Hikma Launches Vecuronium Bromide for Injection
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Anaesthetic Experience with Paediatric Interventional Cardiology
320 Occasional Survey Anaesthetic experience with paediatric inter- Shobha Malviya MD, Frederick A. Burrows MD FRCPC, Albert E. Johnston MD FRCPC, Lee N. Benson MD FRCPC ventional cardiology Anaesthetic and sedation techniques, complications and out- In recent years interventional cardiologic procedures have comes were reviewed in 176 children undergoing 184 interven- replaced thoracic and cardiac surgery in the treatment of tional cardiologic procedures. Techniques included sedation certain cardiac anomalies in children. Balloon dilatation only, and ketamine, inhalational or narcotic anaesthesia. is particularly effective in treating pulmonary valve Ketamine infusion was the technique most frequently used. stenosis and is also performed as a nonsurgical alternative Ketamine was associated with a higher incidence of respiratory for patients with aortic valve stenosis, recoaretation of the complications (P < 0.05) than the other techniques. The higher aorta and pulmonary artery stenosis. Insertion of an incidence of hypercarbia (15.6 per cent), which did not affect occlusion device is used to treat patent ductus arteriosus. outcome, may be attributable to the use of supplemental Procedures performed in the cardiac catheterisation sedatives. The incidence of upper airway obstruction (7.8 per suite require a darkened room, allow poor access to the cenO was similar to that of previous studies. Vascular compro- often critically ill patient and present the potential for mise resulted from the procedure in 33 patients, necessitating sudden devastating complications, including rupture of a surgical correction in 16. Cardiac perforation occurred in four cardiac chamber. In addition, interventional procedures cases, causing one death. Pulmonary valve stenosis was most require a steady haemodynamic state before oxygen amenable to balloon dilatation and aortic valve stenosis least saturation and pressures in the various cardiac chambers amenable. -

12-6202 Document: 01018895054 Date Filed: 08/10/2012 Page: 1 FILED United States Court of Appeals UNITED STATES COURT of APPEALS Tenth Circuit
Appellate Case: 12-6202 Document: 01018895054 Date Filed: 08/10/2012 Page: 1 FILED United States Court of Appeals UNITED STATES COURT OF APPEALS Tenth Circuit FOR THE TENTH CIRCUIT August 10, 2012 Elisabeth A. Shumaker Clerk of Court MICHAEL EDWARD HOOPER, Plaintiff-Appellant, v. No. 12-6202 (D.C. No. 5:12-cv-00758-M) JUSTIN JONES, Director DOC; (W.D. Okla.) RANDALL G. WORKMAN, Warden; DOES, Unknown Executioners, Defendants-Appellees. ORDER AND JUDGMENT* Before MURPHY, O’BRIEN, and HOLMES, Circuit Judges. Michael Edward Hooper, an Oklahoma state prisoner scheduled for execution by lethal injection on August 14, 2012, appeals from the district court’s order * After examining the briefs and appellate record, this panel has determined unanimously that oral argument would not materially assist the determination of this appeal. See Fed. R. App. P. 34(a)(2); 10th Cir. R. 34.1(G). The case is therefore ordered submitted without oral argument. This order and judgment is not binding precedent, except under the doctrines of law of the case, res judicata, and collateral estoppel. It may be cited, however, for its persuasive value consistent with Fed. R. App. P. 32.1 and 10th Cir. R. 32.1. Appellate Case: 12-6202 Document: 01018895054 Date Filed: 08/10/2012 Page: 2 denying his motion for a preliminary injunction seeking to stay his execution. Exercising jurisdiction under 28 U.S.C. § 1292(a)(1), we AFFIRM.1 I Mr. Hooper was tried and convicted on three counts of first-degree murder and sentenced to death. See Hooper v. State, 947 P.2d 1090 (Okla. -

