Acid-Sensing Ion Channels Emerged Over 600 Mya and Are Conserved Throughout the Deuterostomes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Targeting Ion Channels in Cancer: a Novel Frontier in Antineoplastic Therapy A
66 Current Medicinal Chemistry, 2009, 16, 66-93 Targeting Ion Channels in Cancer: A Novel Frontier in Antineoplastic Therapy A. Arcangeli*,1, O. Crociani1, E. Lastraioli1, A. Masi1, S. Pillozzi1 and A. Becchetti2 1Department of Experimental Pathology and Oncology, University of Firenze, Italy; 2Department of Biotechnology and Biosciences, University of Milano-Bicocca, Italy Abstract: Targeted therapy is considerably changing the treatment and prognosis of cancer. Progressive understanding of the molecular mechanisms that regulate the establishment and progression of different tumors is leading to ever more spe- cific and efficacious pharmacological approaches. In this picture, ion channels represent an unexpected, but very promising, player. The expression and activity of different channel types mark and regulate specific stages of cancer progression. Their contribution to the neoplastic phenotype ranges from control of cell proliferation and apoptosis, to regulation of invasiveness and metastatic spread. As is being in- creasingly recognized, some of these roles can be attributed to signaling mechanisms independent of ion flow. Evidence is particularly extensive for K+ channels. Their expression is altered in many primary human cancers, especially in early stages, and they frequently exert pleiotropic effects on the neoplastic cell physiology. For instance, by regulating membrane potential they can control Ca2+ fluxes and thus the cell cycle machinery. Their effects on mitosis can also de- pend on regulation of cell volume, usually in cooperation with chloride channels. However, ion channels are also impli- cated in late neoplastic stages, by stimulating angiogenesis, mediating the cell-matrix interaction and regulating cell motil- ity. Not surprisingly, the mechanisms of these effects are manifold. -

The Chondrocyte Channelome: a Novel Ion Channel Candidate in the Pathogenesis of Pectus Deformities
Old Dominion University ODU Digital Commons Biological Sciences Theses & Dissertations Biological Sciences Summer 2017 The Chondrocyte Channelome: A Novel Ion Channel Candidate in the Pathogenesis of Pectus Deformities Anthony J. Asmar Old Dominion University, [email protected] Follow this and additional works at: https://digitalcommons.odu.edu/biology_etds Part of the Biology Commons, Molecular Biology Commons, and the Physiology Commons Recommended Citation Asmar, Anthony J.. "The Chondrocyte Channelome: A Novel Ion Channel Candidate in the Pathogenesis of Pectus Deformities" (2017). Doctor of Philosophy (PhD), Dissertation, Biological Sciences, Old Dominion University, DOI: 10.25777/pyha-7838 https://digitalcommons.odu.edu/biology_etds/19 This Dissertation is brought to you for free and open access by the Biological Sciences at ODU Digital Commons. It has been accepted for inclusion in Biological Sciences Theses & Dissertations by an authorized administrator of ODU Digital Commons. For more information, please contact [email protected]. THE CHONDROCYTE CHANNELOME: A NOVEL ION CHANNEL CANDIDATE IN THE PATHOGENESIS OF PECTUS DEFORMITIES by Anthony J. Asmar B.S. Biology May 2010, Virginia Polytechnic Institute M.S. Biology May 2013, Old Dominion University A Dissertation Submitted to the Faculty of Old Dominion University in Partial Fulfillment of the Requirements for the Degree of DOCTOR OF PHILOSOPHY BIOMEDICAL SCIENCES OLD DOMINION UNIVERSITY August 2017 Approved by: Christopher Osgood (Co-Director) Michael Stacey (Co-Director) Lesley Greene (Member) Andrei Pakhomov (Member) Jing He (Member) ABSTRACT THE CHONDROCYTE CHANNELOME: A NOVEL ION CHANNEL CANDIDATE IN THE PATHOGENESIS OF PECTUS DEFORMITIES Anthony J. Asmar Old Dominion University, 2017 Co-Directors: Dr. Christopher Osgood Dr. Michael Stacey Costal cartilage is a type of rod-like hyaline cartilage connecting the ribs to the sternum. -

