Podoplanin: Emerging Functions in Development, the Immune System, and Cancer
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Podoplanin Regulates Mammary Stem Cell Function and Tumorigenesis by Potentiating Wnt/Β-Catenin Signaling Laura Bresson1,2,3, Marisa M
© 2018. Published by The Company of Biologists Ltd | Development (2018) 145, dev160382. doi:10.1242/dev.160382 STEM CELLS AND REGENERATION RESEARCH ARTICLE Podoplanin regulates mammary stem cell function and tumorigenesis by potentiating Wnt/β-catenin signaling Laura Bresson1,2,3, Marisa M. Faraldo1,4, Amandine Di-Cicco1, Miguel Quintanilla5, Marina A. Glukhova1,4 and Marie-Ange Deugnier1,4,* ABSTRACT K5/14), P-cadherin, smooth muscle-specific contractile proteins, Δ Stem cells (SCs) drive mammary development, giving rise postnatally and the transcription factors Np63 (an isoform of Trp63) and Slug/ to an epithelial bilayer composed of luminal and basal myoepithelial Snail2, which are essential for the maintenance of basal cell identity cells. Dysregulation of SCs is thought to be at the origin of certain breast (Yalcin-Ozuysal et al., 2010; Guo et al., 2012). The luminal cell cancers; however, the molecular identity of SCs and the factors layer is characterized by the expression of K8/18. It includes a regulating their function remain poorly defined. We identified the subset of hormone-sensing cells that express estrogen, progesterone transmembrane protein podoplanin (Pdpn) as a specific marker of the and prolactin receptors (ER, PR and PrlR, respectively) and produce basal compartment, including multipotent SCs, and found Pdpn local mediators involved in the paracrine control of basal and localized at the basal-luminal interface. Embryonic deletion of Pdpn luminal cell function (Brisken and Ataca, 2015). targeted to basal cells diminished basal and luminal SC activity and It is established that both mammary lineages, basal and luminal, affected the expression of several Wnt/β-catenin signaling components originate from a common embryonic stem cell (SC) expressing basal in basal cells. -

Multivariate Analysis Reveals Differentially Expressed Genes
www.nature.com/scientificreports OPEN Multivariate analysis reveals diferentially expressed genes among distinct subtypes of difuse astrocytic gliomas: diagnostic implications Nerea González‑García1,2, Ana Belén Nieto‑Librero1,2, Ana Luisa Vital3, Herminio José Tao4, María González‑Tablas2,5,6, Álvaro Otero2, Purifcación Galindo‑Villardón1,2, Alberto Orfao2,5,6 & María Dolores Tabernero2,5,6,7* Diagnosis and classifcation of gliomas mostly relies on histopathology and a few genetic markers. Here we interrogated microarray gene expression profles (GEP) of 268 difuse astrocytic gliomas—33 difuse astrocytomas (DA), 52 anaplastic astrocytomas (AA) and 183 primary glioblastoma (GBM)—based on multivariate analysis, to identify discriminatory GEP that might support precise histopathological tumor stratifcation, particularly among inconclusive cases with II–III grade diagnosed, which have diferent prognosis and treatment strategies. Microarrays based GEP was analyzed on 155 difuse astrocytic gliomas (discovery cohort) and validated in another 113 tumors (validation set) via sequential univariate analysis (pairwise comparison) for discriminatory gene selection, followed by nonnegative matrix factorization and canonical biplot for identifcation of discriminatory GEP among the distinct histological tumor subtypes. GEP data analysis identifed a set of 27 genes capable of diferentiating among distinct subtypes of gliomas that might support current histological classifcation. DA + AA showed similar molecular profles with only a few discriminatory genes -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Transcriptional Control of Lung Alveolar Type 1 Cell Development and Maintenance by NK Homeobox 2-1
