Cyclodextrins As Drug Carrier Molecule: a Review
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

16A-Fluoro-5-Androsten-17-0Ne
MILITARY MEDICINE, 167, Suppl. 1:060,2002 Inhibition of Adjuvant-Induced Arthritis by 16a-Fluoro-5-Androsten-17-0ne Guarantor: Arthur G. Schwartz, PhD Contributors: Arthur G. Schwartz, PhD; Laura L. Pashko, PhD The adrenal steroid dehydroepiandrosterone (DHEA) produces tant receptor and that inhibition ofthis enzyme by DHEA me cancer-preventive, antiatherosclerotic, antidiabetic, Immuno diates some of the biological effects of the steroid. DHEA is a modulating, and anti-inflammatory effects in laboratory ani potent uncompetitive inhibitor of G6PDH, the rate-controlling mals. The clinical use of DHEAis limited by its androgenicity. Downloaded from https://academic.oup.com/milmed/article/167/suppl_1/60/4820113 by guest on 01 October 2021 enzyme in thepentose phosphate pathway, 10 and it appearsthat Wehave developed a synthetic congener ofDHEAcalled fluaster the antiproliferative and antitumor-promoting effects of the one that, in animal tests, lacks the androgenicity, estrogenicity, and peroxisome-proliferating effects of DHEA but retains the DHEA class of steroids is mediated through inhibition of cancer-preventive, antidiabetic, and anti-inflammatory efficacy. G6PDH.1,11,12 Two important metabolic consequences ofG6PDH This report discusses how fluasterone ameliorates the develop inhibition are a lowering ofNADPH-reducing equivalents avail ment of joint inflammation in an adjuvant-arthritis model in able forbiosynthetic processes, including ribo- and deoxyribo Lewis rats. nucleotide synthesis, and a reduction in ribose-5-phosphate, -
![©[2010] Diana Y. Lee ALL RIGHTS RESERVED](https://docslib.b-cdn.net/cover/3911/%C2%A9-2010-diana-y-lee-all-rights-reserved-433911.webp)
©[2010] Diana Y. Lee ALL RIGHTS RESERVED
©[2010] Diana Y. Lee ALL RIGHTS RESERVED EFFECTS OF DRYING METHODS ON THE STABILITY OF 2,4-DECADIENAL ENCAPSULATED IN AN O/W NANOEMULSION By DIANA Y. LEE A thesis submitted to the Graduate School-New Brunswick Rutgers, The State University of New Jersey in partial fulfillment of the requirements for the degree of Masters in Science Graduate Program in Food Science Written under the direction of Qingrong Huang and approved by ________________________ ________________________ ________________________ New Brunswick, New Jersey [May, 2010] ABSTRACT OF THE THESIS Effects of drying methods on the stability of 2,4-decadienal encapsulated in an o/w nanoemulsion By DIANA Y. LEE Thesis Director: Qingrong Huang The flavor industry has utilized many encapsulation methods in order to provide customers with stable flavors that maintain their integrity during various processing procedures. Savory flavors in particular have a unique hurdle to overcome, as they are subject to extreme temperature abuse, such as frying, baking, sautéing, and boiling. Highly sensitive compounds such as 2,4-Decadienal, that provide distinct characteristics to savory foods such as french fries and chicken, are particularly susceptible to change during these processes. Using oil in water nanoemulstions of diameters between 20-800 nanometers as well as various drying methods to encapsulate volatile compounds have been an exciting avenue for flavor encapsulation. The present research will focus on the stability of 2,4- Decadienal using oil in water nanoparticles of medium chain triglycerides (Neobee) in addition to multilayer encapsulation; spray drying and freeze drying. ii A slurry of 2,4-decadienal, maltodextrin, gum, Neobee and water were homogenized via high speed at 13,500 rpm and high pressure under 1500 bar to create a stable nanoemulsion. -

