Insulin Dysregulation in a Population of Finnhorses and Associated Phenotypic Markers of Obesity Justin R
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Horse and Burro Management at Sheldon National Wildlife Refuge
U.S. Fish & Wildlife Service Horse and Burro Management at Sheldon National Wildlife Refuge Environmental Assessment Before Horse Gather, August 2004 September 2002 After Horse Gather, August 2005 Front Cover: The left two photographs were taken one year apart at the same site, Big Spring Creek on Sheldon National Wildlife Refuge. The first photograph was taken in August 2004 at the time of a large horse gather on Big Spring Butte which resulted in the removal of 293 horses. These horses were placed in homes through adoption. The photograph shows the extensive damage to vegetation along the ripar- ian area caused by horses. The second photo was taken one-year later (August 2005) at the same posi- tion and angle, and shows the response of vegetation from reduced grazing pressure of horses. Woody vegetation and other responses of the ecosystem will take many years for restoration from the damage. An additional photograph on the right of the page was taken in September 2002 at Big Spring Creek. The tall vegetation was protected from grazing by the cage on the left side of the photograph. Stubble height of vegetation outside the cage was 4 cm, and 35 cm inside the cage (nearly 10 times the height). The intensity of horse grazing pressure was high until the gather in late 2004. Additional photo com- parisons are available from other riparian sites. Photo credit: FWS, David N. Johnson Department of Interior U.S. Fish and Wildlife Service revised, final Environmental Assessment for Horse and Burro Management at Sheldon National Wildlife Refuge April 2008 Prepared by: U.S. -

Digestão Total E Pré-Cecal Dos Nutrientes Em Potros Fistulados No Íleo
R. Bras. Zootec., v.27, n.2, p.331-337, 1998 Digestão Total e Pré-Cecal dos Nutrientes em Potros Fistulados no Íleo Ana Alix Mendes de Almeida Oliveira2, Augusto César de Queiroz3, Sebastião de Campos Valadares Filho3, Maria Ignez Leão3, Paulo Roberto Cecon4, José Carlos Pereira3 RESUMO - Seis potros machos, 1/2 sangue Bretão-Campolina, fistulados no íleo, foram alimentados à vontade com três rações: R1 - capim-elefante, R2 - capim-elefante + milho moído e R3 - capim-elefante + milho moído + farelo de soja, para: 1) estimar e comparar a digestibilidade aparente da matéria seca (MS), obtidas por intermédio do indicador óxido crômico e da coleta total de fezes; 2) avaliar a digestibilidade aparente pré-cecal e pós-ileal da MS, matéria orgânica (MO), proteína bruta (PB) e fibra em detergente neutro (FDN), para as três rações; e 3) calcular, por diferença, o valor energético e protéico do grão de milho moído e sua combinação com o farelo de soja para eqüinos. Análise descritiva foi feita para todos os valores observados. Os coeficientes de digestibilidade aparente, estimados com o óxido crômico para as três dietas, subestimaram os valores obtidos pela coleta total de fezes. Maiores valores de digestibilidade aparente para MO, PB e constituintes da parede celular foram encontrados, quando se adicionou farelo de soja ao capim-elefante e milho moído (R3). A digestibilidade aparente do extrato etéreo foi similar tanto para o milho moído (R2) quanto para o milho moído mais farelo de soja (R3). O capim-elefante teve baixos valores de digestibilidade aparente, pré-cecal e pós-ileal. A digestibilidade aparente pré-cecal da PB, na ração 2, foi inferior à da ração 3 e maior para MS. -