Transcriptional control of lung alveolar type 1 cell development and maintenance by NK homeobox 2-1 Danielle R. Littlea,b, Kamryn N. Gerner-Mauroa, Per Flodbyc, Edward D. Crandallc, Zea Borokc, Haruhiko Akiyamad, Shioko Kimurae, Edwin J. Ostrina,f, and Jichao Chena,1 aDepartment of Pulmonary Medicine, The University of Texas MD Anderson Cancer Center, Houston, TX 77030; bUniversity of Texas Health Graduate School of Biomedical Sciences, The University of Texas MD Anderson Cancer Center, Houston, TX 77030; cDivision of Pulmonary, Critical Care and Sleep Medicine, Department of Medicine and Hastings Center for Pulmonary Research, Keck School of Medicine, University of Southern California, Los Angeles, CA 90033; dDepartment of Orthopedics, Kyoto University, Sakyo, 606-8507 Kyoto, Japan; eLaboratory of Metabolism, Center for Cancer Research, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892; and fDepartment of General Internal Medicine, The University of Texas MD Anderson Cancer Center, Houston, TX 77030 Edited by Clifford J. Tabin, Harvard Medical School, Boston, MA, and approved August 30, 2019 (received for review April 18, 2019) The extraordinarily thin alveolar type 1 (AT1) cell constitutes nearly Hippo signaling promotes progenitor differentiation toward the the entire gas exchange surface and allows passive diffusion of AT1 cell fate (12–14). This growing list of AT1 cell regulators oxygen into the blood stream. Despite such an essential role, the highlights both the underlying complexity and the necessity to transcriptional network controlling AT1 cells remains unclear. Using distinguish direct effects on AT1 cells versus those on progenitors, cell-specific knockout mouse models, genomic profiling, and 3D imag- AT2 cells, or tissue morphology, especially in light of the classical Nkx2-1 ing, we found that NK homeobox 2-1 ( )isexpressedinAT1 observation of rapid AT1 cell-like differentiation of cultured cells and is required for the development and maintenance of AT1 AT2 cells (15). -
Supplementary Figures and Tables
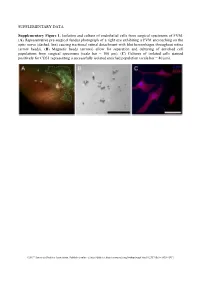
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Transcriptomic Profiles of High and Low Antibody Responders to Smallpox
Genes and Immunity (2013) 14, 277–285 & 2013 Macmillan Publishers Limited All rights reserved 1466-4879/13 www.nature.com/gene ORIGINAL ARTICLE Transcriptomic profiles of high and low antibody responders to smallpox vaccine RB Kennedy1,2, AL Oberg1,3, IG Ovsyannikova1,2, IH Haralambieva1,2, D Grill1,3 and GA Poland1,2 Despite its eradication over 30 years ago, smallpox (as well as other orthopox viruses) remains a pathogen of interest both in terms of biodefense and for its use as a vector for vaccines and immunotherapies. Here we describe the application of mRNA-Seq transcriptome profiling to understanding immune responses in smallpox vaccine recipients. Contrary to other studies examining gene expression in virally infected cell lines, we utilized a mixed population of peripheral blood mononuclear cells in order to capture the essential intercellular interactions that occur in vivo, and would otherwise be lost, using single cell lines or isolated primary cell subsets. In this mixed cell population we were able to detect expression of all annotated vaccinia genes. On the host side, a number of genes encoding cytokines, chemokines, complement factors and intracellular signaling molecules were downregulated upon viral infection, whereas genes encoding histone proteins and the interferon response were upregulated. We also identified a small number of genes that exhibited significantly different expression profiles in subjects with robust humoral immunity compared with those with weaker humoral responses. Our results provide evidence that differential gene regulation patterns may be at work in individuals with robust humoral immunity compared with those with weaker humoral immune responses. Genes and Immunity (2013) 14, 277–285; doi:10.1038/gene.2013.14; published online 18 April 2013 Keywords: Next-generation sequencing; mRNA-Seq; vaccinia virus; smallpox vaccine INTRODUCTION these 44 subjects had two samples (uninfected and vaccinia Vaccinia virus (VACV) is the immunologically cross-protective infected). -