Quantitative Analysis of the Aminosteroidal Muscle Relaxant
J. Ecophysiol. Occup. Hlth. 3 & 4 (2013) 29-37 ® 2013 The Academy of Environmental Biology, India Quantitative Analysis of the Aminosteroidal Muscle Relaxant Vecuronium in Rat Liver and Kidneys using Ion-pairing and Liquid Chromatography-Tandem Mass Spectrometry Risha Jasmine Nathan*, P. Sharma, Lily Saroj Nathan1 Forensic Chemistry and Toxicology, Lok Nayak Jayaprakash Narayan National Institute of Criminology, New Delhi 1Ewing Christian College, University of Allahabad Abstract : Muscle relaxants are occasionally encountered in forensic toxicological cases for identification and quantitation. Due to unavailability of human samples, the analysis of the muscle relaxant vecuronium was done using post-mortem rat visceral samples. Rats were injected with bolus overdose of the drug and the post mortem samples were subject to ion-pair extraction with metanil yellow dye. Method for identification of the drug by LC-MS-MS was established first with secondary standard aqueous vecuronium solutions and then applied on the questioned samples. Quantitation was done to find the quantity of the drug in liver and kidney samples. Keywords : Aminosteroid, Muscle relaxant Vecuronium, Liquid Chromatography-Tandem Mass Spectrometry. Introduction ventilation is made available to the patient so as Muscle relaxants, better known as neuro- to maintain respiration as these drugs may also muscular-blocking drugs, are a class of drugs paralyze the diaphragm at the therapeutic dose that block neuromuscular transmission at required (Dripps, 1959). Despite taking neuromuscular junction which causes paralysis precautions, neuromuscular blocking drugs of the affected skeletal muscles. Of the two have been reported to have inadvertently classes, non-depolarizing neuromuscular caused deaths of patients by accident agents work without depolarizing the motor end (Newsletters, Nurse Advise, 2006). -

Product Monograph Vecuronium Bromide For
PRODUCT MONOGRAPH PrVECURONIUM BROMIDE FOR INJECTION Non-depolarizing Skeletal Neuromuscular Blocking Agent Pharmaceutical Partners of Canada Inc. Date of Preparation: 45 Vogell Road, Suite 200 January 15, 2008 Richmond Hill, ON L4B 3P6 Control No.: 119276 PRODUCT MONOGRAPH PrVECURONIUM BROMIDE FOR INJECTION Non-depolarizing Skeletal Neuromuscular Blocking Agent THIS DRUG SHOULD BE ADMINISTERED ONLY BY ADEQUATELY TRAINED INDIVIDUALS FAMILIAR WITH ITS ACTIONS, CHARACTERISTICS AND HAZARDS ACTIONS AND CLINICAL PHARMACOLOGY Vecuronium Bromide for Injection is a non-depolarizing neuromuscular blocking agent of intermediate duration possessing all of the characteristic pharmacological actions of this class of drugs (curariform). It acts by competing for cholinergic receptors at the motor end-plate. The antagonism to acetylcholine is inhibited and neuromuscular block reversed by acetylcholinesterase inhibitors such as neostigmine. Vecuronium Bromide for Injection is about 1/3 more potent than pancuronium; the duration of neuromuscular blockade produced by Vecuronium Bromide for Injection is shorter than that of pancuronium at initially equipotent doses. The time to onset of paralysis decreases and the duration of maximum effect increases with increasing Vecuronium Bromide for Injection doses. The use of a peripheral nerve stimulator is of benefit in assessing the degree of muscular relaxation. The ED90 (dose required to produce 90% suppression of the muscle twitch response with balanced anesthesia) has averaged 0.057 mg/kg (0.049 to 0.062 mg/kg in various studies). An initial Vecuronium Bromide for Injection dose of 0.08 to 0.10 mg/kg generally produces first depression of twitch in approximately 1 minute, good or excellent intubation conditions within 2.5 to 3 minutes, and maximum neuromuscular blockade within 3 to 5 minutes of injection in Vecuronium Bromide for Injection, Product Monograph Page 2 of 28 most patients. -
PMBJP Product.Pdf
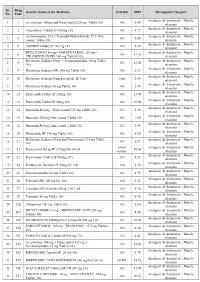
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Vecuronium Bromide As Control
A COMPARATIVE STUDY TO FIND AN IDEAL INTUBATING DOSE OF INJ. ROCURONIUM BROMIDE USING INJ. VECURONIUM BROMIDE AS CONTROL. Dissertation submitted In partial fulfillment for the award of M.D DEGREE EXAMINATION M.D. ANAESTHESIOLOGY & CRITICAL CARE BRANCH X GOVT KILPAUK MEDICAL COLLEGE AND HOSPITAL SUBMITTED TO THE TAMILNADU DR. MGR MEDICAL UNIVERSITY CHENNAI APRIL – 2011 1 DECLARATION I, Dr. D.Shunmuga Priya, solemnly declare that the dissertation, “A COMPARATIVE STUDY TO FIND AN IDEAL INTUBATING DOSE OF INJ. ROCURONIUM BROMIDE USING INJ. VECURONIUM BROMIDE AS CONTROL” is a bonafide work done by me in the Department of Anaesthesiology and Critical Care, Government Kilpauk, Medical College, Chennai under the able guidance of Prof. Dr. P.S. Shanmugam, MD., DA., Professor and HOD, Department of Anaesthesiology and Critical Care, Government Kilpauk Medical College, Chennai. Place : Chennai Date : (Dr. D.SHUNMUGA PRIYA) 5 CERTIFICATE This is to certify that this dissertation titled “A COMPARATIVE STUDY TO FIND AN IDEAL INTUBATING DOSE OF INJ. ROCURONIUM BROMIDE USING INJ. VECURONIUM BROMIDE AS CONTROL” has been prepared by Dr. D.SHUNMUGA PRIYA under my supervision in the Department of Anaesthesiology and Critical Care, Government Kilpauk Medical College, Chennai during the academic period 2008-2011 and is being submitted to the Tamil Nadu Dr. M.G.R. Medical University, Chennai in partial fulfillment of the University regulation for the award of the Degree of Doctor of Medicine (MD Anaesthesiology and Critical Care) and her dissertation is a bonafide work. Prof..Dr. V.Kanagasabai, M.D Prof. Dr. P.S.Shanmugam, M.D., D.A. DEAN Professor & HOD, Government Kilpauk Medical College Department of Anaesthesiology and & Hospital, Critical Care, Chennai. -

