STEP THERAPY CRITERIA BRAND NAME* (Generic) (Desvenlafaxine Fumarate Extended-Release Tablets)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Pristiq (Desvenlafaxine Succinate) – First-Time Generic
Pristiq® (desvenlafaxine succinate) – First-time generic • On March 1, 2017, Teva launched AB-rated generic versions of Pfizer’s Pristiq (desvenlafaxine succinate) 25 mg, 50 mg, and 100 mg extended-release tablets for the treatment of major depressive disorder. — Teva launched the 25 mg tablet with 180-day exclusivity. — In addition, Alembic/Breckenridge, Mylan, and West-Ward have launched AB-rated generic versions of Pristiq 50 mg and 100 mg extended-release tablets. — Greenstone’s launch plans for authorized generic versions of Pristiq 25 mg, 50 mg, and 100 mg tablets are pending. — Lupin and Sandoz received FDA approval of AB-rated generic versions of Pristiq 50 mg and 100 mg tablets on June 29, 2015. Lupin’s and Sandoz’s launch plans are pending. • Other serotonin-norepinephrine reuptake inhibitors approved for the treatment of major depressive disorder include desvenlafaxine fumarate, duloxetine, Fetzima™ (levomilnacipran), Khedezla™ (desvenlafaxine) , venlafaxine, and venlafaxine extended-release. • Pristiq and the other serotonin-norepinephrine reuptake inhibitors carry a boxed warning for suicidal thoughts and behaviors. • According to IMS Health data, the U.S. sales of Pristiq were approximately $883 million for the 12 months ending on December 31, 2016. optumrx.com OptumRx® specializes in the delivery, clinical management and affordability of prescription medications and consumer health products. We are an Optum® company — a leading provider of integrated health services. Learn more at optum.com. All Optum® trademarks and logos are owned by Optum, Inc. All other brand or product names are trademarks or registered marks of their respective owners. This document contains information that is considered proprietary to OptumRx and should not be reproduced without the express written consent of OptumRx. -

Desvenlafaxine
https://providers.amerigroup.com CONTAINS CONFIDENTIAL PATIENT INFORMATION desvenlafaxine Prior Authorization of Benefits (PAB) Form Complete form in its entirety and fax to: Prior Authorization of Benefits Center at 1-844-512-9004 Provider Help Desk 1-800-454-3730 1. PATIENT INFORMATION 2. PHYSICIAN INFORMATION Patient Name: Prescribing Physician: Patient ID #: Physician Address: Patient DOB: Physician Phone #: Date of Rx: Physician Fax #: Patient Phone #: Physician Specialty: Patient Email Address: Physician DEA: Physician NPI #: Physician Email Address: 3. MEDICATION 4. STRENGTH 5. DIRECTIONS 6. QUANTITY PER 30 DAYS desvenlafaxine Specify: 7. DIAGNOSIS: 8. APPROVAL CRITERIA: CHECK ALL BOXES THAT APPLY NOTE: Any areas not filled out are considered not applicable to your patient & MAY AFFECT THE OUTCOME of this request. □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with two preferred generic SSRIs* has been provided If No: □ Yes □ No Documented evidence is provided that the use of these agents would be medically contraindicated □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with one preferred generic SNRI** has been provided If No: □ Yes □ No Documented evidence is provided that the use of these agents would be medically contraindicated □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with one non-SSRI/SNRI generic antidepressant** has been provided If No: □ Yes □ No Documented evidence is provided that the use of these -
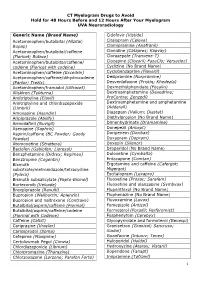
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Antidepressants, Other Review 04/14/2009
Antidepressants, Other Review 04/14/2009 Copyright © 2004 - 2009 by Provider Synergies, L.L.C. All rights reserved. Printed in the United States of America. All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying, recording, digital scanning, or via any information storage and retrieval system without the express written consent of Provider Synergies, L.L.C. All requests for permission should be mailed to: Attention: Copyright Administrator Intellectual Property Department Provider Synergies, L.L.C. 5181 Natorp Blvd., Suite 205 Mason, Ohio 45040 The materials contained herein represent the opinions of the collective authors and editors and should not be construed to be the official representation of any professional organization or group, any state Pharmacy and Therapeutics committee, any state Medicaid Agency, or any other clinical committee. This material is not intended to be relied upon as medical advice for specific medical cases and nothing contained herein should be relied upon by any patient, medical professional or layperson seeking information about a specific course of treatment for a specific medical condition. All readers of this material are responsible for independently obtaining medical advice and guidance from their own physician and/or other medical professional in regard to the best course of treatment for their specific medical condition. This publication, inclusive of all forms contained herein, is intended to be -