Supplementary Table 1. Pain and PTSS Associated Genes (N = 604
Supplementary Table 1. Pain and PTSS associated genes (n = 604) compiled from three established pain gene databases (PainNetworks,[61] Algynomics,[52] and PainGenes[42]) and one PTSS gene database (PTSDgene[88]). These genes were used in in silico analyses aimed at identifying miRNA that are predicted to preferentially target this list genes vs. a random set of genes (of the same length). ABCC4 ACE2 ACHE ACPP ACSL1 ADAM11 ADAMTS5 ADCY5 ADCYAP1 ADCYAP1R1 ADM ADORA2A ADORA2B ADRA1A ADRA1B ADRA1D ADRA2A ADRA2C ADRB1 ADRB2 ADRB3 ADRBK1 ADRBK2 AGTR2 ALOX12 ANO1 ANO3 APOE APP AQP1 AQP4 ARL5B ARRB1 ARRB2 ASIC1 ASIC2 ATF1 ATF3 ATF6B ATP1A1 ATP1B3 ATP2B1 ATP6V1A ATP6V1B2 ATP6V1G2 AVPR1A AVPR2 BACE1 BAMBI BDKRB2 BDNF BHLHE22 BTG2 CA8 CACNA1A CACNA1B CACNA1C CACNA1E CACNA1G CACNA1H CACNA2D1 CACNA2D2 CACNA2D3 CACNB3 CACNG2 CALB1 CALCRL CALM2 CAMK2A CAMK2B CAMK4 CAT CCK CCKAR CCKBR CCL2 CCL3 CCL4 CCR1 CCR7 CD274 CD38 CD4 CD40 CDH11 CDK5 CDK5R1 CDKN1A CHRM1 CHRM2 CHRM3 CHRM5 CHRNA5 CHRNA7 CHRNB2 CHRNB4 CHUK CLCN6 CLOCK CNGA3 CNR1 COL11A2 COL9A1 COMT COQ10A CPN1 CPS1 CREB1 CRH CRHBP CRHR1 CRHR2 CRIP2 CRYAA CSF2 CSF2RB CSK CSMD1 CSNK1A1 CSNK1E CTSB CTSS CX3CL1 CXCL5 CXCR3 CXCR4 CYBB CYP19A1 CYP2D6 CYP3A4 DAB1 DAO DBH DBI DICER1 DISC1 DLG2 DLG4 DPCR1 DPP4 DRD1 DRD2 DRD3 DRD4 DRGX DTNBP1 DUSP6 ECE2 EDN1 EDNRA EDNRB EFNB1 EFNB2 EGF EGFR EGR1 EGR3 ENPP2 EPB41L2 EPHB1 EPHB2 EPHB3 EPHB4 EPHB6 EPHX2 ERBB2 ERBB4 EREG ESR1 ESR2 ETV1 EZR F2R F2RL1 F2RL2 FAAH FAM19A4 FGF2 FKBP5 FLOT1 FMR1 FOS FOSB FOSL2 FOXN1 FRMPD4 FSTL1 FYN GABARAPL1 GABBR1 GABBR2 GABRA2 GABRA4 -

On Obtaining Estimates of Parent-Of-Origin Effects Effectively and Their Exploitation in Association Genetic Mapping
Aus dem Leibniz-Institut für Nutztierbiologie (FBN) Institut für Genetik und Biometrie Dummerstorf ON OBTAINING ESTIMATES OF PARENT-OF-ORIGIN EFFECTS EFFECTIVELY AND THEIR EXPLOITATION IN ASSOCIATION GENETIC MAPPING Dissertation zur Erlangung des Doktorgrades der Agrar- und Ernährungswissenschaftlichen Fakultät der Christian-Albrechts-Universität zu Kiel vorgelegt von M.Sc. agr. Inga Blunk aus Bad Segeberg, Schleswig-Holstein Dekan: Prof. Dr. Joachim Krieter 1. Berichterstatter: Prof. Dr. Norbert Reinsch 2. Berichterstatter: Prof. Dr. Georg Thaller Tag der mündlichen Prüfung: 6. November 2017 Die Dissertation wurde mit dankenswerter finanzieller Unterstützung der H. Wilhelm Schaumann Stiftung, Hamburg, angefertigt. „Das Schönste, was wir erleben können, ist das Geheimnisvolle. …“ ALBERT EINSTEIN TABLE OF CONTENTS General Introduction ………………………………………………………………………. 1 Chapter 1 ………………………………………………………………………………..…… 12 Genetic variance components when fluctuating imprinting patterns are present Chapter 2 ………………………………………………………………………………..…… 21 A new model for parent-of-origin effect analyses applied to Brown Swiss cattle slaughterhouse data Chapter 3 ………………………………………………………………………………..…… 46 Parsimonious model for analyzing parent-of-origin effects related to beef traits in dual-purpose Simmental Chapter 4 ……………………………………………………………………………………. 72 Scanning the genomes of parents for imprinted loci acting in their ungenotyped progeny General Discussion ………………………………………………………………………. 113 Summary ……………………………………………………………………………………. 123 Zusammenfassung ………………………………………………………………………. -