List of Horse Breeds 1 List of Horse Breeds
List of horse breeds 1 List of horse breeds This page is a list of horse and pony breeds, and also includes terms used to describe types of horse that are not breeds but are commonly mistaken for breeds. While there is no scientifically accepted definition of the term "breed,"[1] a breed is defined generally as having distinct true-breeding characteristics over a number of generations; its members may be called "purebred". In most cases, bloodlines of horse breeds are recorded with a breed registry. However, in horses, the concept is somewhat flexible, as open stud books are created for developing horse breeds that are not yet fully true-breeding. Registries also are considered the authority as to whether a given breed is listed as Light or saddle horse breeds a "horse" or a "pony". There are also a number of "color breed", sport horse, and gaited horse registries for horses with various phenotypes or other traits, which admit any animal fitting a given set of physical characteristics, even if there is little or no evidence of the trait being a true-breeding characteristic. Other recording entities or specialty organizations may recognize horses from multiple breeds, thus, for the purposes of this article, such animals are classified as a "type" rather than a "breed". The breeds and types listed here are those that already have a Wikipedia article. For a more extensive list, see the List of all horse breeds in DAD-IS. Heavy or draft horse breeds For additional information, see horse breed, horse breeding and the individual articles listed below. -

Development of a Welfare Index for Thoroughbred Racehorses
DEVELOPMENT OF A WELFARE INDEX FOR THOROUGHBRED RACEHORSES Alison Glen Mactaggart M. Qual. Psychology A thesis submitted for the degree of Doctor of Philosophy at The University of Queensland in 2015 School of Veterinary Science Abstract A uniform method capable of measuring animal welfare within the Thoroughbred Racing Industry (TBRI) does not exist. The aims of this study were to first investigate the importance of different welfare issues for Thoroughbred Racehorses (TBR) in Australia and then to incorporate them into a TBR welfare index (TRWI) that could be utilised in the industry. The second aim was assisted by the first, which utilised the expert opinion of stakeholders with in the TRWI, highlighting those aspects of husbandry requiring most improvement, and validated with behavioural measures. National and State Associations linked to racing were invited to send two delegates (experts) to a stakeholder meeting to determine key welfare issues, which they considered may have negative equine welfare implications. Following this a survey was created which posed vignettes of different combinations of welfare issues, which was subsequently presented to stakeholders around Australia. Fourteen key welfare issues were identified, each with two to four levels that were related to common husbandry practices. The 224 respondents identified the following welfare issues in declining order of importance: horsemanship > health and disease > education of the horse > track design and surface > ventilation > stabling > weaning > transport > nutrition > wastage > heat and humidity > whips > environment > gear. Further analysis of data tested the statistical significance of demographic factors, which determined that the respondents were relatively uniform in their answers. The TRWI which emerged from the responses could potentially be used to identify and improve welfare in training establishments. -
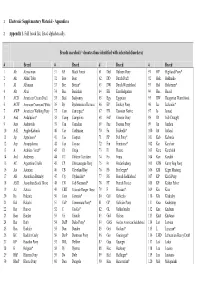
Electronic Supplementary Material - Appendices
1 Electronic Supplementary Material - Appendices 2 Appendix 1. Full breed list, listed alphabetically. Breeds searched (* denotes those identified with inherited disorders) # Breed # Breed # Breed # Breed 1 Ab Abyssinian 31 BF Black Forest 61 Dul Dülmen Pony 91 HP Highland Pony* 2 Ak Akhal Teke 32 Boe Boer 62 DD Dutch Draft 92 Hok Hokkaido 3 Al Albanian 33 Bre Breton* 63 DW Dutch Warmblood 93 Hol Holsteiner* 4 Alt Altai 34 Buc Buckskin 64 EB East Bulgarian 94 Huc Hucul 5 ACD American Cream Draft 35 Bud Budyonny 65 Egy Egyptian 95 HW Hungarian Warmblood 6 ACW American Creme and White 36 By Byelorussian Harness 66 EP Eriskay Pony 96 Ice Icelandic* 7 AWP American Walking Pony 37 Cam Camargue* 67 EN Estonian Native 97 Io Iomud 8 And Andalusian* 38 Camp Campolina 68 ExP Exmoor Pony 98 ID Irish Draught 9 Anv Andravida 39 Can Canadian 69 Fae Faeroes Pony 99 Jin Jinzhou 10 A-K Anglo-Kabarda 40 Car Carthusian 70 Fa Falabella* 100 Jut Jutland 11 Ap Appaloosa* 41 Cas Caspian 71 FP Fell Pony* 101 Kab Kabarda 12 Arp Araappaloosa 42 Cay Cayuse 72 Fin Finnhorse* 102 Kar Karabair 13 A Arabian / Arab* 43 Ch Cheju 73 Fl Fleuve 103 Kara Karabakh 14 Ard Ardennes 44 CC Chilean Corralero 74 Fo Fouta 104 Kaz Kazakh 15 AC Argentine Criollo 45 CP Chincoteague Pony 75 Fr Frederiksborg 105 KPB Kerry Bog Pony 16 Ast Asturian 46 CB Cleveland Bay 76 Fb Freiberger* 106 KM Kiger Mustang 17 AB Australian Brumby 47 Cly Clydesdale* 77 FS French Saddlebred 107 KP Kirdi Pony 18 ASH Australian Stock Horse 48 CN Cob Normand* 78 FT French Trotter 108 KF Kisber Felver 19 Az Azteca -