Encapsulation of Gases in Powder Solid Matrices and Their Applications: a Review
ÔØ ÅÒÙ×Ö ÔØ Encapsulation of gases in powder solid matrices and their applications: a review Thao M. Ho, Tony Howes, Bhesh R. Bhandari PII: S0032-5910(14)00265-4 DOI: doi: 10.1016/j.powtec.2014.03.054 Reference: PTEC 10142 To appear in: Powder Technology Received date: 4 December 2013 Revised date: 18 March 2014 Accepted date: 21 March 2014 Please cite this article as: Thao M. Ho, Tony Howes, Bhesh R. Bhandari, Encapsulation of gases in powder solid matrices and their applications: a review, Powder Technology (2014), doi: 10.1016/j.powtec.2014.03.054 This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. ACCEPTED MANUSCRIPT Encapsulation of gases in powder solid matrices and their applications: a review Thao M. Hoa, Tony Howes b, Bhesh R. Bhandaria * a School of Agriculture and Food Sciences, The University of Queensland, QLD 4072, Australia b School of Chemical Engineering, The University of Queensland, St. Lucia, QLD 4072, Australia * Corresponding author. Address: School of Agriculture and Food Sciences, The University of Queensland, Brisbane, QLD 4072, Australia. Tel.: +61 7 33469192; fax: +61 7 33651177. E-mail address: [email protected] (B.R. -

Synthesis and Properties of Amphiphilic Hyperbranched Poly(Dimethylsiloxane) Possessing Hydrophilic Terminal Group
Polymer Journal, Vol.34, No. 10, pp 755—760 (2002) Synthesis and Properties of Amphiphilic Hyperbranched Poly(dimethylsiloxane) Possessing Hydrophilic Terminal Group † Kyung-Mee KIM, Mitsutoshi JIKEI, and Masa-aki KAKIMOTO Department of Organic and Polymeric Materials, Tokyo Institute of Technology, Meguro-ku, Tokyo 152–8552, Japan (Received May 13, 2002; Accepted August 26, 2002) ABSTRACT: The amphiphilic hyperbranched poly(dimethylsiloxane) (HPDMS) bearing dimethylamine groups on the terminal position was synthesized and characterized. The obtained polymer exhibited low viscosity, and good solu- bility in ether, THF, and acidic aqueous solution. In the acidic aqueous polymer solution, UV-vis absorption and photo luminescence of water-insoluble chromic compounds such as diphenylhexatriene were measured in order to investigate solvating power of hyperbranched poly(dimethylsiloxane)s. It has turned out from the UV absorbance data that a HPDMS solubilized about five molecules of 1,6-diphenylhexatriene. KEY WORDS Amphiphilc Polymer / Hyperbranched Polymer / PDMS / Poly(dimethylsiloxane) / Hydrophilic / Hydrophobic / It is well known that silicon-based polymers, partic- of hyperbranched poly(dimethylsiloxane)s (HPDMSs) ularly polysiloxanes, have unique properties that lead from a novel AB2 type monomer, bis(dimethyl- to a wide range of applications in coatings, adhesives, diethylaminosiloxy)methylsiloxydimethylsilanol.24 cosmetics, and surfactants.1–3 Thus, the preparation The HPDMS end-capped with dimethylphenylsilanol of their hyperbranched -

Molecular Encapsulation in Kinetically Trapped, Hydrogen-Bonded Pyrogallolarene Hexamers
University of Denver Digital Commons @ DU Electronic Theses and Dissertations Graduate Studies 6-1-2014 Molecular Encapsulation in Kinetically Trapped, Hydrogen-Bonded Pyrogallolarene Hexamers Jennifer Christine Chapin Lake University of Denver Follow this and additional works at: https://digitalcommons.du.edu/etd Part of the Organic Chemistry Commons Recommended Citation Chapin Lake, Jennifer Christine, "Molecular Encapsulation in Kinetically Trapped, Hydrogen-Bonded Pyrogallolarene Hexamers" (2014). Electronic Theses and Dissertations. 120. https://digitalcommons.du.edu/etd/120 This Dissertation is brought to you for free and open access by the Graduate Studies at Digital Commons @ DU. It has been accepted for inclusion in Electronic Theses and Dissertations by an authorized administrator of Digital Commons @ DU. For more information, please contact [email protected],[email protected]. MOLECULAR ENCAPSULATION IN KINETICALLY TRAPPED, HYDROGEN-BONDED PYROGALLOLARENE HEXAMERS __________ A Dissertation Presented to the Faculty of Natural Sciences and Mathematics University of Denver __________ In Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy __________ by Jennifer Christine Chapin Lake June 2014 Advisor: Byron W. Purse ©Copyright by Jennifer Christine Chapin Lake 2014 All Rights Reserved Author: Jennifer Christine Chapin Lake Title: MOLECULAR ENCAPSULATION IN KINETICALLY TRAPPED, HYDROGEN-BONDED PYROGALLOLARENE HEXAMERS Advisor: Byron W. Purse Degree Date: June 2014 Abstract Pyrogallolarene and resorcinarene hexamers are hydrogen-bonded capsules that self-assemble in the solid state and can be studied in gaseous and solution phases. Guest loading within pyrogallolarene hexamers in solution has primarily been comprised of solvent molecules with some tertiary amines. A novel solvent-free method for loading guests into the interior of the hexamer has been shown to be effective for encapsulation of a variety of molecules. -

