Valley Fever (Coccidioidomycosis) Tutorial for Primary Care Professionals, Now in Its Second Printing
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Coccidioides Immitis
24/08/2017 FUNGAL AGENTS CAUSING INFECTION OF THE LUNG Microbiology Lectures of the Respiratory Diseases Prepared by: Rizalinda Sjahril Microbiology Department Faculty of Medicine Hasanuddin University 2016 OVERVIEW OF CLINICAL MYCOLOGY . Among 150.000 fungi species only 100-150 are human pathogens 25 spp most common pathogens . Majority are saprophyticLiving on dead or decayed organic matter . Transmission Person to person (rare) SPORE INHALATION OR ENTERS THE TISSUE FROM TRAUMA Animal to person (rare) – usually in dermatophytosis 1 24/08/2017 OVERVIEW OF CLINICAL MYCOLOGY . Human is usually resistant to infection, unless: Immunoscompromised (HIV, DM) Serious underlying disease Corticosteroid/antimetabolite treatment . Predisposing factors: Long term intravenous cannulation Complex surgical procedures Prolonged/excessive antibacterial therapy OVERVIEW OF CLINICAL MYCOLOGY . Several fungi can cause a variety of infections: clinical manifestation and severity varies. True pathogens -- have the ability to cause infection in otherwise healthy individuals 2 24/08/2017 Opportunistic/deep mycoses which affect the respiratory system are: Cryptococcosis Aspergillosis Zygomycosis True pathogens are: Blastomycosis Seldom severe Treatment not required unless extensive tissue Coccidioidomycosis destruction compromising respiratory status Histoplasmosis Or extrapulmonary fungal dissemination Paracoccidioidomycosis COMMON PATHOGENS OBTAINED FROM SPECIMENS OF PATIENTS WITH RESPIRATORY DISEASE Fungi Common site of Mode of Infectious Clinical -

Turning on Virulence: Mechanisms That Underpin the Morphologic Transition and Pathogenicity of Blastomyces
Virulence ISSN: 2150-5594 (Print) 2150-5608 (Online) Journal homepage: http://www.tandfonline.com/loi/kvir20 Turning on Virulence: Mechanisms that underpin the Morphologic Transition and Pathogenicity of Blastomyces Joseph A. McBride, Gregory M. Gauthier & Bruce S. Klein To cite this article: Joseph A. McBride, Gregory M. Gauthier & Bruce S. Klein (2018): Turning on Virulence: Mechanisms that underpin the Morphologic Transition and Pathogenicity of Blastomyces, Virulence, DOI: 10.1080/21505594.2018.1449506 To link to this article: https://doi.org/10.1080/21505594.2018.1449506 © 2018 The Author(s). Published by Informa UK Limited, trading as Taylor & Francis Group© Joseph A. McBride, Gregory M. Gauthier and Bruce S. Klein Accepted author version posted online: 13 Mar 2018. Submit your article to this journal Article views: 15 View related articles View Crossmark data Full Terms & Conditions of access and use can be found at http://www.tandfonline.com/action/journalInformation?journalCode=kvir20 Publisher: Taylor & Francis Journal: Virulence DOI: https://doi.org/10.1080/21505594.2018.1449506 Turning on Virulence: Mechanisms that underpin the Morphologic Transition and Pathogenicity of Blastomyces Joseph A. McBride, MDa,b,d, Gregory M. Gauthier, MDa,d, and Bruce S. Klein, MDa,b,c a Division of Infectious Disease, Department of Medicine, University of Wisconsin School of Medicine and Public Health, 600 Highland Avenue, Madison, WI 53792, USA; b Division of Infectious Disease, Department of Pediatrics, University of Wisconsin School of Medicine and Public Health, 1675 Highland Avenue, Madison, WI 53792, USA; c Department of Medical Microbiology and Immunology, University of Wisconsin School of Medicine and Public Health, 1550 Linden Drive, Madison, WI 53706, USA. -