All Senior Questions Question Answer Source
All Senior Questions Question Answer Source Where in the digestive tract are amino acids Large intestine. HIH 710 synthesized? What unsoundness is most noticeable when Stringhalt. Ensminger, 530 backing the horse? Describe the ideal angles of the horse's front feet Front feet: 45 - 50 degrees. Beeman, 8 and hind feet. Rear feet: 50 - 60 degrees. An excessive reaction of the skin to sunlight is Photosensitivity. Veterinary Medicine, called what? 591 What term is used to describe a hoof wall angle Club foot. Curtis, 45 of 65 degrees or more? The American Paint Horse Association is devoted Paints, Quarter Horses, and Thoroughbreds. HBM strictly to stock horses and bases its registry on the blood of what 3 breeds? What do the letters CF stand for? Crude Fiber. Ensminger, 550 The common digital artery supplies blood to what Phalanges and foot. HBM parts of the horse? What is the interdental space? The gum space between the incisors and the HBM molars. Thursday, January 03, 1980 Page 1 of 95 University of Kentucky, College of Agriculture,Cooperative Extenison Service All Senior Questions Question Answer Source What color horses are more commonly prone to Gray horses. Veterinary Medicine, melanomas? 307 Most of the nutrients are found in what part of the Leaves. HBM forage plant? Excessive granulation tissue rising out of and Proud flesh. Ensminger, 527 above the edges of a wound is called what? Explain the functional difference of arteries and Arteries carry blood away from the heart to the Evans, Borton et all, veins in the horse's body. body tissues. -

THESIS DIETARY INTAKE in a GROUP of OLD MARES FED a SUPPLEMENT CONTAINING LONG CHAIN 18:3 (N-3) FATTY ACID and CHROMIUM Submitte
THESIS DIETARY INTAKE IN A GROUP OF OLD MARES FED A SUPPLEMENT CONTAINING LONG CHAIN 18:3 (N-3) FATTY ACID AND CHROMIUM Submitted by Silvia Otabachian-Smith Department of Animal Sciences In partial fulfillment of the requirements For the Degree of Master of Science Colorado State University Fort Collins, Colorado Spring 2012 Master’s Committee: Advisor: Tanja Hess Elaine Carnevale Terry Engle Gabriele Landolt Copyright by Silvia Otabachian-Smith 2012 All Rights Reserved ABSTRACT DIETARY INTAKE IN A GROUP OF OLD MARES FED A SUPPLEMENT CONTAINING LONG CHAIN 18:3 (N-3) FATTY ACID AND CHROMIUM Introduction: Differences in dietary maintenance requirements for old horses compared to adult horses is unknown (NRC, 2007). Older horses are prone to developing decreased insulin sensitivity due to an increase in inflammation, disease, fat accumulation, and a decrease in physical activity (Adams et al., 2009). Studies show a relationship between obesity, inflammation, and insulin resistance (IR) in horses (Vick et al., a,b). An increased inflammatory status in older horses may cause of pituitary pars intermedia dysfunction (PPID); which, predisposes horses to laminitis and insulin resistance (McFarlane & Holbrook, 2008). Polyunsaturated fatty acids (PUFA), such as n-3 α-linolenic acid (ALA), are absorbed and incorporated into cell membranes. In rat and human studies, PUFAs change fatty acid composition of phospholipids surrounding insulin receptors found in muscle (Luo et al., 1996; Rasic-Milutinovic et al., 2007) and reduce inflammation when incorporated into white blood cells (Calder, 2008). Chromium has been found to be beneficial in diabetic experimental animals and also in conditions resulting from insulin sensitivity and defects in glucose transportation (Liu et al., 2010). -