Transcriptomic Profiling of Ca Transport Systems During
cells Article Transcriptomic Profiling of Ca2+ Transport Systems during the Formation of the Cerebral Cortex in Mice Alexandre Bouron Genetics and Chemogenomics Lab, Université Grenoble Alpes, CNRS, CEA, INSERM, Bâtiment C3, 17 rue des Martyrs, 38054 Grenoble, France; [email protected] Received: 29 June 2020; Accepted: 24 July 2020; Published: 29 July 2020 Abstract: Cytosolic calcium (Ca2+) transients control key neural processes, including neurogenesis, migration, the polarization and growth of neurons, and the establishment and maintenance of synaptic connections. They are thus involved in the development and formation of the neural system. In this study, a publicly available whole transcriptome sequencing (RNA-Seq) dataset was used to examine the expression of genes coding for putative plasma membrane and organellar Ca2+-transporting proteins (channels, pumps, exchangers, and transporters) during the formation of the cerebral cortex in mice. Four ages were considered: embryonic days 11 (E11), 13 (E13), and 17 (E17), and post-natal day 1 (PN1). This transcriptomic profiling was also combined with live-cell Ca2+ imaging recordings to assess the presence of functional Ca2+ transport systems in E13 neurons. The most important Ca2+ routes of the cortical wall at the onset of corticogenesis (E11–E13) were TACAN, GluK5, nAChR β2, Cav3.1, Orai3, transient receptor potential cation channel subfamily M member 7 (TRPM7) non-mitochondrial Na+/Ca2+ exchanger 2 (NCX2), and the connexins CX43/CX45/CX37. Hence, transient receptor potential cation channel mucolipin subfamily member 1 (TRPML1), transmembrane protein 165 (TMEM165), and Ca2+ “leak” channels are prominent intracellular Ca2+ pathways. The Ca2+ pumps sarco/endoplasmic reticulum Ca2+ ATPase 2 (SERCA2) and plasma membrane Ca2+ ATPase 1 (PMCA1) control the resting basal Ca2+ levels. -

Genetic Mapping of ASIC4 and Contrasting Phenotype to Asic1a in Modulating Innate Fear and Anxiety
European Journal of Neuroscience, Vol. 41, pp. 1553–1568, 2015 doi:10.1111/ejn.12905 BEHAVIORAL NEUROSCIENCE Genetic mapping of ASIC4 and contrasting phenotype to ASIC1a in modulating innate fear and anxiety Shing-Hong Lin,1,2 Ya-Chih Chien,2 Wei-Wei Chiang,3 Yan-Zhen Liu,3 Cheng-Chang Lien4 and Chih-Cheng Chen1,2,3 1Graduate institute of Life Sciences, National Defense Medical Center, Taipei, Taiwan 2Institute of Biomedical Sciences, Academia Sinica, Taipei 115, Taiwan 3Taiwan Mouse Clinic-National Comprehensive Mouse Phenotyping and Drug Testing Center, Academia Sinica, Taipei, Taiwan 4Institute of Neuroscience, National Yang-Ming University, Taipei, Taiwan Keywords: ASIC, behavioral phenotyping, Cre, interneuron, knockout mice Abstract Although ASIC4 is a member of the acid-sensing ion channel (ASIC) family, we have limited knowledge of its expression and physiological function in vivo. To trace the expression of this ion channel, we generated the ASIC4-knockout/CreERT2-knockin (Asic4CreERT2) mouse line. After tamoxifen induction in the Asic4CreERT2::CAG-STOPfloxed-Td-tomato double transgenic mice, we mapped the expression of ASIC4 at the cellular level in the central nervous system (CNS). ASIC4 was expressed in many brain regions, including the olfactory bulb, cerebral cortex, striatum, hippocampus, amygdala, thalamus, hypothalamus, brain stem, cer- ebellum, spinal cord and pituitary gland. Colocalisation studies further revealed that ASIC4 was expressed mainly in three types of cells in the CNS: (i) calretinin (CR)-positive and/or vasoactive intestine peptide (VIP)-positive interneurons; (ii) neural/glial anti- gen 2 (NG2)-positive glia, also known as oligodendrocyte precursor cells; and (iii) cerebellar granule cells. To probe the possible role of ASIC4, we hypothesised that ASIC4 could modulate the membrane expression of ASIC1a and thus ASIC1a signaling in vivo. -

