Mcl-1: a Highly Regulated Cell Death and Survival Controller
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

MARCH5 Requires MTCH2 to Coordinate Proteasomal Turnover of the MCL1:NOXA Complex
Cell Death & Differentiation (2020) 27:2484–2499 https://doi.org/10.1038/s41418-020-0517-0 ARTICLE MARCH5 requires MTCH2 to coordinate proteasomal turnover of the MCL1:NOXA complex 1,2 1,2,5 1,2 1,2 1,2,3 1,2 Tirta Mario Djajawi ● Lei Liu ● Jia-nan Gong ● Allan Shuai Huang ● Ming-jie Luo ● Zhen Xu ● 4 1,2 1,2 1,2 Toru Okamoto ● Melissa J. Call ● David C. S. Huang ● Mark F. van Delft Received: 3 July 2019 / Revised: 6 February 2020 / Accepted: 7 February 2020 / Published online: 24 February 2020 © The Author(s) 2020. This article is published with open access Abstract MCL1, a BCL2 relative, is critical for the survival of many cells. Its turnover is often tightly controlled through both ubiquitin-dependent and -independent mechanisms of proteasomal degradation. Several cell stress signals, including DNA damage and cell cycle arrest, are known to elicit distinct E3 ligases to ubiquitinate and degrade MCL1. Another trigger that drives MCL1 degradation is engagement by NOXA, one of its BH3-only protein ligands, but the mechanism responsible has remained unclear. From an unbiased genome-wide CRISPR-Cas9 screen, we discovered that the ubiquitin E3 ligase MARCH5, the ubiquitin E2 conjugating enzyme UBE2K, and the mitochondrial outer membrane protein MTCH2 co- — fi 1234567890();,: 1234567890();,: operate to mark MCL1 for degradation by the proteasome speci cally when MCL1 is engaged by NOXA. This mechanism of degradation also required the MCL1 transmembrane domain and distinct MCL1 lysine residues to proceed, suggesting that the components likely act on the MCL1:NOXA complex by associating with it in a specific orientation within the mitochondrial outer membrane. -

Simultaneously Inhibiting BCL2 and MCL1 Is a Therapeutic Option for Patients with Advanced Melanoma
cancers Article Simultaneously Inhibiting BCL2 and MCL1 Is a Therapeutic Option for Patients with Advanced Melanoma Nabanita Mukherjee 1, Carol M. Amato 2 , Jenette Skees 1, Kaleb J. Todd 1, Karoline A. Lambert 1, William A. Robinson 2, Robert Van Gulick 2, Ryan M. Weight 2, Chiara R. Dart 2, Richard P. Tobin 3, Martin D. McCarter 3, Mayumi Fujita 1,4,5 , David A. Norris 1,4 and Yiqun G. Shellman 1,5,* 1 Department of Dermatology, School of Medicine, University of Colorado Anschutz Medical Campus, Mail Stop 8127, Aurora, CO 80045, USA; [email protected] (N.M.); [email protected] (J.S.); [email protected] (K.J.T.); [email protected] (K.A.L.); [email protected] (M.F.); [email protected] (D.A.N.) 2 Division of Medical Oncology, School of Medicine, University of Colorado Anschutz Medical Campus, Mail Stop 8117, Aurora, CO 80045, USA; [email protected] (C.M.A.); [email protected] (W.A.R.); [email protected] (R.V.G.); [email protected] (R.M.W.); [email protected] (C.R.D.) 3 Division of Surgical Oncology, Department of Surgery, University of Colorado Anschutz Medical Campus, Aurora, CO 80045, USA; [email protected] (R.P.T.); [email protected] (M.D.M.) 4 Dermatology Section, Department of Veterans Affairs Medical Center, Denver, CO 80220, USA 5 Gates Center for Regenerative Medicine, University of Colorado Anschutz Medical Campus, Aurora, CO 80045, USA * Correspondence: [email protected]; Tel.: +1-303-724-4034; Fax: +1-303-724-4048 Received: 30 June 2020; Accepted: 31 July 2020; Published: 5 August 2020 Abstract: There is an urgent need to develop treatments for patients with melanoma who are refractory to or ineligible for immune checkpoint blockade, including patients who lack BRAF-V600E/K mutations. -