BRS Pharmacology
Pharmacology Gary C. Rosenfeld, Ph.D. Professor Department of Integrated Biology and Pharmacology and Graduate School of Biomedical Sciences Assistant Dean for Education Programs University of Texas Medical School at Houston Houston, Texas David S. Loose, Ph.D. Associate Professor Department of Integrated Biology and Pharmacology and Graduate School of Biomedical Sciences University of Texas Medical School at Houston Houston, Texas With special contributions by Medina Kushen, M.D. William Beaumont Hospital Royal Oak, Michigan Todd A. Swanson, M.D., Ph.D. William Beaumont Hospital Royal Oak, Michigan Acquisitions Editor: Charles W. Mitchell Product Manager: Stacey L. Sebring Marketing Manager: Jennifer Kuklinski Production Editor: Paula Williams Copyright C 2010 Lippincott Williams & Wilkins 351 West Camden Street Baltimore, Maryland 21201-2436 USA 530 Walnut Street Philadelphia, PA 19106 All rights reserved. This book is protected by copyright. No part of this book may be reproduced in any form or by any means, including photocopying, or utilized by any information storage and retrieval system without written permission from the copyright owner. The publisher is not responsible (as a matter of product liability, negligence or otherwise) for any injury resulting from any material contained herein. This publication contains information relating to general principles of medical care which should not be construed as specific instructions for individual patients. Manufacturers’ product information and package inserts should be reviewed for current information, including contraindications, dosages and precautions. Printed in the United States of America Library of Congress Cataloging-in-Publication Data Rosenfeld, Gary C. Pharmacology / Gary C. Rosenfeld, David S. Loose ; with special contributions by Medina Kushen, Todd A. -