Multiple Modulation of Acid-Sensing Ion Channel 1A by the Alkaloid Daurisoline
biomolecules Article Multiple Modulation of Acid-Sensing Ion Channel 1a by the Alkaloid Daurisoline Dmitry I. Osmakov 1,2,* , Sergey G. Koshelev 1, Ekaterina N. Lyukmanova 1, Mikhail A. Shulepko 1, Yaroslav A. Andreev 1,2, Peter Illes 3 and Sergey A. Kozlov 1 1 Shemyakin-Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences, ul. Miklukho-Maklaya 16/10, 117997 Moscow, Russia 2 Institute of Molecular Medicine, Sechenov First Moscow State Medical University, Trubetskaya str. 8, bld. 2, 119991 Moscow, Russia 3 Rudolf-Boehm-Institut für Pharmakologie und Toxikologie, University of Leipzig, 04107 Leipzig, Germany * Correspondence: [email protected] Received: 26 June 2019; Accepted: 1 August 2019; Published: 2 August 2019 Abstract: Acid-sensing ion channels (ASICs) are proton-gated sodium-selective channels that are expressed in the peripheral and central nervous systems. ASIC1a is one of the most intensively studied isoforms due to its importance and wide representation in organisms, but it is still largely unexplored as a target for therapy. In this study, we demonstrated response of the ASIC1a to acidification in the presence of the daurisoline (DAU) ligand. DAU alone did not activate the channel, but in combination with protons, it produced the second peak component of the ASIC1a current. This second peak differs from the sustained component (which is induced by RF-amide peptides), as the second (DAU-induced) peak is completely desensitized, with the same kinetics as the main peak. The co-application of DAU and mambalgin-2 indicated that their binding sites do not overlap. Additionally,we found an asymmetry in the pH activation curve of the channel, which was well-described by a mathematical model based on the multiplied probabilities of protons binding with a pool of high-cooperative sites and a single proton binding with a non-cooperative site. -

Ion Channels
UC Davis UC Davis Previously Published Works Title THE CONCISE GUIDE TO PHARMACOLOGY 2019/20: Ion channels. Permalink https://escholarship.org/uc/item/1442g5hg Journal British journal of pharmacology, 176 Suppl 1(S1) ISSN 0007-1188 Authors Alexander, Stephen PH Mathie, Alistair Peters, John A et al. Publication Date 2019-12-01 DOI 10.1111/bph.14749 License https://creativecommons.org/licenses/by/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California S.P.H. Alexander et al. The Concise Guide to PHARMACOLOGY 2019/20: Ion channels. British Journal of Pharmacology (2019) 176, S142–S228 THE CONCISE GUIDE TO PHARMACOLOGY 2019/20: Ion channels Stephen PH Alexander1 , Alistair Mathie2 ,JohnAPeters3 , Emma L Veale2 , Jörg Striessnig4 , Eamonn Kelly5, Jane F Armstrong6 , Elena Faccenda6 ,SimonDHarding6 ,AdamJPawson6 , Joanna L Sharman6 , Christopher Southan6 , Jamie A Davies6 and CGTP Collaborators 1School of Life Sciences, University of Nottingham Medical School, Nottingham, NG7 2UH, UK 2Medway School of Pharmacy, The Universities of Greenwich and Kent at Medway, Anson Building, Central Avenue, Chatham Maritime, Chatham, Kent, ME4 4TB, UK 3Neuroscience Division, Medical Education Institute, Ninewells Hospital and Medical School, University of Dundee, Dundee, DD1 9SY, UK 4Pharmacology and Toxicology, Institute of Pharmacy, University of Innsbruck, A-6020 Innsbruck, Austria 5School of Physiology, Pharmacology and Neuroscience, University of Bristol, Bristol, BS8 1TD, UK 6Centre for Discovery Brain Science, University of Edinburgh, Edinburgh, EH8 9XD, UK Abstract The Concise Guide to PHARMACOLOGY 2019/20 is the fourth in this series of biennial publications. The Concise Guide provides concise overviews of the key properties of nearly 1800 human drug targets with an emphasis on selective pharmacology (where available), plus links to the open access knowledgebase source of drug targets and their ligands (www.guidetopharmacology.org), which provides more detailed views of target and ligand properties. -