Discriminant Canonical Analysis of the Contribution of Spanish and Arabian Purebred Horses to the Genetic Diversity and Population Structure of Hispano-Arabian Horses
UC Davis UC Davis Previously Published Works Title Discriminant Canonical Analysis of the Contribution of Spanish and Arabian Purebred Horses to the Genetic Diversity and Population Structure of Hispano-Arabian Horses. Permalink https://escholarship.org/uc/item/8w77w522 Journal Animals : an open access journal from MDPI, 11(2) ISSN 2076-2615 Authors Marín Navas, Carmen Delgado Bermejo, Juan Vicente McLean, Amy Katherine et al. Publication Date 2021-01-21 DOI 10.3390/ani11020269 Peer reviewed eScholarship.org Powered by the California Digital Library University of California animals Article Discriminant Canonical Analysis of the Contribution of Spanish and Arabian Purebred Horses to the Genetic Diversity and Population Structure of Hispano-Arabian Horses Carmen Marín Navas 1 , Juan Vicente Delgado Bermejo 1 , Amy Katherine McLean 2 , José Manuel León Jurado 3, Antonio Rodriguez de la Borbolla y Ruiberriz de Torres 4 and Francisco Javier Navas González 1,* 1 Department of Genetics, Faculty of Veterinary Sciences, University of Córdoba, 14071 Córdoba, Spain; [email protected] (C.M.N.); [email protected] (J.V.D.B.) 2 Department of Animal Science, University of California Davis, Davis, CA 95617, USA; [email protected] 3 Centro Agropecuario Provincial de Córdoba, Diputación Provincial de Córdoba, 14071 Córdoba, Spain; [email protected] 4 Unión Española de Ganaderos de Pura Raza Hispano-Árabe, 41001 Sevilla, Spain; [email protected] * Correspondence: [email protected]; Tel.: +34-957-21-87-06 Simple Summary: The demographic and genetic diversity structure and the contributions of Spanish (PRE) and Arabian Purebred (PRá) horses to the process of conformation of the Hispano-Arabian Citation: Marín Navas, C.; Delgado (Há) horse breed were evaluated. -

Feeding Race Prospects and Racehorse in Training
E-533 12-02 eeding Race Prospects F Racehorses & inTraining P. G. Gibbs, G. D. Potter and B. D. Scott eeding F Race Prospects & Racehorses in Training P. G. Gibbs, G. D. Potter and B. D. Scott* n recent years, significant research attention be closely related to that horse’s fitness and diet. has been directed toward the equine athlete, If the horse has the available energy and the Iparticularly racehorses and young horses des- nutrients to use that energy, it can voluntarily run tined for the track. New information is becoming faster and perform at a higher level than horses available and new concepts are being formed with insufficient fuel and other nutrients to per- about the physiology and nutrition of racehorses. form these tasks. One reason for this attention is that over the To ensure that racehorses can perform at opti- past 50 years, the physical performance of race- mum levels, trainers need to pay close attention horses has improved very little. Although racing to nutrition, providing the appropriate amounts times over common distances have improved and forms of energy, protein, vitamins and miner- some, the magnitude of improvement has been als for young prospects as well as for racehorses relatively small compared to that of human ath- in training. If the nutritional requirements are met letes. This is in spite of efforts to breed horses accurately and feeding management is conducted with greater racing ability. Further, too many properly, racehorses’ performances will be horses continue to succumb to crippling injuries improved over those horses fed imbalanced diets brought on by acute fatigue and a compromised in irregular amounts at inappropriate times. -