Synergy of Bcl2 and Histone Deacetylase Inhibition Against Leukemic Cells from Cutaneous T-Cell Lymphoma Patients Benoit Cyrenne
Yale University EliScholar – A Digital Platform for Scholarly Publishing at Yale Yale Medicine Thesis Digital Library School of Medicine January 2018 Synergy Of Bcl2 And Histone Deacetylase Inhibition Against Leukemic Cells From Cutaneous T-Cell Lymphoma Patients Benoit Cyrenne Follow this and additional works at: https://elischolar.library.yale.edu/ymtdl Recommended Citation Cyrenne, Benoit, "Synergy Of Bcl2 And Histone Deacetylase Inhibition Against Leukemic Cells From Cutaneous T-Cell Lymphoma Patients" (2018). Yale Medicine Thesis Digital Library. 3388. https://elischolar.library.yale.edu/ymtdl/3388 This Open Access Thesis is brought to you for free and open access by the School of Medicine at EliScholar – A Digital Platform for Scholarly Publishing at Yale. It has been accepted for inclusion in Yale Medicine Thesis Digital Library by an authorized administrator of EliScholar – A Digital Platform for Scholarly Publishing at Yale. For more information, please contact [email protected]. i Synergy of BCL2 and histone deacetylase inhibition against leukemic cells from cutaneous T-cell lymphoma patients A Thesis Submitted to the Yale University School of Medicine in Partial Fulfillment of the Requirements for the Degree of Doctor of Medicine Benoit M. Cyrenne 2018 ii SYNERGY OF BCL2 AND HISTONE DEACETYLASE INHIBITION AGAINST LEUKEMIC CELLS FROM CUTANEOUS T-CELL LYMPHOMA PATIENTS. Benoit Cyrenne, Julia Lewis, Jason Weed, Kacie Carlson, Fatima Mirza, Francine Foss, and Michael Girardi. Department of Dermatology, Yale University, School of Medicine, New Haven, CT. The presence and degree of peripheral blood involvement in patients with cutaneous T-cell lymphoma (CTCL) portend a worse clinical outcome. Available systemic therapies for CTCL may variably decrease tumor burden and improve quality of life, but offer limited effects on survival; thus, novel approaches to the treatment of advanced stages of this non-Hodgkin lymphoma are clearly warranted. -

BCL-2 Family Proteins: Changing Partners in the Dance Towards Death
Cell Death and Differentiation (2018) 25, 65–80 OPEN Official journal of the Cell Death Differentiation Association www.nature.com/cdd Review BCL-2 family proteins: changing partners in the dance towards death Justin Kale1, Elizabeth J Osterlund1,2 and David W Andrews*,1,2,3 The BCL-2 family of proteins controls cell death primarily by direct binding interactions that regulate mitochondrial outer membrane permeabilization (MOMP) leading to the irreversible release of intermembrane space proteins, subsequent caspase activation and apoptosis. The affinities and relative abundance of the BCL-2 family proteins dictate the predominate interactions between anti-apoptotic and pro-apoptotic BCL-2 family proteins that regulate MOMP. We highlight the core mechanisms of BCL-2 family regulation of MOMP with an emphasis on how the interactions between the BCL-2 family proteins govern cell fate. We address the critical importance of both the concentration and affinities of BCL-2 family proteins and show how differences in either can greatly change the outcome. Further, we explain the importance of using full-length BCL-2 family proteins (versus truncated versions or peptides) to parse out the core mechanisms of MOMP regulation by the BCL-2 family. Finally, we discuss how post- translational modifications and differing intracellular localizations alter the mechanisms of apoptosis regulation by BCL-2 family proteins. Successful therapeutic intervention of MOMP regulation in human disease requires an understanding of the factors that mediate the major binding interactions between BCL-2 family proteins in cells. Cell Death and Differentiation (2018) 25, 65–80; doi:10.1038/cdd.2017.186; published online 17 November 2017 The membrane plays an active role in most BCL-2 family interactions by changing the affinities and local relative abundance of these proteins. -