HIV Infection and AIDS
G Maartens 12 HIV infection and AIDS Clinical examination in HIV disease 306 Prevention of opportunistic infections 323 Epidemiology 308 Preventing exposure 323 Global and regional epidemics 308 Chemoprophylaxis 323 Modes of transmission 308 Immunisation 324 Virology and immunology 309 Antiretroviral therapy 324 ART complications 325 Diagnosis and investigations 310 ART in special situations 326 Diagnosing HIV infection 310 Prevention of HIV 327 Viral load and CD4 counts 311 Clinical manifestations of HIV 311 Presenting problems in HIV infection 312 Lymphadenopathy 313 Weight loss 313 Fever 313 Mucocutaneous disease 314 Gastrointestinal disease 316 Hepatobiliary disease 317 Respiratory disease 318 Nervous system and eye disease 319 Rheumatological disease 321 Haematological abnormalities 322 Renal disease 322 Cardiac disease 322 HIV-related cancers 322 306 • HIV INFECTION AND AIDS Clinical examination in HIV disease 2 Oropharynx 34Neck Eyes Mucous membranes Lymph node enlargement Retina Tuberculosis Toxoplasmosis Lymphoma HIV retinopathy Kaposi’s sarcoma Progressive outer retinal Persistent generalised necrosis lymphadenopathy Parotidomegaly Oropharyngeal candidiasis Cytomegalovirus retinitis Cervical lymphadenopathy 3 Oral hairy leucoplakia 5 Central nervous system Herpes simplex Higher mental function Aphthous ulcers 4 HIV dementia Kaposi’s sarcoma Progressive multifocal leucoencephalopathy Teeth Focal signs 5 Toxoplasmosis Primary CNS lymphoma Neck stiffness Cryptococcal meningitis 2 Tuberculous meningitis Pneumococcal meningitis 6 -

List Item Posaconazole SP-H-C-611-II
European Medicines Agency London, 4 December 2006 Product Name: POSACONAZOLE SP Procedure number: EMEA/H/C/611/II/01 authorised SCIENTIFIC DISCUSSION longer no product Medicinal 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20) 74 18 84 00 Fax (44-20) 74 18 86 68 E-mail: [email protected] http://www.emea.europa.eu 1 Introduction Fungal infections are a major cause of morbidity and mortality in immunocompromised patients. Filamentous mould and yeast-like fungi are ubiquitous organisms found worldwide in many different media. The Candida species are the most common cause of fungal infections. However, epidemiologic shifts have begun to occur, most likely due to the prophylactic and empiric use of antifungal agents. Emerging fungal pathogens, such as Aspergillus, Fusarium, and Zygomycetes, are changing the clinical spectrum of fungal diagnoses. Pathogens General risk factors for invasive fungal infections are exposure to pathogens, an impaired immune system, and fungal spores. The presence of a colonised environment, partnered with a disruption in a physiologic barrier, potentiates the risk of an invasive fungal infection in an immunologically impaired host, such as a patient infected with HIV, someone taking chronic systemic steroids, or a transplant recipient. In addition, contaminated implanted devices (e.g., catheters, prostheses), external devices (e.g., contact lenses), and community reservoirs (e.g., hand lotion, pepper shakers) have all been implicated as sources of fungal outbreaks. Candida albicans continues to be the most frequent cause of invasive fungal infections in most patient populations. However, prophylaxis and the widespread use of antifungal agents as empiric therapy for neutropenic fever have led to a shift in the epidemiology of invasive Candida infections. -

Fungal Diseases
Abigail Zuger Fungal Diseases For creatures your size I offer a free choice of habitat, so settle yourselves in the zone that suits you best, in the pools of my pores or the tropical forests of arm-pit and crotch, in the deserts of my fore-arms, or the cool woods of my scalp Build colonies: I will supply adequate warmth and moisture, the sebum and lipids you need, on condition you never do me annoy with your presence, but behave as good guests should not rioting into acne or athlete's-foot or a boil. from "A New Year Greeting" by W.H. Auden. Introduction Most of the important contacts between human beings and the fungi occur outside medicine. Fungi give us beer, bread, antibiotics, mushroom omelets, mildew, and some devastating crop diseases; their ability to cause human disease is relatively small. Of approximately 100,000 known species of fungi, only a few hundred are human pathogens. Of these, only a handful are significant enough to be included in medical texts and introductory courses like this one. On the other hand, while fungal virulence for human beings is uncommon, the fungi are not casual pathogens. In the spectrum of infectious diseases, they can cause some of the most devastating and stubborn infections we see. Most human beings have a strong natural immunity to the fungi, but when this immunity is breached the consequences can be dramatic and severe. As modern medicine becomes increasingly adept in prolonging the survival of some patients with naturally-occurring immunocompromise (diabetes, cancer, AIDS), and causing iatrogenic immunocompromise in others (antibiotics, cytotoxic and MID 25 & 26 immunomodulating drugs), fungal infections are becoming increasingly important. -