Desvenlafaxine Drug Assessment Pristiq® in Major Depressive Report Disorder in Adults @DTB Navarre.Es More Cost for Less
03 2015 DESVENLAFAXINE DRUG ASSESSMENT Pristiq® in major depressive REPORT www.dtb.navarra.es disorder in adults @DTB_Navarre.es More cost for less... ABSTRACT Desvenlafaxine is an active me- tabolite of venlafaxine. Indications1 In the study against escitalopram4 flexible Treatment of major depressive disorder doses of desvenlafaxine were used (100 and In three placebo-controlled 200 mg daily) in postmenopausal women. No trials, the results on the reduc- 1 Mechanism of action advantage of desvenlafaxine over escitalopram tion in HAM-D17 score were not This is an active metabolite of venlafaxine, that was found. consistent. inhibits the reuptake of serotonine and nora- There is only one long-term study16 that eva- dernaline. Its bioavailability reaches up to 80% luated relapse prevention. Patients responding The most common adverse while elimination occurs without alteration after 8 weeks of treatment with desvenlafaxine effects are of gastrointestinal through the urine (45%) and metabolism by 50 mg daily and with a stable response up to origin or sleep disorders. There glucurono conjugation (19%). week 20 were randomized either to placebo or are no available long-term safe- desvenlafaxine 50 mg daily for 6 months. The ty data. Dosage and administration1 endpoint was time to relapse (defined as HAM- The recommended dose is 50 mg daily. The ta- D17 score ≥16), treatment withdrawal due to In the only head-to-head trial blets are swallowed wholly, with liquid with or unsatisfactory response, hospital admission carried out in post-menopause without food and at the same time. The increa- due to depression, suicide attempt or suicide. women, desvenlafaxine at high se in doses should be gradual and up to a maxi- Time to relapse was significantly lower in the doses did not show superiority mum of 200 mg daily and at intervals of at least case of placebo compared to desvenlafaxine versus escitalopram. -

Pharmacokinetic and Pharmacodynamic Interactions Between Antiepileptics and Antidepressants Domenico Italiano University of Messina, Italy
University of Kentucky UKnowledge Psychiatry Faculty Publications Psychiatry 11-2014 Pharmacokinetic and Pharmacodynamic Interactions between Antiepileptics and Antidepressants Domenico Italiano University of Messina, Italy Edoardo Spina University of Messina, Italy Jose de Leon University of Kentucky, [email protected] Right click to open a feedback form in a new tab to let us know how this document benefits oy u. Follow this and additional works at: https://uknowledge.uky.edu/psychiatry_facpub Part of the Psychiatry and Psychology Commons Repository Citation Italiano, Domenico; Spina, Edoardo; and de Leon, Jose, "Pharmacokinetic and Pharmacodynamic Interactions between Antiepileptics and Antidepressants" (2014). Psychiatry Faculty Publications. 40. https://uknowledge.uky.edu/psychiatry_facpub/40 This Article is brought to you for free and open access by the Psychiatry at UKnowledge. It has been accepted for inclusion in Psychiatry Faculty Publications by an authorized administrator of UKnowledge. For more information, please contact [email protected]. Pharmacokinetic and Pharmacodynamic Interactions between Antiepileptics and Antidepressants Notes/Citation Information Published in Expert Opinion on Drug Metabolism & Toxicology, v. 10, Issue 11, p. 1457-1489. © 2014 Taylor & Francis Group This is an Accepted Manuscript of an article published by Taylor & Francis Group in Expert Opinion on Drug Metabolism & Toxicology in Nov. 2014, available online: http://www.tandfonline.com/10.1517/ 17425255.2014.956081 Digital Object Identifier (DOI) http://dx.doi.org/10.1517/17425255.2014.956081 This article is available at UKnowledge: https://uknowledge.uky.edu/psychiatry_facpub/40 1 This is an Accepted Manuscript of an article published by Taylor & Francis Group in Expert Opinion on Drug Metabolism & Toxicology in Nov. -

