute of Pathology (NIP). The study also aimed to assess the usefulness and validity of using retrospective laboratory results of different patients in varying degrees of health and which were produced using various methods in different laboratories in Namibia. Methods: 254,271 test results (female: 134,261, male = 117,091, unknown gender= 2,919) consisting of Haemoglobin, serum Urea, serum Creatinine, plasma Glucose (fasting and random), serum Cholesterol, serum Triglycerides and serum Uric Acid was extracted from NIP Laboratory Information System over a period of four years and of the 13 different regions of Namibia were analyzed. -

Comparison of Histopathology, Immunofluorescence, and Serology
Global Dermatology Cae Report ISSN: 2056-7863 Comparison of histopathology, immunofluorescence, and serology for the diagnosis of autoimmune bullous disorders: an update Seline Ali E1, Seline Lauren N1, Sokumbi Olayemi1* and Motaparthi Kiran2,3 1Department of Dermatology, Medical College of Wisconsin, Milwaukee, WI, USA 2Dermatopathology, Miraca Life Sciences, USA 3Department of Dermatology, University of Texas Southwestern Medical Center, Dallas, TX, USA Introduction In an ELISA, the target antigen of interest (such as the NC16a domain of BP180) is immobilized by physical adsorption or by The diagnosis of autoimmune bullous disorders (AIBDs) relies on antibody capture. When antibody capture is utilized, this is referred to several different diagnostic methods. These include histopathology, as “sandwich ELISA” because the target antigen is bound between the direct immunofluorescence (DIF), indirect immunofluorescence immobilizing antibody and the primary antibody. Primary antibodies (IIF), enzyme-linked immunosorbent assay (ELISA) and are present in the patient’s serum. Enzyme-linked secondary antibodies immunoblotting. When faced with a presumptive AIBD, the are then added which bind the Fc region of primary antibodies. most widely employed method for diagnosis by dermatologists is Substrate is added and converted by the enzyme into a signal. A a combination of histopathology and DIF. While DIF is still the resulting color change, fluorescence, or electrochemical signal is diagnostic method of choice for linear IgA bullous disease and IgA quantitatively measured and reported [5]. pemphigus, ELISA is a more accurate, cost-effective and less invasive method of diagnosis for several AIBDs including pemphigus vulgaris Western blot is synonymous with immunoblot. For this method, and foliaceus, based on currently available evidence [1-3]. -

Online-Only Appendix: List of Inclusion and Exclusion Criteria
ONLINE-ONLY APPENDIX: LIST OF INCLUSION AND EXCLUSION CRITERIA INCLUSION CRITERIA [1] Male subjects between the ages of 35 and 70 years, inclusive, with Type 2 diabetes mellitus as defined by the American Diabetes Association (Diabetes care, Vol. 21: S5- S19, 1998) for more than one year. [2] Subjects must have Body Mass Index (BMI) < 36 kg/m². [3] Stable glycemic control (Hb A1C <11%). [4] Subjects must be off all oral hypoglycemic agents 24 hours prior to each study dosing day and off any investigational drug for at least four weeks. [5] Subjects must refrain from strenuous physical activity beginning 72 hours prior to admission and through the duration of the study. [6] Subjects must be willing and able to be confined to the Clinical Research Unit as required by the protocol. [7] Subjects must be willing and able to provide written informed consent. EXCLUSION CRITERIA [1] History or presence of clinically significant cardiovascular, respiratory, hepatic, renal, gastrointestinal, neurological or infectious disorders capable of altering the absorption, metabolism or elimination of drugs, or of constituting a risk factor when taking the study medication. [2] Subjects with gastroparesis, orthostatic hypotension and hypoglycemia unawareness (autonomic neuropathy). [3] Subjects with “brittle” diabetes or predisposition to severe hypoglycemia, e.g., 2 or more serious hypoglycemic episodes (requiring another’s assistance) within the past year, or any hospitalization or emergency room visit due to poor diabetic control within the past 6 months. [4] Evidence of significant active hematological disease and/or cumulative blood donation of 1 unit (500 mL) or more including blood drawn during clinical trials in the last 3 months. -

Application of Quantitative Immunofluorescence to Clinical Serology: Antibody Levels of Treponema Pallidum GRACE L
JOURNAL OF CLINICAL MICROBIOLOGY, May 1992, p. 1294-1296 Vol. 30, No. 5 0095-1137/92/051294-03$02.00/0 Copyright C 1992, American Society for Microbiology Application of Quantitative Immunofluorescence to Clinical Serology: Antibody Levels of Treponema pallidum GRACE L. PICCIOLO* AND DAVID S. KAPLAN Center for Devices and Radiological Health, Food and Drug Administration, 12200 Wilkins Avenue, Rockville, Maryland 20852 Received 27 March 1991/Accepted 20 January 1992 A previously reported method of quantitative immunofluorescence, employing a calibrated photometric system and chemically stabilized fluorescence intensity, was used to replace the subjective, visual method of endpoint determination with a quantitative, calibrated measurement of antibodies to Treponema paUlidum in serum. The results of the quantitative immunofluorescence method showed a 90% correlation with the subjective determinations of the visual method. A quantitative immunofluorescence (QIF) method to de- measured by using an acridine orange filter set, with a 400- to termine immunofluorescence with a quantitative, calibrated 440-nm excitation and LP 470 emission filters. photometric intensity of the fluorescent reaction product has Reducing agent. Dithioerythritol (DTE) (Sigma Chemical been reported previously by us (4). This method used uranyl Co., St. Louis, Mo.) was prepared as stock solution contain- glass slides in the calibration and standardization of the ing 0.3 M reducing agent in 0.5 M Tris buffer, pH 8.2 (Sigma microscope-photometer voltage measurements. To stabilize Chemical Co.). DTE was diluted 1:9 with standard buffered the fluorescence emission, dithioerythritol (DTE) was incor- glycerol mounting medium (Clinical Sciences, Inc., Whip- porated into the buffered glycerol mounting medium of the pany, N.J.) (4, 8). -