The AAA-Atpase P97 Is Essential for Outer Mitochondrial Membrane Protein Turnover
MBoC | ARTICLE The AAA-ATPase p97 is essential for outer mitochondrial membrane protein turnover Shan Xu a, b,* , Guihong Peng a, b,* , Yang Wang a, c , Shengyun Fang a, c , and Mariusz Karbowskia, b a Center for Biomedical Engineering and Technology, University of Maryland, Baltimore, MD 21201; b Department of Biochemistry & Molecular Biology, c Department of Physiology, University of Maryland School of Medicine, Baltimore, MD 21201 ABSTRACT Recent studies have revealed a role for the ubiquitin/proteasome system in the Monitoring Editor regulation and turnover of outer mitochondrial membrane (OMM)-associated proteins. Al- Benjamin Glick though several molecular components required for this process have been identifi ed, the University of Chicago mechanism of proteasome-dependent degradation of OMM-associated proteins is currently Received: Sep 3 , 2010 unclear. We show that an AAA-ATPase, p97, is required for the proteasomal degradation of Revised: Oct 26 , 2010 Mcl1 and Mfn1, two unrelated OMM proteins with short half-lives. A number of biochemical Accepted: Nov 18 , 2010 assays, as well as imaging of changes in localization of photoactivable GFP-fused Mcl1, re- vealed that p97 regulates the retrotranslocation of Mcl1 from mitochondria to the cytosol, prior to, or concurrent with, proteasomal degradation. Mcl1 retrotranslocation from the OMM depends on the activity of the ATPase domain of p97. Furthermore, p97-mediated retrotranslocation of Mcl1 can be recapitulated in vitro, confi rming a direct mitochondrial role for p97. Our results establish p97 as a novel and essential component of the OMM-associated protein degradation pathway. INTRODUCTION Mitochondria are the primary site of energy production in animal such as mitochondrial membrane dynamics. -

Multi-Modal Effects of 1B3, a Novel Synthetic Mir-193A-3P Mimic, Support Strong Potential for Therapeutic Intervention in Oncology
www.oncotarget.com Oncotarget, 2021, Vol. 12, (No. 5), pp: 422-439 Research Paper Multi-modal effects of 1B3, a novel synthetic miR-193a-3p mimic, support strong potential for therapeutic intervention in oncology Bryony J. Telford1, Sanaz Yahyanejad1, Thijs de Gunst1, Harm C. den Boer1, Rogier M. Vos1, Marieke Stegink1, Marion T.J. van den Bosch1, Mir Farshid Alemdehy1, Laurens A.H. van Pinxteren1, Roel Q.J. Schaapveld1 and Michel Janicot1 1InteRNA Technologies BV, Utrecht, The Netherlands Correspondence to: Michel Janicot, email: [email protected] Keywords: miR-193a-3p; microRNA mimic; microRNA delivery in vivo; pleiotropic mechanism of microRNA Received: October 16, 2020 Accepted: February 01, 2021 Published: March 02, 2021 Copyright: © 2021 Telford et al. This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Compelling evidence demonstrates that miR-193a-3p is a tumor suppressor microRNA in many cancer types, and its reduced expression is linked to cancer initiation and progression, metastasis, and therapy resistance. However, its mechanism of action is not consistently described between studies, and often contradicts the pleiotropic role of a microRNA in manipulating several different mRNA targets. We therefore comprehensively investigated miRNA-193a-3p's mode of action in a panel of human cancer cell lines, with a variety of genetic backgrounds, using 1B3, a synthetic microRNA mimic. Interestingly, the exact mechanism through which 1B3 reduced cell proliferation varied between cell lines. 1B3 efficiently reduced target gene expression, leading to reduced cell proliferation/survival, cell cycle arrest, induction of apoptosis, increased cell senescence, DNA damage, and inhibition of migration. -

Anti-NOXA Antibody (ARG64942)
Product datasheet [email protected] ARG64942 Package: 100 μg, 50 μg anti-NOXA antibody Store at: -20°C Summary Product Description Goat Polyclonal antibody recognizes NOXA Tested Reactivity Hu Tested Application WB Host Goat Clonality Polyclonal Isotype IgG Target Name NOXA Antigen Species Human Immunogen C-TQLRRFGDKLNFRQK Conjugation Un-conjugated Alternate Names NOXA; APR; Phorbol-12-myristate-13-acetate-induced protein 1; PMA-induced protein 1; Immediate- early-response protein APR; Protein Noxa Application Instructions Application table Application Dilution WB 0.03 - 0.1 µg/ml Application Note WB: Recommend incubate at RT for 1h. * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Calculated Mw 6 kDa Properties Form Liquid Purification Purified from goat serum by ammonium sulphate precipitation followed by antigen affinity chromatography using the immunizing peptide. Buffer Tris saline (pH 7.3), 0.02% Sodium azide and 0.5% BSA Preservative 0.02% Sodium azide Stabilizer 0.5% BSA Concentration 0.5 mg/ml Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. www.arigobio.com 1/2 Note For laboratory research only, not for drug, diagnostic or other use. Bioinformation Database links GeneID: 5366 Human Swiss-port # Q13794 Human Gene Symbol PMAIP1 Gene Full Name phorbol-12-myristate-13-acetate-induced protein 1 Function Promotes activation of caspases and apoptosis. -