Anticonvulsants Antipsychotics Benzodiazepines/Anxiolytics ADHD
Anticonvulsants Antidepressants Generic Name Brand Name Generic Name Brand Name Carbamazepine Tegretol SSRI Divalproex Depakote Citalopram Celexa Lamotrigine Lamictal Escitalopram Lexapro Topirimate Topamax Fluoxetine Prozac Fluvoxamine Luvox Paroxetine Paxil Antipsychotics Sertraline Zoloft Generic Name Brand Name SNRI Typical Desvenlafaxine Pristiq Chlorpromazine Thorazine Duloextine Cymbalta Fluphenazine Prolixin Milnacipran Savella Haloperidol Haldol Venlafaxine Effexor Perphenazine Trilafon SARI Atypical Nefazodone Serzone Aripiprazole Abilify Trazodone Desyrel Clozapine Clozaril TCA Lurasidone Latuda Clomimpramine Enafranil Olanzapine Zyprexa Despiramine Norpramin Quetiapine Seroquel Nortriptyline Pamelor Risperidone Risperal MAOI Ziprasidone Geodon Phenylzine Nardil Selegiline Emsam Tranylcypromine Parnate Benzodiazepines/Anxiolytics DNRI Generic Name Brand Name Bupropion Wellbutrin Alprazolam Xanax Clonazepam Klonopin Sedative‐Hypnotics Lorazepam Ativan Generic Name Brand Name Diazepam Valium Clonidine Kapvay Buspirone Buspar Eszopiclone Lunesta Pentobarbital Nembutal Phenobarbital Luminal ADHD Medicines Zaleplon Sonata Generic Name Brand Name Zolpidem Ambien Stimulant Amphetamine Adderall Others Dexmethylphenidate Focalin Generic Name Brand Name Dextroamphetamine Dexedrine Antihistamine Methylphenidate Ritalin Non‐stimulant Diphenhydramine Bendryl Atomoxetine Strattera Anti‐tremor Guanfacine Intuniv Benztropine Cogentin Mood stabilizer (bipolar treatment) Lithium Lithane Never Mix, Never Worry: What Clinicians Need to Know about -