A Novel Role for TRPM8 in Visceral Afferent Function
PAINÒ 152 (2011) 1459–1468 www.elsevier.com/locate/pain Research papers A novel role for TRPM8 in visceral afferent function ⇑ Andrea M. Harrington a,b, , Patrick A. Hughes a,b, Christopher M. Martin a, Jing Yang a,b, Joel Castro a, Nicole J. Isaacs a, L. Ashley Blackshaw a,b,c, Stuart M. Brierley a,b,c a Nerve-Gut Research Laboratory, Department of Gastroenterology & Hepatology, Hanson Institute, Royal Adelaide Hospital, Adelaide, South Australia, Australia b Discipline of Medicine, University of Adelaide, Adelaide, South Australia, Australia c Discipline of Physiology, University of Adelaide, Adelaide, South Australia, Australia Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article. article info abstract Article history: Transient receptor potential ion channel melastatin subtype 8 (TRPM8) is activated by cold temperatures Received 15 July 2010 and cooling agents, such as menthol and icilin. Compounds containing peppermint are reported to reduce Received in revised form 24 November 2010 symptoms of bowel hypersensitivity; however, the underlying mechanisms of action are unclear. Here Accepted 14 January 2011 we determined the role of TRPM8 in colonic sensory pathways. Laser capture microdissection, quantita- tive reverse transcription-polymerase chain reaction (RT-PCR), immunofluorescence, and retrograde trac- ing were used to localise TRPM8 to colonic primary afferent neurons. In vitro extracellular single-fibre Keywords: afferent recordings were used to determine the effect of TRPM8 channel activation on the chemosensory Transient receptor potential ion channel and mechanosensory function of colonic high-threshold afferent fibres. TRPM8 mRNA was present in melastatin 8 Mechanosensation colonic DRG neurons, whereas TRPM8 protein was present on nerve fibres throughout the wall of the Chemosensation colon. -
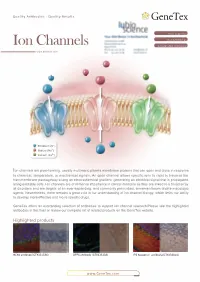
Ion Channels Accelerated D1scovery
Quality Antibodies · Quality Results $-GeneTex Your Expertise Our Antibod1es Ion Channels Accelerated D1scovery --------- www.genetex.com Potassium (K+ ) Sodium (Na+ ) Calcium (Ca2+ ) Ion channels are pore-forming, usually multimeric plasma membrane proteins that can open and close in response to chemical, temperature, or mechanical signals. An open channel allows specific ions to rapid ly traverse the transmembrane passageway a long an electrochemical gradient, generating an electrical signal that is propagated along excitable cells. Ion channels are of immense importance in clinical medicine as they are linked to a broad array of disorders and are targets of an ever-expanding, and commonly prescribed, armamentarium of pharmacologic agents. Nevertheless, there remains a great void in our understanding of ion channel biology, which limits our ability to develop more effective andmo re specific drugs. GeneTex offers an outstanding selection of antibodies to support ion channel research.Please see the highlighted antibodies in this flyer or review our complete list of related products on the GeneTex website. Highlighted products HCNl antibody (GTX131334} DPP6 antibody (GTX133338} IP3 Receptor I antibody (GTX133104} (...___ ___w _w _w_ ._G_e_n_e_T_e_x_._c_o_m_ _____,) ` a n a 匱 。 'ot I: . ·""·,, " . 鼴 丶•' · ,' , `> ,,, 丶 B -- ,:: ·-- o .i :' T. 、 冨 ,. \\.◄ .•}' r<• .. , -·•Jt.<_,,,, . ◄ · ◄ / .Jj -~~ 盆;i . '. ., CACNB4 (GTX100202) VGluTl antibody (GTX133148) P2X7一 antibody (GTX104288) Cavl.2 antibody (GTX54754) Cav2.1 antibody (GTX54753) -