Valley Fever a K a Coccidioidomycosis Coccidioidosis Coccidiodal Granuloma San Joaquin Valley Fever Desert Rheumatism Valley Bumps Cocci Cox C
2019 Lung Infection Symposium - Libke 10/26/2019 58 YO ♂ • 1974 PRESENTED WITH HEADACHE – DX = COCCI MENINGITIS WITH HYDROCEPHALUS – Rx = IV AMPHOTERICIN X 6 WKS – VP SHUNT – INTRACISTERNAL AMPHO B X 2.5 YRS (>200 PUNCTURES) • 1978 – 2011 VP SHUNT REVISIONS X 5 • 1974 – 2019 GAINFULLY EMPLOYED, RAISED FAMILY, RETIRED AND CALLS OCCASIONALLY TO SEE HOW I’M DOING. VALLEY FEVER A K A COCCIDIOIDOMYCOSIS COCCIDIOIDOSIS COCCIDIODAL GRANULOMA SAN JOAQUIN VALLEY FEVER DESERT RHEUMATISM VALLEY BUMPS COCCI COX C 1 2019 Lung Infection Symposium - Libke 10/26/2019 COCCIDIOIDOMYCOSIS • DISEASE FIRST DESCRIBED IN 1892 – POSADAS –ARGENTINA – RIXFORD & GILCHRIST - CALIFORNIA – INITIALLY THOUGHT PARASITE – RESEMBLED COCCIDIA “COCCIDIOIDES” – “IMMITIS” = NOT MINOR COCCIDIOIDOMYCOSIS • 1900 ORGANISM IDENTIFIED AS FUNGUS – OPHULS AND MOFFITT – ORGANISM CULTURED FROM TISSUES OF PATIENT – LIFE CYCLE DEFINED – FULFULLED KOCH’S POSTULATES 2 2019 Lung Infection Symposium - Libke 10/26/2019 COCCIDIOIDOMYCOSIS • 1932 ORGANISM IN SOIL SAMPLE FROM DELANO – UNDER BUNKHOUSE OF 4 PATIENTS – DISEASE FATAL • 1937 DICKSON & GIFFORD CONNECTED “VALLEY FEVER” TO C. IMMITIS – USUALLY SELF LIMITED – FREQUENTLY SEEN IN SAN JOAQUIN VALLEY – RESPIRATORY TRACT THE PORTAL OF ENTRY The usual cause for coccidioidomycosis in Arizona is C. immitis A. True B. False 3 2019 Lung Infection Symposium - Libke 10/26/2019 COCCIDIOIDAL SPECIES • COCCIDIOIDES IMMITIS – CALIFORNIA • COCCIDIOIDES POSADASII – NON-CALIFORNIA • ARIZONA, MEXICO • OVERLAP IN SAN DIEGO AREA THE MICROBIAL WORLD • PRIONS -

Fungal Serology Update – Test Algorithms and Methodologies
LABSTRACT – October 2019 Fungal Serology Update – Test algorithms and methodologies Audience Health care providers and laboratories involved in submission of specimens for fungal serology testing. Overview Beginning October 2019, the fungal serology testing algorithms and methodologies for Aspergillus, Histoplasma capsulatum, Blastomyces dermatitidis and Coccidiodies immitis used at Public Health Ontario have changed. All assays are now Health Canada licensed. Complement fixation (CF) assays have been discontinued. Summary of fungal serology assay methods now in use at PHO Laboratory Serologic Test Method used at PHO Analyte Detected Ordered Aspergillus Enzyme Immunoassay (EIA) IgG antibodies Histoplasma Immunodiffusion M and H band precipitins Blastomyces Immunodiffusion A band precipitins IgM (against TP antigens) and Screen, EIA IgG (against CF antigens) Coccidioides IDTP and IDCF band Confirmation, Immunodiffusion precipitins Page 1 of 4 Fungal Serology Update – Test algorithms and methodologies LAB-SD-134-000 Background information Please note, culture remains the gold standard for the diagnosis of most invasive fungal infections. However, fungal infections can be challenging to diagnose as symptoms are often non-specific and can mimic bacterial, viral, other fungal infections or malignancy. Results of fungal serological tests can be used to aid in the diagnosis and/or management of specific fungal infections and fungal allergies. Aspergillus species are ubiquitous environmental moulds which people inhale each day. While invasive disease is typically restricted to immunocompromised individuals, immunocompetent individuals can have certain allergic manifestations, or develop chronic pulmonary aspergillosis (CPA). Histoplasma capsulatum, Blastomyces dermatitidis and Coccidioides immitis are endemic, dimorphic fungi which can infect otherwise healthy individuals. Most infected individuals are exposed by inhaling fungal spores from the environment and present with initial clinical symptoms of respiratory illness. -