Knockout of Vdac1 Activates Hypoxia-Inducible Factor Through
Brahimi-Horn et al. Cancer & Metabolism (2015) 3:8 DOI 10.1186/s40170-015-0133-5 Cancer & Metabolism RESEARCH Open Access Knockout of Vdac1 activates hypoxia- inducible factor through reactive oxygen species generation and induces tumor growth by promoting metabolic reprogramming and inflammation M. Christiane Brahimi-Horn1†, Sandy Giuliano1†, Estelle Saland2, Sandra Lacas-Gervais3, Tatiana Sheiko4, Joffrey Pelletier1, Isabelle Bourget5, Frédéric Bost6, Chloé Féral5, Etienne Boulter5, Michel Tauc7, Mircea Ivan8, Barbara Garmy-Susini9, Alexandra Popa10, Bernard Mari10, Jean-Emmanuel Sarry2, William J. Craigen4, Jacques Pouysségur1,11 and Nathalie M. Mazure1* Abstract Background: Mitochondria are more than just the powerhouse of cells; they dictate if a cell dies or survives. Mitochondria are dynamic organelles that constantly undergo fusion and fission in response to environmental conditions. We showed previously that mitochondria of cells in a low oxygen environment (hypoxia) hyperfuse to form enlarged or highly interconnected networks with enhanced metabolic efficacy and resistance to apoptosis. Modifications to the appearance and metabolic capacity of mitochondria have been reported in cancer. However, the precise mechanisms regulating mitochondrial dynamics and metabolism in cancer are unknown. Since hypoxia plays a role in the generation of these abnormal mitochondria, we questioned if it modulates mitochondrial function. The mitochondrial outer-membrane voltage-dependent anion channel 1 (VDAC1) is at center stage in regulating metabolism and apoptosis. We demonstrated previously that VDAC1 was post-translationally C-terminal cleaved not only in various hypoxic cancer cells but also in tumor tissues of patients with lung adenocarcinomas. Cells with enlarged mitochondria and cleaved VDAC1 were also more resistant to chemotherapy-stimulated cell death than normoxic cancer cells. -
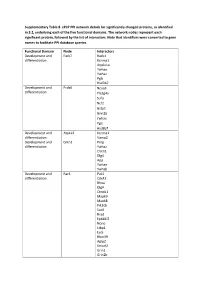
Supplementary Table 8. Cpcp PPI Network Details for Significantly Changed Proteins, As Identified in 3.2, Underlying Each of the Five Functional Domains
Supplementary Table 8. cPCP PPI network details for significantly changed proteins, as identified in 3.2, underlying each of the five functional domains. The network nodes represent each significant protein, followed by the list of interactors. Note that identifiers were converted to gene names to facilitate PPI database queries. Functional Domain Node Interactors Development and Park7 Rack1 differentiation Kcnma1 Atp6v1a Ywhae Ywhaz Pgls Hsd3b7 Development and Prdx6 Ncoa3 differentiation Pla2g4a Sufu Ncf2 Gstp1 Grin2b Ywhae Pgls Hsd3b7 Development and Atp1a2 Kcnma1 differentiation Vamp2 Development and Cntn1 Prnp differentiation Ywhaz Clstn1 Dlg4 App Ywhae Ywhab Development and Rac1 Pak1 differentiation Cdc42 Rhoa Dlg4 Ctnnb1 Mapk9 Mapk8 Pik3cb Sod1 Rrad Epb41l2 Nono Ltbp1 Evi5 Rbm39 Aplp2 Smurf2 Grin1 Grin2b Xiap Chn2 Cav1 Cybb Pgls Ywhae Development and Hbb-b1 Atp5b differentiation Hba Kcnma1 Got1 Aldoa Ywhaz Pgls Hsd3b4 Hsd3b7 Ywhae Development and Myh6 Mybpc3 differentiation Prkce Ywhae Development and Amph Capn2 differentiation Ap2a2 Dnm1 Dnm3 Dnm2 Atp6v1a Ywhab Development and Dnm3 Bin1 differentiation Amph Pacsin1 Grb2 Ywhae Bsn Development and Eef2 Ywhaz differentiation Rpgrip1l Atp6v1a Nphp1 Iqcb1 Ezh2 Ywhae Ywhab Pgls Hsd3b7 Hsd3b4 Development and Gnai1 Dlg4 differentiation Development and Gnao1 Dlg4 differentiation Vamp2 App Ywhae Ywhab Development and Psmd3 Rpgrip1l differentiation Psmd4 Hmga2 Development and Thy1 Syp differentiation Atp6v1a App Ywhae Ywhaz Ywhab Hsd3b7 Hsd3b4 Development and Tubb2a Ywhaz differentiation Nphp4 -