Structure of the Acid-Sensing Ion Channel 1 in Complex with the Gating Modifi Er Psalmotoxin 1
ARTICLE Received 17 Jan 2012 | Accepted 18 May 2012 | Published 3 Jul 2012 DOI: 10.1038/ncomms1917 Structure of the Acid-sensing ion channel 1 in complex with the gating modifi er Psalmotoxin 1 Roger J.P. Dawson1 , J ö r g B e n z1 , Peter Stohler1 , Tim Tetaz 1 , Catherine Joseph1 , Sylwia Huber 1 , Georg Schmid 1 , Daniela H ü gin 1 , Pascal Pfl imlin 2 , Gerd Trube 2 , Markus G. Rudolph1 , Michael Hennig 1 & A r m i n R u f 1 Venom-derived peptide toxins can modify the gating characteristics of excitatory channels in neurons. How they bind and interfere with the fl ow of ions without directly blocking the ion permeation pathway remains elusive. Here we report the crystal structure of the trimeric chicken Acid-sensing ion channel 1 in complex with the highly selective gating modifi er Psalmotoxin 1 at 3.0 Å resolution. The structure reveals the molecular interactions of three toxin molecules binding at the proton-sensitive acidic pockets of Acid-sensing ion channel 1 and electron density consistent with a cation trapped in the central vestibule above the ion pathway. A hydrophobic patch and a basic cluster are the key structural elements of Psalmotoxin 1 binding, locking two separate regulatory regions in their relative, desensitized-like arrangement. Our results provide a general concept for gating modifi er toxin binding suggesting that both surface motifs are required to modify the gating characteristics of an ion channel. 1 F. Hoffmann-La Roche AG, pRED, Pharma Research & Early Development, Discovery Technologies , Grenzacherstrasse 124, Basel CH4070 , Switzerland . -
A Variant of ASIC2 Mediates Sodium Retention in Nephrotic Syndrome
A variant of ASIC2 mediates sodium retention in nephrotic syndrome Marc Fila, … , Gilles Crambert, Alain Doucet JCI Insight. 2021. https://doi.org/10.1172/jci.insight.148588. Research In-Press Preview Nephrology Graphical abstract Find the latest version: https://jci.me/148588/pdf 1 A variant of ASIC2 mediates sodium retention in nephrotic syndrome 1,2Marc Fila *, 1,2Ali Sassi*, 1,2Gaëlle Brideau, 1,2Lydie Cheval, 1,2 Luciana Morla, 1,2Pascal Houillier, 1,2Christine Walter, 1,2Michel Gennaoui, 1,2Laure Collignon L, 1,2Mathilde Keck, 1,2Gabrielle Planelles, 1,2Naziha Bakouh, 3Michel Peuchmaur, 4Georges Deschênes, 5Ignacio Anegon, 5Séverine Remy, 6Bruno Vogt, 1,2 Gilles Crambert#, 1,2Alain Doucet 1Centre de Recherche des Cordeliers, Sorbonne Universités, INSERM, Université de Paris, Laboratoire de Physiologie Rénale et Tubulopathies, F-75006, Paris, France 2CNRS, ERL8228, F-75006, Paris, France 3Cytology and Pathology DePartment, Robert Debré HosPital, F-75019, Paris, France 4Pediatric NePhrology DePartment, Robert Debré HosPital, F-75019, Paris, France 5INSERM UMR 1064, Centre de Recherches en TransPlantation et Immunologie (CRTI), Transgenic Rats ImmunoPhenomic facility, Nantes, France. 6DePartment of NePhrology and HyPertension, InselsPital, Bern University HosPital, CH- 3010, Bern, Switzerland Present address: Marc Fila, Pediatric NePhrology DePartment, HôPital Arnaud de Villeneuve Institut de Génomique Fonctionnelle UMR9023 CNRS U661 INSERM, MontPellier, France; Ali Sassi, DePartment of Cellular Physiology and Metabolism, University