HIGHLIGHTS of PRESCRIBING INFORMATION These Highlights Do Not Include All the Information Needed to Use APTRYXOL Safely and Effe
HIGHLIGHTS OF PRESCRIBING INFORMATION within 7 days of stopping treatment with desvenlafaxine extended- These highlights do not include all the information needed to use release tablets. Do not use desvenlafaxine extended release tablets APTRYXOL safely and effectively. See full prescribing information for within 14 days of stopping an MAOI intended to treat psychiatric APTRYXOL. disorders. In addition, do not start desvenlafaxine extended release tablets in a patient who is being treated with linezolid or intravenous APTRYXOL™ (desvenlafaxine) extended-release tablets, for oral use methylene blue (4). Initial U.S. Approval: 2008 -----------------------WARNINGS AND PRECAUTIONS----------------------- • Serotonin Syndrome: Increased risk when co-administered with other WARNING: SUICIDAL THOUGHTS AND BEHAVIORS serotonergic agents (e.g., SSRI, SNRI, triptans), but also when taken See full prescribing information for complete boxed warning. alone. If it occurs, discontinue desvenlafaxine extended-release tablets and initiate supportive treatment (5.2). • Increased risk of suicidal thoughts and behaviors in children, • Elevated Blood Pressure: Control hypertension before initiating adolescents and young adults taking antidepressants (5.1). treatment. Monitor blood pressure regularly during treatment (5.3). • Closely monitor for clinical worsening and emergence of suicidal • Increased Risk of Bleeding: Concomitant use of aspirin, NSAIDs, other thoughts and behaviors (5.1). antiplatelet drugs, warfarin and other anticoagulants may increase this • Desvenlafaxine is not approved for use in pediatric patients (8.4). risk (5.4). • Angle Closure Glaucoma: Avoid use of antidepressants, including desvenlafaxine extended-release tablets, in patients with untreated anatomically narrow angles treated (5.5) ----------------------------INDICATIONS AND USAGE-------------------------- • Activation of Mania/Hypomania: Use cautiously in patients with Bipolar APTRYXOL (desvenlafaxine), is a serotonin and norepinephrine reuptake Disorder. -

Desvenlafaxine Versus Venlafaxine for the Treatment of Adult Patients with Major Depressive Disorder: a Review of the Comparative Clinical and Cost-Effectiveness
CADTH RAPID RESPONSE REPORT: SUMMARY WITH CRITICAL APPRAISAL Desvenlafaxine versus Venlafaxine for the Treatment of Adult Patients with Major Depressive Disorder: A Review of the Comparative Clinical and Cost-Effectiveness Service Line: Rapid Response Service Version: 1.0 Publication Date: October 25, 2017 Report Length: 15 Pages Authors: Veronica Poitras, Sarah Visintini Cite As: Desv enlaf axine v ersus Venlaf axine for the Treatment of Adult Patients with Major Depressiv e Disorder: A Rev iew of the Comparativ e Clinical and Cost-Ef f ectiveness. Ottawa: CADTH; 2017 Oct. (CADTH rapid response report: summary with critical appraisal). Acknowledgments: ISSN: 1922-8147 (online) Disclaimer: The inf ormation in this document is intended to help Canadian health care decision-makers, health care prof essionals, health sy stems leaders, and policy -makers make well-inf ormed decisions and thereby improv e the quality of health care serv ices. While patients and others may access this document, the document is made av ailable f or inf ormational purposes only and no representations or warranties are made with respect to its f itness f or any particular purpose. The inf ormation in this document should not be used as a substitute f or prof essional medical adv ice or as a substitute f or the application of clinical judgment in respect of the care of a particular patient or other prof essional judgment in any decision-making process. The Canadian Agency f or Drugs and Technologies in Health (CADTH) does not endorse any inf ormation, drugs, therapies, treatments, products, processes, or serv ic es. While care has been taken to ensure that the inf ormation prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was f irst published by CADTH, CADTH does not make any guarantees to that ef f ect. -

Preferred Drug List
October 2021 Preferred Drug List The Preferred Drug List, administered by CVS Caremark® on behalf of Siemens, is a guide within select therapeutic categories for clients, plan members and health care providers. Generics should be considered the first line of prescribing. If there is no generic available, there may be more than one brand-name medicine to treat a condition. These preferred brand-name medicines are listed to help identify products that are clinically appropriate and cost-effective. Generics listed in therapeutic categories are for representational purposes only. This is not an all-inclusive list. This list represents brand products in CAPS, branded generics in upper- and lowercase Italics, and generic products in lowercase italics. PLAN MEMBER HEALTH CARE PROVIDER Your benefit plan provides you with a prescription benefit program Your patient is covered under a prescription benefit plan administered administered by CVS Caremark. Ask your doctor to consider by CVS Caremark. As a way to help manage health care costs, prescribing, when medically appropriate, a preferred medicine from authorize generic substitution whenever possible. If you believe a this list. Take this list along when you or a covered family member brand-name product is necessary, consider prescribing a brand name sees a doctor. on this list. Please note: Please note: • Your specific prescription benefit plan design may not cover • Generics should be considered the first line of prescribing. certain products or categories, regardless of their appearance in • This drug list represents a summary of prescription coverage. It is this document. Products recently approved by the U.S. Food and not all-inclusive and does not guarantee coverage. -