Clinical Placements Serology Screening for Students
Gawler Place Medical Practice Level 1, Key Invest Building 49 Gawler Place Adelaide SA 5000 T: 08 8212 7175 F: 08 8212 1993 E: [email protected] www.adelaideunicare.com.au CLINICAL PLACEMENTS SEROLOGY SCREENING FOR STUDENTS Gawler Place Medical Practice have put together this Question and Answer sheet to assist you in meeting the requirements of SA Health’s Immunisation for Health Care Workers in South Australia Policy – August 2017. It is a requirement that all Health Care Workers have adequate immunisation against certain diseases prior to working in a clinical environment. What is Serology Screening and why do I need to do it? Serology Screening is a blood test that looks for antibodies in your blood. The purpose of such a test is to detect serum antibodies to prove protective immunity against a disease. Where can I have Serology Screening done? Gawler Place Medical Practice offer Serology Screening appointments to allow incoming students to have their serology screening completed, and where required, obtain their required vaccinations to meet SA Health’s Immunisation for Health Care Workers in South Australia – August 2017. Australian Clinical Labs are located within our Practice so that you can have your blood taken (if required) as soon as you have seen the doctor. How do I make an appointment? You may contact the practice to organise an appointment, our telephone number is 8212 7175 or make a booking online. Please make a long appointment booking. If you need to change or cancel an appointment please contact our Practice on (08) 2127175 as soon as possible so that your appointment can be offered to another person. -

Calcium, Plasma, Total
Division of Laboratory Medicine Biochemistry Calcium, plasma, total Calcium is the most abundant mineral element in the body with about 99 percent in the bones primarily as hydroxyapatite. The remaining calcium is distributed between the various tissues and the extracellular fluids where it performs a vital role for many life sustaining processes. Among the extra skeletal functions of calcium are involvement in blood coagulation, neuromuscular conduction, excitability of skeletal and cardiac muscle, enzyme activation, and the preservation of cell membrane integrity and permeability. Calcium levels and hence the body content are controlled by parathyroid hormone (PTH), calcitonin, and vitamin D. To maintain calcium homeostasis, we require sufficient intake, normal metabolism and appropriate excretion. An imbalance in any of these modulators leads to alterations of the body and circulating calcium levels. Increases in serum PTH or vitamin D are usually associated with hypercalcemia. Increased calcium levels may also be observed in multiple myeloma and other neoplastic diseases. Hypocalcemia may be observed e.g. in hypoparathyroidism, nephrosis, and pancreatitis. Total Calcium is reported as direct and after adjustment for plasma albumin level. Adjusted total calcium has the same reference interval as total calcium and is a surrogate for ionised calcium. General information Collection container: Adults – serum (with gel separator, 4.9mL brown top Sarstedt tube) Paediatrics – lithium heparin plasma (1.2mL orange top Sarstedt tube) Type and volume of sample: The tubes should be thoroughly mixed before transport to the lab. 1mL whole blood is required as a minimum volume. Specimen transport/special precautions: N/A Laboratory information Method principle: Calcium ions react with 5‑nitro‑5’‑methyl‑BAPTA (NM‑BAPTA) under alkaline conditions to form a complex. -

Lab Dept: Chemistry Test Name: VENOUS BLOOD GAS (VBG)
Lab Dept: Chemistry Test Name: VENOUS BLOOD GAS (VBG) General Information Lab Order Codes: VBG Synonyms: Venous blood gas CPT Codes: 82803 - Gases, blood, any combination of pH, pCO2, pO2, CO2, HCO3 (including calculated O2 saturation) Test Includes: VpH (no units), VpCO2 and VpO2 measured in mmHg, VsO2 and VO2AD measured in %, HCO3 and BE measured in mmol/L, Temperature (degrees C) and ST (specimen type) Logistics Test Indications: Useful for evaluating oxygen and carbon dioxide gas exchange; respiratory function, including hypoxia; and acid/base balance. It is also useful in assessment of asthma; chronic obstructive pulmonary disease and other types of lung disease; embolism, including fat embolism; and coronary artery disease. Lab Testing Sections: Chemistry Phone Numbers: MIN Lab: 612-813-6280 STP Lab: 651-220-6550 Test Availability: Daily, 24 hours Turnaround Time: 30 minutes Special Instructions: See Collection and Patient Preparation Specimen Specimen Type: Whole blood Container: Preferred: Sims Portex® syringe (PB151) or Smooth-E syringe (956- 463) Draw Volume: 0.4 mL (Minimum: 0.2 mL) blood Note: Submission of 0.2 mL of blood does not allow for repeat analysis. Processed Volume: 0.2 mL blood per analysis Collection: Avoid using a tourniquet. Anaerobically collect blood into a heparinized blood gas syringe (See Container. Once the puncture has been performed or the line specimen drawn, immediately remove all air from the syringe. Remove the needle, cap tightly and gently mix. Do not expose the specimen to air. Forward the specimen immediately at ambient temperature. Specimens cannot be stored. Note: When drawing from an indwelling catheter, the line must be thoroughly flushed with blood before drawing the sample.