Androgen-Induced Masculinization in Rainbow Trout Results
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role of LIM Kinase 1 and Its Substrates in Cell Cycle Progression
University of Central Florida STARS Electronic Theses and Dissertations, 2004-2019 2014 The Role of LIM Kinase 1 and its Substrates in Cell Cycle Progression Lisa Ritchey University of Central Florida Part of the Medical Sciences Commons Find similar works at: https://stars.library.ucf.edu/etd University of Central Florida Libraries http://library.ucf.edu This Doctoral Dissertation (Open Access) is brought to you for free and open access by STARS. It has been accepted for inclusion in Electronic Theses and Dissertations, 2004-2019 by an authorized administrator of STARS. For more information, please contact [email protected]. STARS Citation Ritchey, Lisa, "The Role of LIM Kinase 1 and its Substrates in Cell Cycle Progression" (2014). Electronic Theses and Dissertations, 2004-2019. 1300. https://stars.library.ucf.edu/etd/1300 THE ROLE OF LIM KINASE 1 AND ITS SUBSTRATES IN CELL CYCLE PROGRESSION by LISA RITCHEY B.S. Florida State University 2007 M.S. University of Central Florida 2010 A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Burnett School of Biomedical Sciences in the College of Graduate Studies at the University of Central Florida Orlando, Florida Summer Term 2014 Major Professor: Ratna Chakrabarti © 2014 Lisa Ritchey ii ABSTRACT LIM Kinase 1 (LIMK1), a modulator of actin and microtubule dynamics, has been shown to be involved in cell cycle progression. In this study we examine the role of LIMK1 in G1 phase and mitosis. We found ectopic expression of LIMK1 resulted in altered expression of p27Kip1, the G1 phase Cyclin D1/Cdk4 inhibitor. -

Dynamics of Meiotic Sex Chromosome Inactivation And
bioRxiv preprint doi: https://doi.org/10.1101/665372; this version posted June 10, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. 1 Dynamics of Meiotic Sex Chromosome Inactivation 2 and Pachytene Activation in Mice Spermatogenesis 3 4 Ábel Vértesy1,2; Javier Frias-Aldeguer1,4; Zeliha Sahin1,3; Nicolas Rivron1,4; Alexander van 5 Oudenaarden1,2 and Niels Geijsen1,5 6 7 1. Hubrecht Institute-KNAW (Royal Netherlands Academy of Arts and Sciences) and 8 University Medical Center, 3584 CT Utrecht, The Netherlands 9 2. Department of Genetics, Center for Molecular Medicine, Cancer Genomics Netherlands, 10 University Medical Center Utrecht, The Netherlands 11 3. Academic Medical Center, Amsterdam, The Netherlands 12 4. MERLN Institute for Technology-Inspired Regenerative Medicine, Maastricht University, 13 The Netherlands 14 5. Faculty of Veterinary Medicine, Clinical Sciences of Companion Animals, Utrecht 15 University, The Netherlands 16 17 Abstract 18 During germ cell development, cells undergo a drastic switch from mitosis to meiosis to 19 form haploid germ cells. Sequencing and computational technologies now allow studying 20 development at the single-cell level. Here we developed a multiplexed trajectory 21 reconstruction to create a high-resolution developmental map of spermatogonia and 22 prophase-I spermatocytes from testes of a Dazl-GFP reporter mouse. We identified three 23 main transitions in the meiotic prophase-I: meiotic entry, the meiotic sex chromosome 24 inactivation (MSCI), and concomitant pachytene activation. -

Analysis of Microarray Data on Gene Expression and Methylation to Identify Long Non-Coding Rnas in Non-Small Cell Lung Cancer
www.nature.com/scientificreports OPEN Analysis of Microarray Data on Gene Expression and Methylation to Identify Long Non-coding RNAs Received: 16 August 2016 Accepted: 26 October 2016 in Non-small Cell Lung Cancer Published: 16 November 2016 Nannan Feng1,*, Travers Ching2,*, Yu Wang3, Ben Liu3, Hongyan Lin1, Oumin Shi1, Xiaohong Zhang1, Min Zheng1, Xin Zheng1, Ming Gao3, Zhi-jie Zheng1, Herbert Yu2, Lana Garmire2,† & Biyun Qian1,† To identify what long non-coding RNAs (lncRNAs) are involved in non-small cell lung cancer (NSCLC), we analyzed microarray data on gene expression and methylation. Gene expression chip and HumanMethylation450BeadChip were used to interrogate genome-wide expression and methylation in tumor samples. Differential expression and methylation were analyzed through comparing tumors with adjacent non-tumor tissues. LncRNAs expressed differentially and correlated with coding genes and DNA methylation were validated in additional tumor samples using RT-qPCR and pyrosequencing. In vitro experiments were performed to evaluate lncRNA’s effects on tumor cells. We identified 8,500 lncRNAs expressed differentially between tumor and non-tumor tissues, of which 1,504 were correlated with mRNA expression. Two of the lncRNAs, LOC146880 and ENST00000439577, were positively correlated with expression of two cancer-related genes, KPNA2 and RCC2, respectively. High expression of LOC146880 and ENST00000439577 were also associated with poor survival. Analysis of lncRNA expression in relation to DNA methylation showed that LOC146880 expression was down-regulated by DNA methylation in its promoter. Lowering the expression of LOC146880 or ENST00000439577 in tumor cells could inhibit cell proliferation, invasion and migration. Analysis of microarray data on gene expression and methylation allows us to identify two lncRNAs, LOC146880 and ENST00000439577, which may promote the progression of NSCLC. -