Doxacurium Chloride Injection Abbott Laboratories
NUROMAX - doxacurium chloride injection Abbott Laboratories ---------- NUROMAX® (doxacurium chloride) Injection This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL DESCRIPTION NUROMAX (doxacurium chloride) is a long-acting, nondepolarizing skeletal muscle relaxant for intravenous administration. Doxacurium chloride is [1α,2β(1'S*,2'R*)]-2,2' -[(1,4-dioxo-1,4- butanediyl)bis(oxy-3,1-propanediyl)]bis[1,2,3,4-tetrahydro-6,7,8-trimethoxy-2- methyl-1-[(3,4,5- trimethoxyphenyl)methyl]isoquinolinium] dichloride (meso form). The molecular formula is C56H78CI2N2O16 and the molecular weight is 1106.14. The compound does not partition into the 1- octanol phase of a distilled water/ 1-octanol system, i.e., the n-octanol:water partition coefficient is 0. Doxacurium chloride is a mixture of three trans, trans stereoisomers, a dl pair [(1R,1'R ,2S,2'S ) and (1S,1'S ,2R,2'R )] and a meso form (1R,1'S,2S,2'R). The meso form is illustrated below: NUROMAX Injection is a sterile, nonpyrogenic aqueous solution (pH 3.9 to 5.0) containing doxacurium chloride equivalent to 1 mg/mL doxacurium in Water for Injection. Hydrochloric acid may have been added to adjust pH. NUROMAX Injection contains 0.9% w/v benzyl alcohol. Reference ID: 2867706 CLINICAL PHARMACOLOGY NUROMAX binds competitively to cholinergic receptors on the motor end-plate to antagonize the action of acetylcholine, resulting in a block of neuromuscular transmission. This action is antagonized by acetylcholinesterase inhibitors, such as neostigmine. Pharmacodynamics NUROMAX is approximately 2.5 to 3 times more potent than pancuronium and 10 to 12 times more potent than metocurine. -

Vecureinj.Pdf
NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted with 5 mL water for injection corresponds to 2 mg vecuronium bromide per mL in a clear almost clear isotonic solution. For the full list of excipients, see section 6.1 3. Pharmaceutical Form White, freeze dried powder for injection. Following reconstitution with solvent (water for injection) the resultant solution is isotonic and has a pH of 4. 4. Clinical Particulars 4.1 Therapeutic indications Vecure is indicated as an adjunct to general anaesthesia to facilitate tracheal intubation and to provide skeletal muscle relaxation during surgery. 4.2 Dose and method of administration Dosage Like other neuromuscular blocking agents, VECURE should only be administered by, or under supervision of, experienced clinicians who are familiar with the action and use of these agents. Like with other neuromuscular blocking agents, the dosage of VECURE should be individualised in each patient. The anaesthetic method used, the expected duration of surgery, the possible interaction with other medicines that are administered before or during anaesthesia and the condition of the patient should be taken into account when determining the dose. The use of an appropriate neuromuscular monitoring technique is recommended to monitor neuromuscular block and recovery. Inhalational anaesthetics do potentiate the neuromuscular blocking effects of VECURE. This potentiation however, becomes clinically relevant in the course of anaesthesia, when the volatile agents have reached the tissue concentrations required for this interaction. Consequently, adjustments with VECURE should be made by administering smaller maintenance doses at less frequent intervals or by using lower infusion rates of VECURE during long lasting procedures (longer than 1 hour) under inhalational anaesthesia (see section 4.5). -

Atracurium Besylate Injection
PRODUCT MONOGRAPH PrATRACURIUM BESYLATE INJECTION 10 mg/mL Skeletal Neuromuscular Blocking Agent Sandoz Canada Inc. Date of Revision: March 25, 2013 145 Jules-Léger Boucherville, QC, Canada J4B 7K8 Control no.: 161881 Atracurium Besylate Injection Page 1 of 19 PrATRACURIUM BESYLATE INJECTION 10 mg/mL THERAPEUTIC CLASSIFICATION Skeletal Neuromuscular Blocking Agent ACTIONS AND CLINICAL PHARMACOLOGY Atracurium besylate is a nondepolarizing, intermediate-duration, skeletal neuromuscular blocking agent. Nondepolarizing agents antagonize the neurotransmitter action of acetylcholine by binding competitively to cholinergic receptor sites on the motor end-plate. This antagonism is inhibited, and neuromuscular block reversed, by acetylcholinesterase inhibitors such as neostigmine, edrophonium and pyridostigmine. The duration of neuromuscular blockade produced by atracurium besylate is approximately one-third to one-half the duration seen with d-tubocurarine, metocurine and pancuronium at equipotent doses. As with other nondepolarizing neuromuscular blockers, the time to onset of paralysis decreases and the duration of maximum effect increases with increasing atracurium besylate doses. The pharmacokinetics of atracurium besylate in man are essentially linear within the 0.3 to 0.6 mg/kg dose range. The elimination half-life is approximately 20 minutes. The duration of neuromuscular blockade does not correlate with plasma pseudocholinesterase levels and is not altered by the absence of renal function. INDICATIONS AND CLINICAL USE Atracurium Besylate Injection is indicated, as an adjunct to general anaesthesia, to facilitate endotracheal intubation and to provide skeletal muscle relaxation during surgery or mechanical ventilation. It can be used most advantageously if muscle twitch response to peripheral nerve stimulation is monitored. CONTRAINDICATIONS Atracurium besylate is contraindicated in patients known to have a hypersensitivity to it. -