STEP THERAPY CRITERIA BRAND NAME (Generic) (Desvenlafaxine Fumarate Extended-Release Tablets)
STEP THERAPY CRITERIA BRAND NAME (generic) (desvenlafaxine fumarate extended-release tablets) FETZIMA (levomilnacipran) KHEDEZLA (desvenlafaxine extended release tablets) PRISTIQ (desvenlafaxine succinate extended release tablets) Status: CVS Caremark Criteria Type: Initial Step Therapy; Post Step Therapy Prior Authorization with Limit POLICY FDA-APPROVED INDICATIONS Desvenlafaxine Desvenlafaxine, a serotonin and norepinephrine reuptake inhibitor (SNRI), is indicated for the treatment of major depressive disorder (MDD). The efficacy of desvenlafaxine has been established in four short-term (8-week, placebo- controlled studies) of outpatients who met DSM-IV criteria for major depressive disorder. Fetzima Fetzima, a serotonin and norepinephrine reuptake inhibitor (SNRI) is indicated for the treatment of major depressive disorder (MDD). The efficacy of Fetzima was established in three 8-week, randomized, double-blind, placebo-controlled studies in adult patients with a diagnosis of MDD Limitation of Use: Fetzima is not approved for the management of fibromyalgia. The efficacy and safety of Fetzima for the management of fibromyalgia have not been established. Khedezla Khedezla, a serotonin and norepinephrine reuptake inhibitor (SNRI), is indicated for the treatment of major depressive disorder (MDD). The efficacy of desvenlafaxine has been established in four short-term (8-week, placebo-controlled studies) in adult outpatients who met DSM-IV criteria for major depressive disorder. Pristiq Pristiq is indicated for the treatment of adults with -

Antidepressants for Major Depressive Disorder: Drug Information to Support Drug Therapy Decisions
Antidepressants for Major Depressive Disorder: Drug Information to Support Drug Therapy Decisions B.C. Provincial Academic Detailing (PAD) Service March 2020 Background This PAD education session focuses on commonly prescribed and recently marketed antidepressants for major depressive disorder. After achieving remission of an initial depressive episode, clinical practice guidelines recommend several months of continued antidepressant therapy (recommendations vary from 4 to 12 months) and longer if someone is at risk for relapse or has experienced recurrent episodes.1-6 A recent primary care trial found that discontinuing antidepressant therapy can be challenging even for people considered candidates for discontinuation according to guidelines.7 51% of people in the trial agreed to try a taper to discontinue their antidepressant therapy when provided with the recommendation from their primary care physician. Ten of 146 people considered eligible for antidepressant discontinuation were no longer taking an antidepressant at the end of follow up. Acknowledging that the most appropriate duration of antidepressant therapy is not known,8 participants in this PAD session will have the opportunity to discuss drug information that informs several decisions regarding antidepressants for non-pregnant adults, including: 1. How the efficacy of antidepressants is Antidepressant Brand Name Marketed9 measured in clinical trials and reported in meta-analyses Fluoxetine Prozac 1989 Sertraline Zoloft 1992 2. The quality and quantity of evidence that informs conclusions regarding Paroxetine Paxil 1993 antidepressant comparisons and Venlafaxine Effexor 1994 combinations Bupropion Wellbutrin 1998 3. Drug information relevant to the Citalopram Celexa 1999 prescribing, deprescribing and monitoring Mirtazapine Remeron 2001 of antidepressants, including: Escitalopram Cipralex 2005 . dose response Duloxetine Cymbalta 2008 .