RCC2 (NM 018715) Human Tagged ORF Clone Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for RC208811 RCC2 (NM_018715) Human Tagged ORF Clone Product data: Product Type: Expression Plasmids Product Name: RCC2 (NM_018715) Human Tagged ORF Clone Tag: Myc-DDK Symbol: RCC2 Synonyms: TD-60 Vector: pCMV6-Entry (PS100001) E. coli Selection: Kanamycin (25 ug/mL) Cell Selection: Neomycin This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 4 RCC2 (NM_018715) Human Tagged ORF Clone – RC208811 ORF Nucleotide >RC208811 representing NM_018715 Sequence: Red=Cloning site Blue=ORF Green=Tags(s) TTTTGTAATACGACTCACTATAGGGCGGCCGGGAATTCGTCGACTGGATCCGGTACCGAGGAGATCTGCC GCCGCGATCGCC ATGCCCAGGAAGAAGGCGGCGGCGGCGGCCTGGGAGGAGCCGAGCTCGGGCAACGGCACTGCCCGCGCCG GGCCCAGGAAACGCGGCGGCCCGGCGGGCAGGAAGCGCGAGCGGCCCGAGCGCTGCAGTAGCAGCAGCGG CGGCGGCAGCAGCGGCGACGAGGACGGCCTGGAGCTCGACGGGGCCCCCGGCGGGGGCAAGCGCGCGGCG CGGCCGGCGACAGCAGGCAAGGCGGGCGGCGCGGCCGTGGTCATCACCGAACCCGAGCACACCAAGGAGC GCGTCAAACTTGAAGGGTCAAAGTGCAAAGGGCAGCTTTTGATTTTTGGGGCAACCAACTGGGACTTGAT TGGTCGAAAAGAAGTGCCTAAACAGCAAGCTGCTTACCGCAATCTCGGTCAGAATTTGTGGGGGCCCCAC AGATATGGGTGCCTGGCGGGGGTCCGGGTGCGGACAGTGGTCTCGGGCTCGTGTGCTGCACACAGCCTCC TCATCACCACGGAAGGGAAGCTGTGGAGCTGGGGTCGAAATGAGAAGGGGCAGCTGGGACATGGTGACAC CAAGAGAGTAGAAGCCCCTAGACTCATCGAGGGTCTTAGCCACGAAGTGATTGTGTCTGCAGCATGTGGG -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -
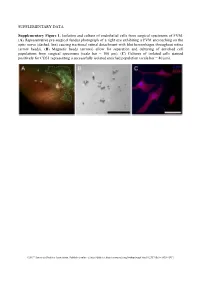
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

How Does SUMO Participate in Spindle Organization?
cells Review How Does SUMO Participate in Spindle Organization? Ariane Abrieu * and Dimitris Liakopoulos * CRBM, CNRS UMR5237, Université de Montpellier, 1919 route de Mende, 34090 Montpellier, France * Correspondence: [email protected] (A.A.); [email protected] (D.L.) Received: 5 July 2019; Accepted: 30 July 2019; Published: 31 July 2019 Abstract: The ubiquitin-like protein SUMO is a regulator involved in most cellular mechanisms. Recent studies have discovered new modes of function for this protein. Of particular interest is the ability of SUMO to organize proteins in larger assemblies, as well as the role of SUMO-dependent ubiquitylation in their disassembly. These mechanisms have been largely described in the context of DNA repair, transcriptional regulation, or signaling, while much less is known on how SUMO facilitates organization of microtubule-dependent processes during mitosis. Remarkably however, SUMO has been known for a long time to modify kinetochore proteins, while more recently, extensive proteomic screens have identified a large number of microtubule- and spindle-associated proteins that are SUMOylated. The aim of this review is to focus on the possible role of SUMOylation in organization of the spindle and kinetochore complexes. We summarize mitotic and microtubule/spindle-associated proteins that have been identified as SUMO conjugates and present examples regarding their regulation by SUMO. Moreover, we discuss the possible contribution of SUMOylation in organization of larger protein assemblies on the spindle, as well as the role of SUMO-targeted ubiquitylation in control of kinetochore assembly and function. Finally, we propose future directions regarding the study of SUMOylation in regulation of spindle organization and examine the potential of SUMO and SUMO-mediated degradation as target for antimitotic-based therapies. -