Encapsulation of Risperidone by Methylated Β-Cyclodextrins: Physicochemical and Molecular Modeling Studies
molecules Article Encapsulation of Risperidone by Methylated β-Cyclodextrins: Physicochemical and Molecular Modeling Studies 1,2 3 1,2, 1,2 Laura Sbârcea , Ionut, -Mihai Tănase , Adriana Ledet, i * , Denisa Cîrcioban , Gabriela Vlase 4, Paul Barvinschi 5, Marinela Miclău 6, Renata-Maria Văru¸t 7, 8 1,2,3 Cristina Trandafirescu and Ionut, Ledet, i 1 Department of Pharmacy I, Faculty of Pharmacy, “Victor Babe¸s”University of Medicine and Pharmacy, 2 Eftimie Murgu Square, 300041 Timisoara, Romania; [email protected] (L.S.); [email protected] (D.C.); [email protected] (I.L.) 2 Advanced Instrumental Screening Center, Faculty of Pharmacy, “Victor Babes” University of Medicine and Pharmacy, 2 Eftimie Murgu Square, 300041 Timisoara, Romania 3 Faculty of Industrial Chemistry and Environmental Engineering, Politehnica University of Timisoara, 6 Vasile Parvan Blvd, 300223 Timisoara, Romania; [email protected] 4 Research Centre for Thermal Analysis in Environmental Problems, West University of Timisoara, 16 Pestalozzi Street, 300115 Timisoara, Romania; [email protected] 5 Faculty of Physics, West University of Timisoara, 4 Vasile Parvan Blvd, 300223 Timisoara, Romania; [email protected] 6 National Institute for Research and Development in Electrochemistry and Condensed Matter, 144 Dr. A. Păunescu-Podeanu Street, 300587 Timisoara, Romania; [email protected] 7 Faculty of Pharmacy, University of Medicine and Pharmacy Craiova, 2–4 Petru Rares Street, 200349 Craiova, Romania; [email protected] 8 Department of -

Photocontrolled Self-Assembly of a Fluorescent Anthracene at Nano- and Microscales Christiaan J.F
Photocontrolled self-assembly of a fluorescent anthracene at nano- and microscales Christiaan J.F. de Vet To cite this version: Christiaan J.F. de Vet. Photocontrolled self-assembly of a fluorescent anthracene at nano- and mi- croscales. Organic chemistry. Université de Bordeaux, 2016. English. NNT : 2016BORD0333. tel-02003479 HAL Id: tel-02003479 https://tel.archives-ouvertes.fr/tel-02003479 Submitted on 1 Feb 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. THÈSE PRÉSENTÉE POUR OBTENIR LE GRADE DE DOCTEUR DE L’UNIVERSITÉ DE BORDEAUX ÉCOLE DOCTORALE DES SCIENCES CHIMIQUES SPÉCIALITÉ CHIMIE ORGANIQUE Christiaan J.F. de Vet Auto-assemblage d'un anthracène fluorescent aux échelles nano- et micrométriques par photoréaction contrôlée Photocontrolled self-assembly of a fluorescent anthracene at nano- and microscales Sous la direction de: Prof. André DEL GUERZO Soutenue le 9 Décembre 2016 Membres du jury : Mme. ISHOW, Elena Professeur, Univ. de Nantes Rapporteur M. SOPPERA, Olivier Directeur de recherche, Univ. de Haute Alsace Rapporteur M. BARTHELEMY, Philippe Professeur, Univ. de Bordeaux Président M. DEL GUERZO, André Professeur, Univ. de Bordeaux Directeur Voor ome Ad… Remerciements These three years have been an interesting experience which did not only consist of dealing with the challenges of scientific research, but also a change of culture and language. -