Expression Profile of BCL-2, BCL-XL and MCL-1 Predicts Pharmacological
Author Manuscript Published OnlineFirst on March 3, 2016; DOI: 10.1158/1535-7163.MCT-15-0730 Author manuscripts have been peer reviewed and accepted for publication but have not yet been edited. Expression profile of BCL-2, BCL-XL and MCL-1 predicts pharmacological response to the BCL-2 selective antagonist venetoclax in multiple myeloma models Elizabeth Punnoose1*, Joel D Leverson2*, Franklin Peale3, Erwin R Boghaert4, Lisa Belmont5, Nguyen Tan5, Amy Young6, Michael Mitten4, Ellen Ingalla6, Walter Darbonne1, Anatol Oleksijew4, Paul Tapang4, Peng Yue5, Jason Oeh6, Leslie Lee6, Sophie Maiga7, Wayne J Fairbrother8, Martine Amiot7, Andrew J Souers4, Deepak Sampath6# 1Oncology Biomarkers, Genentech 2Oncology Development, AbbVie, Inc. 3Research Pathology, Genentech 4Oncology Discovery, AbbVie, Inc. 5Discovery Oncology, Genentech 6Translational Oncology, Genentech 7INSERM UMR892, CNRS UMR6299, University of Nantes, France 8Early Discovery Biochemistry, Genentech *These authors contributed equally #Address correspondence to Deepak Sampath, Genentech, 1 DNA Way, South San Francisco, CA 94080. [email protected] Running Title: Predictive biomarkers of venetoclax efficacy in myeloma 1 Downloaded from mct.aacrjournals.org on September 24, 2021. © 2016 American Association for Cancer Research. Author Manuscript Published OnlineFirst on March 3, 2016; DOI: 10.1158/1535-7163.MCT-15-0730 Author manuscripts have been peer reviewed and accepted for publication but have not yet been edited. Keywords: BCL-2 pro-survival proteins, multiple myeloma, drug resistance, predictive biomarkers Abbreviations: BCL-2 (B-cell lymphoma 2), BCL-XL (B-cell lymphoma-extra large or BCL2-like 1 isoform 1), MCL-1 (Myeloid Cell Leukemia 1), BIM (BCL-2-like protein 11), MM (Multiple Myeloma), HMCL (human myeloma cell line). -

Targeting of BCL2 Family Proteins With
Published OnlineFirst October 14, 2015; DOI: 10.1158/1078-0432.CCR-15-1191 Cancer Therapy: Preclinical Clinical Cancer Research Targeting of BCL2 Family Proteins with ABT-199 and Homoharringtonine Reveals BCL2- and MCL1-Dependent Subgroups of Diffuse Large B-Cell Lymphoma Magdalena Klanova1,2, Ladislav Andera3, Jan Brazina3, Jan Svadlenka3, Simona Benesova3, Jan Soukup4, Dana Prukova1, Dana Vejmelkova2, Radek Jaksa5, Karel Helman6, Petra Vockova1,2, Lucie Lateckova1,2, Jan Molinsky1,2, Bokang Calvin Lenyeletse Maswabi1, Mahmudul Alam1, Roman Kodet4, Robert Pytlik2, Marek Trneny2, and Pavel Klener1,2 Abstract Purpose: To investigate the roles of BCL2, MCL1, and BCL-XL dependent and MCL1-dependent subgroups with a less pro- in the survival of diffuse large B-cell lymphoma (DLBCL). nounced role left for BCL-XL. Derived DLBCL clones with manip- Experimental designs: Immunohistochemical analysis of 105 ulated expressions of BCL2, MCL1, and BCL-XL, as well as the primary DLBCL samples, and Western blot analysis of 18 DLBCL immunoprecipitation experiments, which analyzed MCL1 pro- cell lines for the expression of BCL2, MCL1, and BCL-XL. Phar- tein complexes, confirmed these findings at the molecular level. macologic targeting of BCL2, MCL1, and BCL-XL with ABT-199, We demonstrated that concurrent inhibition of BCL2 and MCL1 homoharringtonine (HHT), and ABT-737. Analysis of DLBCL with ABT-199 and HHT induced significant synthetic lethality in clones with manipulated expressions of BCL2, MCL1, and BCL- most BCL2-expressing DLBCL cell lines. The marked cytotoxic XL. Immunoprecipitation of MCL1 complexes in selected DLBCL synergy between ABT-199 and HHT was also confirmed in vivo cell lines. -
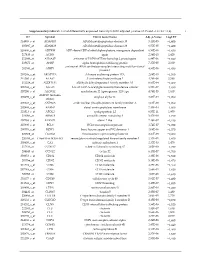
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at