Fungal Group Fungal Disease Source Guidelines
Fungal Fungal disease Source Guidelines Relevant articles Group ESCMID guideline for the diagnosis and management of Candida diseases 2012: 1. Developing European guidelines in clinical microbiology and infectious diseases 2. Diagnostic procedures 3. Non-neutropenic adult patients 4. Prevention and management of Candida diseases ESCMID invasive infections in neonates and children caused by Candida spp 5. Adults with haematological malignancies and after haematopoietic stem cell transplantation (HCT) 6. Patients with HIV infection or AIDS Candidaemia and IDSA clinical practice guidelines 2016 IDSA invasive candidiasis ISPD ISPD guidelines/recommendations Candida peritonitis Special article: reducing the risks of peritoneal dialysis-related infections Invasive IDSA IDSA clinical practice guidelines 2010 WHO management guidelines WHO Cryptococcal meningitis Guidelines for the prevention and AIDSinfo treatment of opportunistic infections in HIV-infected Adults and adolescents Southern Guideline for the prevention, African diagnosis and management of HIV cryptococcal meningitis among HIV- clinicians infection persons: 2013 update society IDSA Clinical practice guidelines 2007 IDSA Histoplasmosis disseminated Guidelines for the prevention and treatment of opportunistic infections in AIDSinfo HIV-infected Adults and adolescents IDSA Clinical practice guidelines 2007 Histoplasmosis IDSA acute pulmonary AIDSinfo Guidelines for the prevention and treatment of opportunistic infections in HIV-infected Adults and adolescents Invasive IDSA Clinical -
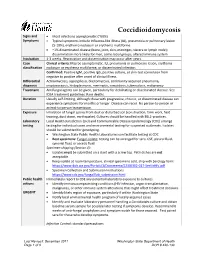
Coccidioidomycosis Reporting and Investigation Guideline
Coccidioidomycosis Signs and • Most infections asymptomatic (~60%) Symptoms • Typical symptoms include influenza-like illness (ILI), pneumonia or pulmonary lesion (5-10%), erythema nodosum or erythema multiforme • ~1% disseminated disease (bone, joint, skin, meninges, viscera or lymph node); dissemination more likely for men, some racial groups, altered immune system Incubation 1-3 weeks. Reactivation and dissemination may occur after years. Case Clinical criteria: May be asymptomatic. ILI, pneumonia or pulmonary lesion, erythema classification nodosum or erythema multiforme, or disseminated infection. Confirmed: Positive IgM, positive IgG, positive culture, or skin-test conversion from negative to positive after onset of clinical illness Differential Actinomycosis, aspergillosis, blastomycosis, community acquired pneumonia, diagnosis cryptococcosis, histoplasmosis, meningitis, sarcoidosis, tuberculosis, malignancy Treatment Antifungal agents can be given, particularly for debilitating or disseminated disease. See IDSA treatment guidelines. Rare deaths. Duration Usually self-limiting, although those with progressive, chronic, or disseminated disease can experience symptoms for months or longer. Disease can recur. No person-to-person or animal-to-person transmission. Exposure Inhalation of fungal spores from dust or disturbed soil (construction, farm work, field training, dust storm, earthquake). Cultures should be handled with BSL2-practices. Laboratory Local Health Jurisdiction (LHJ) and Communicable Disease Epidemiology (CDE) arrange testing testing for individual cases and environmental testing for suspected outbreaks. Isolates should be submitted for genotyping. • Washington State Public Health Laboratories can facilitate testing at CDC • Best specimens: Fungal isolate; testing can be arranged for sera, CSF, pleural fluid, synovial fluid, or ascetic fluid Specimen shipping (Section 4): • Isolates must be submitted on a slant with a screw top. Petri dishes are not acceptable. -