Mouse Rcc2 Conditional Knockout Project (CRISPR/Cas9)
https://www.alphaknockout.com Mouse Rcc2 Conditional Knockout Project (CRISPR/Cas9) Objective: To create a Rcc2 conditional knockout Mouse model (C57BL/6J) by CRISPR/Cas-mediated genome engineering. Strategy summary: The Rcc2 gene (NCBI Reference Sequence: NM_173867 ; Ensembl: ENSMUSG00000040945 ) is located on Mouse chromosome 4. 13 exons are identified, with the ATG start codon in exon 2 and the TGA stop codon in exon 13 (Transcript: ENSMUST00000038893). Exon 3 will be selected as conditional knockout region (cKO region). Deletion of this region should result in the loss of function of the Mouse Rcc2 gene. To engineer the targeting vector, homologous arms and cKO region will be generated by PCR using BAC clone RP23-275J10 as template. Cas9, gRNA and targeting vector will be co-injected into fertilized eggs for cKO Mouse production. The pups will be genotyped by PCR followed by sequencing analysis. Note: Exon 3 starts from about 17.95% of the coding region. The knockout of Exon 3 will result in frameshift of the gene. The size of intron 2 for 5'-loxP site insertion: 5515 bp, and the size of intron 3 for 3'-loxP site insertion: 2288 bp. The size of effective cKO region: ~594 bp. The cKO region does not have any other known gene. Page 1 of 7 https://www.alphaknockout.com Overview of the Targeting Strategy Wildtype allele gRNA region 5' gRNA region 3' 1 3 13 Targeting vector Targeted allele Constitutive KO allele (After Cre recombination) Legends Exon of mouse Rcc2 Homology arm cKO region loxP site Page 2 of 7 https://www.alphaknockout.com Overview of the Dot Plot Window size: 10 bp Forward Reverse Complement Sequence 12 Note: The sequence of homologous arms and cKO region is aligned with itself to determine if there are tandem repeats. -

2021.06.15.448470V1.Full.Pdf
bioRxiv preprint doi: https://doi.org/10.1101/2021.06.15.448470; this version posted June 15, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Systematic Chromosome Rearrangement Induced by CRISPR-Cas9 Reshapes the Genome and Transcriptome of Human Cells Ying Liu1#, Guangwei Ma1,2#, Zenghong Gao1, Jian Li1, Jin Wang1, Xiangping Zhu1, Jiawen Yang1,3, Yiting Zhou1, Kaishun, Hu1, Yin Zhang1*, Yabin Guo1* Affiliation: 1Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Medical Research Center, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou, 510120; 2Ministry of Education Key Laboratory for Ecology of Tropical Islands, Key Laboratory of Tropical Animal and Plant Ecology of Hainan Province, College of Life Sciences, Hainan Normal University, Haikou 571158; 3State Key Laboratory of Biocontrol, School of Life Sciences, Sun Yat-sen University, Guangzhou, China. #equal contributions *Correspondence: [email protected]; [email protected] Running title: Genome shuffle by CRISPR-Cas9 Highlights: Repetitive retroelements with large copy numbers were targeted using CRISPR-Cas9 in human cells. Cells survived after their chromosomal DNAs were heavily cleaved and systematic chromosome rearrangement was achieved. Systematic chromosome rearrangement reshaped the genome and transcriptome of the cells. bioRxiv preprint doi: https://doi.org/10.1101/2021.06.15.448470; this version posted June 15, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract Chromosome rearrangement plays important roles in development, carcinogenesis and evolution. -