An Orally Bioavailable Synthetic Analog of an Active Dehydroepiandrosterone Metabolite Reduces Established Disease in Rodent Models of Rheumatoid Arthritis
0022-3565/09/3293-1100–1109$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 329, No. 3 Copyright © 2009 by The American Society for Pharmacology and Experimental Therapeutics 145086/3474987 JPET 329:1100–1109, 2009 Printed in U.S.A. An Orally Bioavailable Synthetic Analog of an Active Dehydroepiandrosterone Metabolite Reduces Established Disease in Rodent Models of Rheumatoid Arthritis Halina Offner, Gary S. Firestein, David L. Boyle, Raymond Pieters, James M. Frincke, Armando Garsd, Steven K. White, Christopher L. Reading, and Dominick L. Auci Department of Neurology, Oregon Health and Science University, Portland, Oregon (H.O.); Institute for Risk Assessment Sciences-Immunotoxicology, Utrecht University, Utrecht, The Netherlands (R.P.); Division of Rheumatology, Allergy and Immunology, University of California at San Diego School of Medicine, La Jolla, California (D.L.B., G.S.F.); and Hollis-Eden Pharmaceuticals Inc., San Diego, California (J.M.F., A.G., S.K.W., C.L.R., D.L.A.) Downloaded from Received August 20, 2008; accepted March 17, 2009 ABSTRACT Dehydroepiandrosterone (DHEA) treatment provides diverse anti- even when treatments were delayed until 7 days after the onset jpet.aspetjournals.org inflammatory benefits in rodent models of diseases, including of arthritis. Furthermore, dose-dependent benefit was observed rheumatoid arthritis (RA), but only limited benefits to patients. In in the DBA mouse model of collagen antibody-induced arthritis, rodents, DHEA is metabolized to (among others) androstene- as well as reductions in IL-6 and matrix metalloproteinase-3 3,7,17-triol (AET), which retains potent anti-inflammatory mRNA levels in joints at the peak of disease and at the end of activity. -
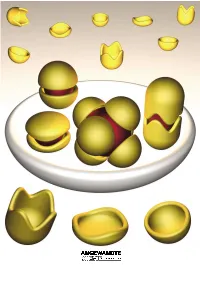
Molecular Encapsulation
REVIEWS Molecular Encapsulation Fraser Hof, Stephen L. Craig, Colin Nuckolls, and Julius Rebek, Jr.* Dedicated to Ivar Ugi Louis Kahn, architect of the Salk nents. This review is about molecular ble–an expression of the molecule×s Institute in La Jolla, said[1] ™even a aggregates of a certain sort, namely, desire to be something more than it common, ordinary brick wants to be those that assemble and more or less is–results from instructions engi- something more than it is.∫ Suppose completely surround other molecules. neered into the molecules during their that were also true of molecules. We Taking part in this intimacy imparts creation. know that they can and do aggregate; unique properties to the participants, they give complex structures, and by and new functions emerge from the Keywords: host ± guest systems ¥ mo- doing so they acquire new properties– aggregate as a whole. For the most lecular capsules ¥ molecular recogni- functions that may not be apparent part, we emphasize self-complementa- tion ¥ self-assembly from a study of the individual compo- ry structures. Their ability to assem- 1. Introduction reaction templates were devised[17] and more complex systems were contrived. This is engineering (or is it art?)[18] at the In the 1980s, most of the publications on molecular molecular level.[19] It did not matter that these systems weren×t recognition dealt with the selectivity of synthetic receptors particularly efficient, what mattered was that they improved and the energetics of intermolecular forces, and were confined the understanding of three-component systems. for the most part to bimolecular systems. -

Novel Architectures in Cavitand Chemistry: Shaping Molecular Inner Space
Novel Architectures in Cavitand Chemistry: Shaping Molecular Inner Space DISSERTATION Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the Graduate School of The Ohio State University By Keith Robert Hermann Graduate Program in Chemistry The Ohio State University 2014 Dissertation Committee: Professor Jovica D. Badjić, Advisor Professor T.V. RajanBabu Professor Psaras L. McGrier Copyright by Keith Robert Hermann 2014 Abstract As long as chemists have marveled at the specificity of interactions in enzymes, nucleic acids, and other biological motifs that contain an inner cavity, there has been much desire to construct molecules that mimic these in function. Inspired by work performed by Charles J. Pederson on crown ethers, Donald J. Cram conducted seminal research into the construction and the host-guest interactions of a number of molecular architectures, specifically with his work on carcerands and hemicarcerands. Since Cram’s early work, the field of cavitand chemistry has taken off, providing endless examples in architecture capable of enclosing the space around a guest molecule. Some common examples of artificial hosts are cryptophanes, cucurbit[n]urils, and calixaranes. Function and application vary as much as structure, and range from stabilizing reactive intermediates and probing fundamental questions in physical organic chemistry, to drug delivery and chiral separations. Meanwhile, in the Badjić Group, the development of cavitands functionalized with dynamic apertures, or gates, has allowed us to probe fundamental questions in the kinetics of encapsulation. These studies, however, have relied upon a single C3v symmetric host. As a result, the group has taken on the challenge of constructing new architectures giving consideration toward the inner space geometry to allow for installation of “gate” moieties onto these architectures. -