Disseminated Infection Due to Chrysosporium Zonatum in A
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 1999, p. 18–25 Vol. 37, No. 1 0095-1137/99/$04.0010 Copyright © 1999, American Society for Microbiology. All Rights Reserved. Disseminated Infection Due to Chrysosporium zonatum in a Patient with Chronic Granulomatous Disease and Review of Non-Aspergillus Fungal Infections in Patients with This Disease EMMANUEL ROILIDES,1* LYNNE SIGLER,2 EVANGELIA BIBASHI,3 HELEN KATSIFA,1 4 1 NICOLAS FLARIS, AND CHRISTOS PANTELIADIS Third Department of Pediatrics, Aristotle University of Thessaloniki,1 and Microbiology3 and Pathology Departments,4 Hippokration Hospital, Thessaloniki, Greece, and Microfungus Collection and Herbarium, Devonian Botanic Garden, University of Alberta, Edmonton, Alberta, Canada2 Received 15 June 1998/Returned for modification 1 August 1998/Accepted 6 October 1998 We report the first case of Chrysosporium zonatum infection in a 15-year-old male with chronic granuloma- tous disease who developed a lobar pneumonia and tibia osteomyelitis while on prophylaxis with gamma inter- feron. The fungus was isolated from sputum and affected bone, and hyphae were observed in the bone by his- topathology. Therapy with amphotericin B eradicated the osteomyelitis and pneumonia, but pneumonia recurred in association with pericarditis and pleuritis during therapy with itraconazole. These manifestations subsided, and no recurrences occurred with liposomal amphotericin B therapy. Infections caused by Chrysosporium spe- cies are very rare, and C. zonatum has not previously been reported to cause mycosis in humans. This species, the anamorph of the heterothallic ascomycete Uncinocarpus orissi (family Onygenaceae), is distinguished by its thermotolerance, by colonies which darken from yellowish white to buff, and by club-shaped terminal aleurio- conidia borne at the ends of short, typically curved stalks. -

Coccidioidomycosis in New York State
Synopses Coccidioidomycosis in New York State Vishnu Chaturvedi,* Rama Ramani,* Sally Gromadzki,* Birgit Rodeghier,* Hwa-Gan Chang,† and Dale L. Morse*† New York State Department of Health, Albany, New York, USA; and †School of Public Health, University at Albany, SUNY, Albany, New York, USA Coccidioidomycosis, a systemic fungal disease caused by Coccidioides immitis, is endemic in the southwestern United States and in parts of Mexico and Central and South America. Only sporadic cases have been reported in areas (including New York) where the disease is not endemic. We used hospital discharge records and state mycology laboratory data to investigate the characteristics of C. immitis infections among New York State residents. From 1992 to 1997, 161 persons had hospital discharge diagnoses of coccidioidomycosis (ICD9 Code 114.0 - 114.5, 114.9). From 1989 to 1997, 49 cultures from patients were confirmed as C. immitis; 26 of these patients had traveled to disease-endemic areas. Fourteen of 16 isolates had multilocus genotypes similar to those of Arizona isolates, which corroborates the travel-related acquisition of the disease. Our results indicate that coccidioidomycosis may be more common in New York residents than previously recognized. Increased awareness among health-care providers should improve timely diagnosis of coccidioidomycosis and prevention of associated illnesses and deaths among patients in nondisease- endemic areas. Coccidioidomycosis, a systemic disease which fragment into endospores. When released caused by the dimorphic fungus Coccidioides from the spherule, each endospore can act as a immitis, is endemic in the southwestern United new infectious unit in vivo (1). C. immitis, one of States and parts of Mexico and in Central and the most virulent and infectious fungal South America (1,2). -

Black Fungus: a New Threat Uddin KN
Editorial (BIRDEM Med J 2021; 11(3): 164-165) Black fungus: a new threat Uddin KN Fungal infections, also known as mycoses, are Candida spp. including non-albicans Candida (causing traditionally divided into superficial, subcutaneous and candidiasis), p. Aspergillus spp. (causing aspergillosis), systemic mycoses. Cryptococcus (causing cryptococcosis), Mucormycosis previously called zygomycosis caused by Zygomycetes. What are systemic mycoses? These fungi are found in or on normal skin, decaying Systemic mycoses are fungal infections affecting vegetable matter and bird droppings respectively but internal organs. In the right circumstances, the fungi not exclusively. They are present throughout the world. enter the body via the lungs, through the gut, paranasal sinuses or skin. The fungi can then spread via the Who are at risk of systemic mycoses? bloodstream to multiple organs, often causing multiple Immunocompromised people are at risk of systemic organs to fail and eventually, result in the death of the mycoses. Immunodeficiency can result from: human patient. immunodeficiency virus (HIV) infection, systemic malignancy (cancer), neutropenia, organ transplant What causes systemic mycoses? recipients including haematological stem cell transplant, Patients who are immunocompromised are predisposed after a major surgical operation, poorly controlled to systemic mycoses but systemic mycosis can develop diabetes mellitus, adult-onset immunodeficiency in otherwise healthy patients. Systemic mycoses can syndrome, very old or very young. be split between two main varieties, endemic respiratory infections and opportunistic infections. What are the clinical features of systemic mycoses? The clinical features of a systemic mycosis depend on Endemic respiratory infections the specific infection and which organs have been Fungi that can cause systemic infection in people with affected.