Pdpn Podoplanin
1 Running title: Podoplanin and EMT NEW INSIGHTS INTO THE ROLE OF PODOPLANIN IN THE EPITHELIAL TO MESENCHYMAL TRANSITION Jaime Renart*, Patricia Carrasco-Ramírez, Beatriz Fernández-Muñoz1, Ester Martín- Villar, Lucía Montero, María M. Yurrita and Miguel Quintanilla. Instituto de Investigaciones Biomédicas Alberto Sols, CSIC-UAM *Corresponding author at: Instituto de Investigaciones Biomédicas Alberto Sols, CSIC- UAM. Arturo Duperier 4. 28029-Madrid. Spain. Tel: +34915854439. Fax: +34915854401. E-mail address: [email protected] 1Present address: Laboratorio Andaluz de Reprogramación Celular, Parque Tecnológico y Científico Cartuja 93. 41092-Sevilla, Spain. 2 CONTENTS 1. Introduction 2. Podoplanin 2.1. Protein structure 2.1.1. The ectodomain 2.1.2. The transmembrane domain 2.1.3. The cytoplasmic domain 2.2. Gene structure 2.3. Expression of podoplanin 2.3.1. Tissue distribution 2.3.2. Transcriptional regulation 2.3.3. Post-translational regulation 2.3.3.1. miRNAs 2.3.3.2. Glycosylation 2.3.3.3. Proteolytic processing 2.3.3.4. Phosphorylation 2.4. Podoplanin partners 2.4.1. CLEC-2 2.4.2. Tetraspanin CD9 2.4.3. Galectin 8 2.4.4. Heat shock protein A9 2.4.5. CD44 2.4.6. ERM proteins 3 2.4.7. Others 2.5. Signaling and molecular mechanisms 2.6. Podoplanin in development 2.7. Podoplanin in cancer 2.7.1. Expression in tumors 2.7.2. Role in migration, invasion and progression 2.7.3. Presence in tumor stroma 3. Epithelial to mesenchymal transition 3.1. Molecular mechanisms of EMT 3.1.1. Properties of the core Transcription factors 3.1.2. -

Mouse Embryonic Fibroblasts Exhibit Extensive Developmental and Phenotypic Diversity
Mouse embryonic fibroblasts exhibit extensive developmental and phenotypic diversity Prabhat K. Singhala,b, Slim Sassia,c, Lan Lana,b, Patrick Aud, Stefan C. Halvorsena, Dai Fukumurad, Rakesh K. Jaind,1, and Brian Seeda,b,1 aCenter for Computational and Integrative Biology, Massachusetts General Hospital, Boston, MA 02114; bDepartment of Genetics, Harvard Medical School, Boston, MA 02115; cDepartment of Orthopedics, Harvard Medical School, Boston, MA 02115; and dSteele Laboratory of Tumor Biology, Department of Radiation Oncology, Massachusetts General Hospital and Harvard Medical School, Boston, MA 02114 Contributed by Rakesh K. Jain, November 16, 2015 (sent for review August 22, 2015; reviewed by Patricia A. D’Amore, Massimo Pinzani, and David Tuveson) Analysis of embryonic fibroblasts from GFP reporter mice indicates that light or electron microscopy, but exhibit variable GFP expression. the fibroblast cell type harbors a large collection of developmentally Populations from earlier stages, days E12.5 and E14.5, exhibit lower and phenotypically heterogeneous subtypes. Some of these cells frequencies of GFP-positive cells compared with cultures prepared exhibit multipotency, whereas others do not. Multiparameter flow from day E16.5 and E18.5 embryos (Fig. 1A). GFP-positive cells cytometry analysis shows that a large number of distinct populations typically replicate more rapidly than GFP-negative cells under stan- of fibroblast-like cells can be found in cultures initiated from different dard culture conditions, leading to an increase in their prevalence embryonic organs, and cells sorted according to their surface pheno- with increasing culture passage (Fig. 1B). type typically retain their characteristics on continued propagation in Following fluorescence-activated cell sorting of newly initiated culture. -

Molecular Taxonomy of Human Ocular Outflow Tissues Defined by Single-Cell Transcriptomics