(12) Patent Application Publication (10) Pub. No.: US 2010/009874.6 A1 King (43) Pub
US 201000987.46A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2010/009874.6 A1 King (43) Pub. Date: Apr. 22, 2010 (54) COMPOSITIONS AND METHODS FOR Related U.S. Application Data TREATING PERIODONTAL DISEASE COMPRISING CLONDINE, SULINDAC (60) Provisional application No. 61/106,815, filed on Oct. AND/OR FLUOCNOLONE 20, 2008. Publication Classification (75) Inventor: Vanja Margareta King, Memphis, TN (US) (51) Int. Cl. A6F I3/00 (2006.01) Correspondence Address: A6II 3/56 (2006.01) MEDTRONIC (52) U.S. Cl. ......................................... 424/435: 514/171 Attn: Noreen Johnson - IP Legal Department (57) ABSTRACT 2600 Sofamor Danek Drive MEMPHIS, TN38132 (US) Effective treatments of periodontal disease for extended peri ods of time are provided. Through the administration of an (73) Assignee: Warsaw Orthopedic, Inc., Warsaw, effective amount of clonidine, Sulindac, and/or fluocinolone IN (US) at or near a target site, one can reduce, prevent, and/or treat periodontal disease. In some embodiments, when appropriate (21) Appl. No.: 12/572,387 formulations are provided within biodegradable polymers, treatment can be continued for at least two weeks to two (22) Filed: Oct. 2, 2009 months. Patent Application Publication Apr. 22, 2010 Sheet 1 of 3 US 2010/009874.6 A1 FIG. 1 Clonidine 15 o 9. 10 wd O O 2 SS 5 O RANK Control 100 M 10 M M 0.1 LM Concentration Patent Application Publication Apr. 22, 2010 Sheet 2 of 3 US 2010/009874.6 A1 FIG. 2 Sulindac 5 1 O RANK Control 70 M 35M 7.5 M ConCentration Patent Application Publication Apr. -

COMPARISON of ROCURONIUM BROMIDE and SUCCINYLCHOLINE CHLORIDE for USE DURING RAPID SEQUENCE INTUBATION in ADULTS Ch
DOI: 10.18410/jebmh/2015/672 ORIGINAL ARTICLE COMPARISON OF ROCURONIUM BROMIDE AND SUCCINYLCHOLINE CHLORIDE FOR USE DURING RAPID SEQUENCE INTUBATION IN ADULTS Ch. Penchalaiah1, N. Jagadeesh Babu2, T. Kumar3 HOW TO CITE THIS ARTICLE: Ch. Penchalaiah, N. Jagadeesh Babu, T. Kumar. ”Comparison of Rocuronium Bromide and Succinylcholine Chloride for Use during Rapid Sequence Intubation in Adults”. Journal of Evidence based Medicine and Healthcare; Volume 2, Issue 32, August 10, 2015; Page: 4796-4806, DOI: 10.18410/jebmh/2015/672 ABSTRACT: BACKGROUND AND OBJECTIVE: The goal of rapid sequence intubation is to secure the patients airway smoothly and quickly, minimizing the chances of regurgitation and aspiration of gastric contents. Traditionally succinylcholine chloride has been the neuromuscular blocking drug of choice for use in rapid sequence intubation because of its rapid onset of action and profound relaxation. Succinylcholine chloride remains unsurpassed in providing ideal intubating conditions. However the use of succinylcholine chloride is associated with many side effects like muscle pain, bradycardia, hyperkalaemia and rise in intragastric and intraocular pressure. Rocuronium bromide is the only drug currently available which has the rapidity of onset of action like succinylcholine chloride. Hence the present study was undertaken to compare rocuronium bromide with succinylcholine chloride for use during rapid sequence intubation in adult patients. METHODOLOGY: The study population consisted of 90 patients aged between 18-60 years posted for various elective surgeries requiring general anaesthesia. Study population was randomly divided into 3 groups with 30 patients in each sub group. 1. Group I: Intubated with 1 mg kg-1 of succinylcholine chloride (n=30).