1 Nature of Horse Breeds
1 Nature of Horse Breeds The horse captures our imagination because of its beauty, power and, most of all, its personality. Today, we encounter a wide array of horse breeds, developed for diverse purposes. Much of this diversity did not exist at the time of domestication of the horse, 5500 years ago (Chapter 2). Modern breeds were developed through genetic selection and based on the variety of uses of horses during the advance of civilization. Domestication of the horse revolutionized civilization. A rider could go farther and faster than people had ever gone before. Horses provided power to till more land and move heavier loads. Any sort of horse could provide these benefits, as long as it could be domesticated. However, over time people became more discern- ing about the characteristics of their horses. The intuitive and genetic principle that “like begat like” led people to choose the best horses as breeding stock. At the same time, people in different parts of the world used different criteria when select- ing horses. The horses were raised in different climates, fed different rations, exposed to different infectious diseases, and asked to do different types of work. Genetic differences could and did have a large impact on these traits. Over time, selection led to the creation of diverse types of horse around the world. We use a variety of terms to describe the genetic diversity among groups of animals, both to distinguish horses from other animals and examine differences among the different types of horses. Those terms include genus, species, popula- tion, landrace, and breed. -

Regional Hippology Contest – 2016 Written Exam NAME
Junior High Division (6th - 8th Grades) Regional Hippology Contest – 2016 Written Exam NAME: ________________________________________________________ COUNTY: _________________________ (Mark correct LETTER on answer sheet) Multiple Choice: 1. Name the only draft horse to originate and be recognized as a breed in the United States. A. Shire B. Clydesdale C. American Cream D. Belgian 2. What are the 12 front teeth in the horse’s mouth called? A. Incisors B. Molars C. Pre-molars D. Wolf teeth 3. Blister beetles can infect what type of forage? A. Timothy hay B. Alfalfa hay C. Bermuda grass hay D. Orchard grass hay 4. What part of the Western saddle is located directly behind the rider’s seat? A. Saddle flap B. Skirt C. Cantle D. Pommel 5. Fever, loss of appetite, and unwillingness to swallow are early signs or what disease? A. Equine Infectious Anemia B. Equine Strangles C. Equine Influenza D. Equine Tetanus 6. What external parasite lays eggs on the legs of horses? A. Horse fly B. Ascarids C. Deer fly D. Bot fly 7. In equine nutrition, what do the initials TDN indicate? A. Total Digestible Nitrogen B. Total Disposable Nutrition C. Total Degraded Nitrogen D. Total Digestible Nutrients 8. What part of the English bridle fits around the horse’s forehead, between the ears and eyes? A. Crown piece B. Brow band C. Cavesson D. Cheek piece 9. What is the main site for nutrient absorption in the horse? A. Small intestine B. Large intestine C. Cecum D. Stomach 10. What type of bit applies direct pressure to the horse’s mouth? A. -

Arabian Horses and the Cavalry
BLOODLINE © www.tuttoarabi.com RUSSIA ARABIAN HORSES AND THE CAVALRY Many Russian breeders in the 1700s and 1800s had the necessary expertise and huge resources to use the Arabian horse as a foundation for the creation of many breeds. In fact, the Arabian heralded in a new era for these astute breeders. They utilised these wonderful animals to increase the endurance, movement, working ability, soundness, and thriftiness, as well as beauty of their native stock. It was noted the Arabian had the amazing ability to transmit these outstanding characteristics onto their descendants for several generations. In fact, the Arabian had a phenomenal effect on Russia’s horse breeding activities. The Orlov Trotter (and Orlov Riding Horse) is one of Russia’s most famous breeds and was founded by the Arabian stallions Smetanka and Saltan I. The grey Smetanka, reputed to be a magnificent horse, was purchased by Count Alexis Orlov-Chesmenskii from a Turkish pasha in 1774. Smetanka travelled to Count Orlov’s Khrenovoya Stud in Russia with a military escort, the journey taking two years to complete. Smetanka only stood at stud in 1777 and was crossed with a Danish mare to start the foundation of the Orlov Trotter breed. The chestnut stallion Saltan I came to Russia from Arabia in 1775 and died in 1776, leaving three sons (confusingly named Saltan) and one daughter to carry on his influence as co-founder of the Orlov Trotter. Count Orlov also imported many other horses of Oriental blood to Artwork by Karen Hodges. his Khrenovoya Stud. The characteristics of the Orlov Trotter appeared to mirror in many respects the excellent features found in their Arabian ancestors.