Molecular taxonomy of human ocular outflow tissues defined by single-cell transcriptomics Gaurang Patela,1, Wen Furya,1, Hua Yanga, Maria Gomez-Caraballob, Yu Baia, Tao Yanga, Christina Adlera, Yi Weia, Min Nia, Heather Schmittb, Ying Hua, George Yancopoulosa, W. Daniel Stamerb,2, and Carmelo Romanoa,2 aRegeneron Pharmaceuticals, Inc., Tarrytown, NY 10591; and bDepartment of Ophthalmology, Duke University, Durham, NC 27710 Edited by Joel S. Schuman, New York University, New York, NY, and accepted by Editorial Board Member Jeremy Nathans April 16, 2020 (received for review January 31, 2020) The conventional outflow pathway is a complex tissue responsible refs. 7 and 8). Cells that populate the TM are all of neural crest in for maintaining intraocular pressure (IOP) homeostasis. The co- origin (13), displaying different morphologies depending upon their ordinated effort of multiple cells with differing responsibilities tissue location. Thus, TM cells in uveal/corneoscleral meshwork ensures healthy outflow function and IOP maintenance. Dysfunc- display endothelial and macrophage properties, maintaining patent tion of one or more resident cell types results in ocular hyperten- flow passageways by secreting antithrombotic molecules (14), sion and risk for glaucoma, a leading cause of blindness. In this phagocytosing cellular debris, neutralizing reactive oxygen species study, single-cell RNA sequencing was performed to generate a (15–17), and mediating immune function (18, 19), while the cells in comprehensive cell atlas of human conventional outflow tissues. the JCT region display fibroblast- and smooth muscle-like proper- We obtained expression profiles of 17,757 genes from 8,758 cells ties, playing an important role in the generation and control of from eight eyes of human donors representing the outflow cell outflow resistance (7). -

The Impact of Transcription Factor Prospero Homeobox 1 on the Regulation of Thyroid Cancer Malignancy
International Journal of Molecular Sciences Review The Impact of Transcription Factor Prospero Homeobox 1 on the Regulation of Thyroid Cancer Malignancy Magdalena Rudzi ´nska 1,2 and Barbara Czarnocka 1,* 1 Department of Biochemistry and Molecular Biology, Centre of Postgraduate Medical Education, 01-813 Warsaw, Poland; [email protected] 2 Institute of Molecular Medicine, Sechenov First Moscow State Medical University, 119991 Moscow, Russia * Correspondence: [email protected]; Tel.: +48-225693812; Fax: +48-225693712 Received: 7 April 2020; Accepted: 30 April 2020; Published: 2 May 2020 Abstract: Transcription factor Prospero homeobox 1 (PROX1) is continuously expressed in the lymphatic endothelial cells, playing an essential role in their differentiation. Many reports have shown that PROX1 is implicated in cancer development and acts as an oncoprotein or suppressor in a tissue-dependent manner. Additionally, the PROX1 expression in many types of tumors has prognostic significance and is associated with patient outcomes. In our previous experimental studies, we showed that PROX1 is present in the thyroid cancer (THC) cells of different origins and has a high impact on follicular thyroid cancer (FTC) phenotypes, regulating migration, invasion, focal adhesion, cytoskeleton reorganization, and angiogenesis. Herein, we discuss the PROX1 transcript and protein structures, the expression pattern of PROX1 in THC specimens, and its epigenetic regulation. Next, we emphasize the biological processes and genes regulated by PROX1 in CGTH-W-1 cells, derived from squamous cell carcinoma of the thyroid gland. Finally, we discuss the interaction of PROX1 with other lymphatic factors. In our review, we aimed to highlight the importance of vascular molecules in cancer development and provide an update on the functionality of PROX1 in THC biology regulation. -

EVOLUTIONARY HISTORY of the PODOPLANIN GENE Jaime Renart