Prediction Model for Aneuploidy in Early Human Embryo Development Revealed by Single-Cell Analysis
ARTICLE Received 26 Sep 2014 | Accepted 22 May 2015 | Published 7 Jul 2015 DOI: 10.1038/ncomms8601 OPEN Prediction model for aneuploidy in early human embryo development revealed by single-cell analysis Maria Vera-Rodriguez1,2,3, Shawn L. Chavez1,2,w, Carmen Rubio3,4, Renee A. Reijo Pera1,2,w & Carlos Simon2,3,4 Aneuploidies are prevalent in the human embryo and impair proper development, leading to cell cycle arrest. Recent advances in imaging and molecular and genetic analyses are postulated as promising strategies to unveil the mechanisms involved in aneuploidy generation. Here we combine time-lapse, complete chromosomal assessment and single-cell RT–qPCR to simultaneously obtain information from all cells that compose a human embryo until the approximately eight-cell stage (n ¼ 85). Our data indicate that the chromosomal status of aneuploid embryos (n ¼ 26), including those that are mosaic (n ¼ 3), correlates with significant differences in the duration of the first mitotic phase when compared with euploid embryos (n ¼ 28). Moreover, gene expression profiling suggests that a subset of genes is differentially expressed in aneuploid embryos during the first 30 h of development. Thus, we propose that the chromosomal fate of an embryo is likely determined as early as the pronuclear stage and may be predicted by a 12-gene transcriptomic signature. 1 Institute for Stem Cell Biology and Regenerative Medicine, Center for Reproductive and Stem Cell Biology, Stanford, California 94305, USA. 2 Department of Obstetrics and Gynecology, Institute for Stem Cell Biology and Regenerative Medicine, Center for Reproductive and Stem Cell Biology, Stanford, California 94305, USA. -

Exome Sequencing and Array-Based Comparative Genomic Hybridisation Analysis of Preferential 6-Methylmercaptopurine Producers
The Pharmacogenomics Journal (2015) 15, 414–421 © 2015 Macmillan Publishers Limited All rights reserved 1470-269X/15 www.nature.com/tpj ORIGINAL ARTICLE Exome sequencing and array-based comparative genomic hybridisation analysis of preferential 6-methylmercaptopurine producers EW Chua1,2, S Cree1, ML Barclay3,4, K Doudney5, K Lehnert6, A Aitchison1 and MA Kennedy1 Preferential conversion of azathioprine or 6-mercaptopurine into methylated metabolites is a major cause of thiopurine resistance. To seek potentially Mendelian causes of thiopurine hypermethylation, we recruited 12 individuals who exhibited extreme therapeutic resistance while taking azathioprine or 6-mercaptopurine and performed whole-exome sequencing (WES) and copy- number variant analysis by array-based comparative genomic hybridisation (aCGH). Exome-wide variant filtering highlighted four genes potentially associated with thiopurine metabolism (ENOSF1 and NFS1), transport (SLC17A4) or therapeutic action (RCC2). However, variants of each gene were found only in two or three patients, and it is unclear whether these genes could influence thiopurine hypermethylation. Analysis by aCGH did not identify any unusual or pathogenic copy-number variants. This suggests that if causative mutations for the hypermethylation phenotype exist they may be heterogeneous, occurring in several different genes, or they may lie within regulatory regions not captured by WES. Alternatively, hypermethylation may arise from the involvement of multiple genes with small effects. To test this hypothesis would require recruitment of large patient samples and application of genome-wide association studies. The Pharmacogenomics Journal (2015) 15, 414–421; doi:10.1038/tpj.2015.9; published online 10 March 2015 INTRODUCTION retrospective study (n = 1879), 2.5% of New Zealand individuals Azathioprine and 6-mercaptopurine structurally resemble endo- receiving thiopurine treatment were found to have a very high 8 genous purines, differing from the latter at the sixth carbon atom, 6-MMP/6-TGN ratio of 4100. -

RCC2 Is a Novel P53 Target in Suppressing Metastasis
OPEN Oncogene (2018) 37,8–17 www.nature.com/onc ORIGINAL ARTICLE RCC2 is a novel p53 target in suppressing metastasis C Song1, L Liang1, Y Jin, Y Li, Y Liu, L Guo, C Wu, C-H Yun and Y Yin RCC2 (also known as TD60) is a highly conserved protein involved in prognosis in colorectal cancer. However, its relationship with tumor development is less understood. Here we demonstrate a signaling pathway defining regulation of RCC2 and its functions in tumor progression. We report that p53 is a transcriptional regulator of RCC2 that acts through its binding to a palindromic motif in the RCC2 promoter. RCC2 physically interacts and deactivates a small GTPase Rac1 that is known to be involved in metastasis. We solved a high-resolution crystal structure of RCC2 and revealed one RCC1-like domain with a unique β-hairpin that is requisite for RCC2 interaction with Rac1. p53 or RCC2 deficiency leads to activation of Rac1 and deterioration of extracellular matrix sensing (haptotaxis) of surface-bound gradients. Ectopic expression of RCC2 restores directional migration in p53-null cells. Our results demonstrate that p53 and RCC2 signaling is important for regulation of cell migration and suppression of metastasis. We propose that the p53/RCC2/Rac1 axis is a potential target for cancer therapy. Oncogene (2018) 37, 8–17; doi:10.1038/onc.2017.306; published online 4 September 2017 INTRODUCTION outside of the area of mitotic control. However, the relation of Cell migration is fundamental to development and maintenance RCC2 to cancer development and the function of this molecule of organisms during embryonic development and wound healing.