Shear-Induced Microporous Nanocomposite Epoxy Thermosets (Minet) Molla Hasan, Yogin Patel, Arielle R
Shear-Induced Microporous Nanocomposite Epoxy Thermosets (MiNET) Molla Hasan, Yogin Patel, Arielle R. Gamboa, Michael J. Grzenda, Valeria Saro-Cortes, Vivek Mhatre, Jonathan P. Singer Abstract To create microporous nanocomposite epoxy thermosets (MiNET), a mixing pathway is demonstrated in which a bicontinuous emulsion gel (bijel) like viscous fluid is kinetically trapped by high shear mixing of immiscible liquids, surfactant, and nanoparticles. The MiNETs are prepared from common ingredients, that are widely employed in industry, including epoxy resin, vegetable oil, epoxidized soybean oil, and different types of nanoparticles such as silica, activated carbon, alumina, and zinc oxide. MiNETs prepared by the presented route are processed at ambient conditions and exhibit low shrinkage (less than 2%). Furthermore, they are suitable to erect macro- to microscale structures with high precision and various porosity. The interconnected porous architecture of MiNET is even preserved in microscale features and thus ensures the mass transport in microstructures. With facile processability and tunability of pore sizes in a wide range (~100 nm to few microns), the proposed route overcomes the two major roadblocks – difficulty in fabrication and large domain size (on the order of 5µm or larger) – of bijel-like materials to apply in catalysis, energy storage, and molecular encapsulation. Introduction: Porous materials with tunable surface to volume ratio and chemistry have numerous applications, including tissue engineering,[1] catalysis,[2] smart filtering,[3, 4], fuel cells,[5] and sensors.[6, 7] These materials are also essential to unlock the physics of mass transport phenomena in porous media.[7, 8] To synthesize the porous materials in the aforementioned applications and studies, numerous methods have been developed, for example metal-organic frameworks (MOFs), sol-gel, particles sintering, and emulsion templating. -

Inhibition of Carcinogen-Activating Enzymes by 16␣-Fluoro-5-Androsten-17-One
[CANCER RESEARCH 62, 3685–3690, July 1, 2002] Inhibition of Carcinogen-activating Enzymes by 16␣-Fluoro-5-androsten-17-one Henry P. Ciolino,1 Christopher J. MacDonald, and Grace Chao Yeh Cellular Defense and Carcinogenesis Section, Basic Research Laboratory, Center for Cancer Research, National Cancer Institute at Frederick, NIH, Frederick, Maryland 21702- 1201 ABSTRACT decrease in the incidence of a number of different types of cancer in humans (5, 6). In animal models, DHEA has been shown to inhibit In the present study, we examined the effect of a synthetic analogue of both spontaneous and chemically induced carcinogenesis in rodents the chemopreventive hormone dehydroepiandrosterone, 16␣-fluoro-5- (7–9). Specifically, DHEA inhibits both skin and mammary tumori- androsten-17-one, also known as fluasterone, on the activity and expres- sion of carcinogen-activating enzymes in MCF-7 cells. The increase in genesis caused by the PAH DMBA (10–13). DHEA has been shown cytochrome P450 (CYP) 1A1 and 1B1 activity, as measured by to inhibit DMBA activation in vitro (14) and DMBA-DNA binding in ethoxyresorufin-O-deethylase activity, in cells treated with the carcino- vivo (15–18). The dramatic decline of DHEA levels in humans with gens dimethylbenzanthracene (DMBA) or 2,3,5,7-tetrachlorodibenzo-p- advancing age is, therefore, of substantial concern. However, DHEA dioxin (TCDD), was inhibited by cotreatment with fluasterone. However, supplementation is problematic, because it can be converted to both treatment of the cells with fluasterone after induction with DMBA or testosterone and estrone (19), and it exhibits considerable liver tox- TCDD failed to decrease enzyme activity, indicating that inhibition was icity (20) and hepatocarcinogenicity (21).