*Revised Manuscript (unmarked) 1 EVOLUTIONARY HISTORY OF THE PODOPLANIN GENE§ Jaime Renart1*, Diego San Mauro2, Ainhoa Agorreta2, Kim Rutherford3, Neil J. Gemmell3, Miguel Quintanilla1 1Instituto de Investigaciones Biomédicas Alberto Sols, Consejo Superior de Investigaciones Científicas (CSIC)-Universidad Autónoma de Madrid. Spain 2Department of Biodiversity, Ecology, and Evolution. Faculty of Biological Sciences. Universidad Complutense de Madrid. 28040 Madrid. Spain 3Department of Anatomy, School of Biomedical Sciences. University of Otago, PO Box 56, Dunedin 9054, New Zealand *Corresponding author: Jaime Renart Instituto de Investigaciones Biomédicas Alberto Sols, CSIC-UAM Arturo Duperier 4. 28029-Madrid. Spain. T: +34 915854412 [email protected] §We wish to dedicate this publication to the memory of our friend and colleague Luis Álvarez (†2016) 2 Keywords: PDPN, Evolution, Gnathostomes, exon/intron gain Abbreviations: BLAST, Basic Local Alignment Search Tool; CT, cytoplasmic domain; EC, extracellular domain; NCBI, National Center for Biotechnology Information; PDPN, podoplanin; SRA, Sequence Read Archive; TAE, Tris Acetate-EDTA buffer; PCR, polymerase chain reaction; UTR, untranslated region 3 ABSTRACT Podoplanin is a type I small mucin-like protein involved in cell motility. We have identified and studied the podoplanin coding sequence in 201 species of vertebrates, ranging from cartilaginous fishes to mammals. The N-terminal signal peptide is coded by the first exon; the transmembrane and intracellular domains are coded by the third exon (except for the last amino acid, coded in another exon with a long 3’-UTR). The extracellular domain has undergone variation during evolutionary time, having a single exon in cartilaginous fishes, teleosts, coelacanths and lungfishes. In amphibians, this single exon has been split in two, and in amniotes, another exon has been acquired, although it has been secondarily lost in Squamata. -

Evolution of Fibroblasts in the Lung Metastatic Microenvironment
RESEARCH ARTICLE Evolution of fibroblasts in the lung metastatic microenvironment is driven by stage-specific transcriptional plasticity Ophir Shani1†, Yael Raz1,2†, Lea Monteran1, Ye’ela Scharff1, Oshrat Levi-Galibov3, Or Megides4, Hila Shacham4, Noam Cohen1, Dana Silverbush5, Camilla Avivi6, Roded Sharan5, Asaf Madi1, Ruth Scherz-Shouval3, Iris Barshack6, Ilan Tsarfaty4, Neta Erez1* 1Department of Pathology, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel; 2Department of Obstetrics and Gynecology, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel; 3Department of Biomolecular Sciences, The Weizmann Institute of Science, Rehovot, Israel; 4Department of Clinical Microbiology and Immunology, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel; 5Blavatnik School of Computer Sciences, Faculty of Exact Sciences, Tel Aviv University, Tel Aviv, Israel; 6Department of Pathology, Sheba Medical Center, Tel Hashomer, affiliated with Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel Abstract Mortality from breast cancer is almost exclusively a result of tumor metastasis, and lungs are one of the main metastatic sites. Cancer-associated fibroblasts are prominent players in the microenvironment of breast cancer. However, their role in the metastatic niche is largely unknown. In this study, we profiled the transcriptional co-evolution of lung fibroblasts isolated from transgenic mice at defined stage-specific time points of metastases formation. Employing multiple *For correspondence: knowledge-based platforms of data analysis provided powerful insights on functional and temporal [email protected] regulation of the transcriptome of fibroblasts. We demonstrate that fibroblasts in lung metastases † These authors contributed are transcriptionally dynamic and plastic, and reveal stage-specific gene signatures that imply equally to this work functional tasks, including extracellular matrix remodeling, stress response, and shaping the Competing interests: The inflammatory microenvironment.