By C K Cheung Atomic Physics
Matter ----- composed of atoms and molecules. Atom ----- composed of ??????
J.J. Thomson Experiment (1897 )
Thomson’s ‘ plum – pudding ‘model of atoms:
electron 10 – 10 m +ve charged matter spread uniformly over the entire region
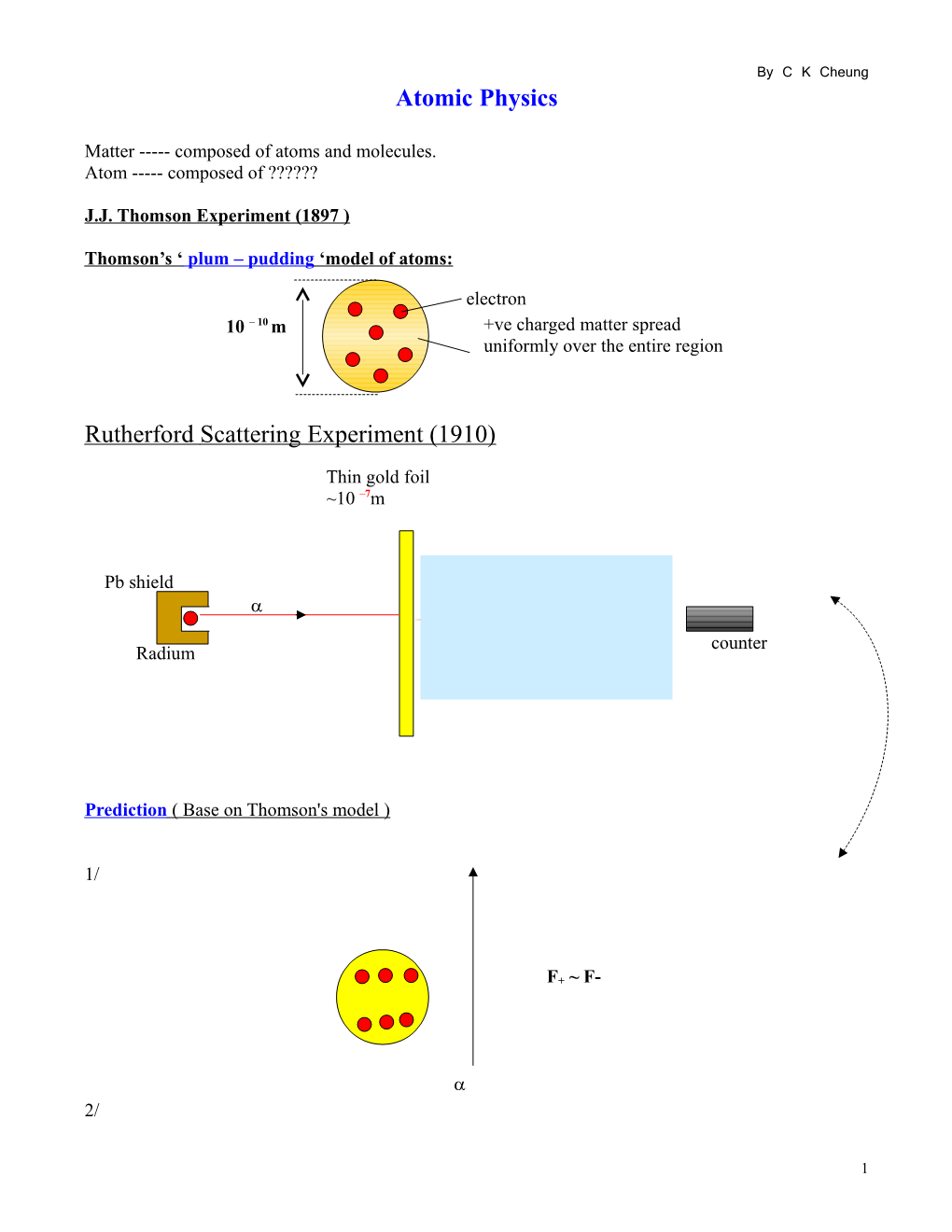
Rutherford Scattering Experiment (1910)
Thin gold foil ~10 –7m
Pb shield
< 100 counter Radium
Prediction ( Base on Thomson's model )
1/
F+ ~ F-
2/
1 By C K Cheung
F+ ~ F+’ F- ~ F-‘
Hence, no matter where the particles hit, they should be deflected only by a small angle. Experimental results: ..\powerpoint\atomic structure\Rutherford's expt..ppt
Thin gold foil ~10 –7m
Pb
- < 100 counter Radium very few ( 1 in 8000 )
1/ many ( > 99.5 % ) of the particles go straight through the foil and were deflected through very small angles ( < 100 ).
2/ very few ( ~ 1 in 8000 ) suffered deflections of more than 900.
Note: 1. gold is ductile 2. thin gold foil to avoid multiple collisions inside the foil.
2 By C K Cheung Rutherford’s model of atom
1/ Due to large-angle scattering of very few particles a small massive +ve core or nucleus.
2/ The –ve charge at relatively large distances from the nucleus so that their negative charge did not act as a shield to the +ve nucleus charge when the particles penetrate the atom.
3/ The –ve charge in circular motion to balance the electrostatic attraction.
Rutherford’s model of atoms (1911 )
3 By C K Cheung Rutherford’s estimate of nucleus size
r b
Z u v = 0 R KEPE
Z = atomic no. ..\powerpoint\atomic structure\size of atom.ppt = no. of proton inside nucleus KE = PE
1 2 1 (2e)(Ze) mu 2 40 r
Ze2 r = 2 0mu
For closest approach: r = b
Ze2 b = 2 0mu
Take : Z = 79 e = 1.6x10 – 19 - 12 0= 8.86x10 – 27 m=6.7x 10 u = 2x10 7 ms - 1
b ~ 10 –14 m
b R ~ ~ 10 – 15 m 2
4 By C K Cheung
Limitations of Rutherford’s model
1/
By classical theories, all accelerating charged particles will emit radiation ( energy ). If Rutherford’s model is correct, the accelerating electrons will lose energy continuously, then they will be adhered to the nucleus and all atoms will finally be collapsed!
2/ Rutherford’s model does not predict the existence of discrete energy levels in atoms.
3/ Rutherford’s scattering experiment founded the high energy physics.
5 By C K Cheung
Bohr’s Theory of atoms (1913)
Consider an H atom:
e
p
r
1 (e)(e) mv 2 2 = 4 0 r r e2 v2 = ………………(1) 4 0mr
First Theory: Only those orbits occur for which the angular momenta of the planetary electron are integral h h multiples of or n( ) 2 2 h mvr = n( ) 2 h v = n( )…………………(2) 2mr
(2) (1)
n 2 h 2 e2 2 2 2 = 4 m r 4 0mr
h2 r = 0 (n2 ) n2 me2
r is in discrete values.
6 By C K Cheung
For H-atom,
r = 0.529( n2) x 10 – 10 m
If n = 1, r1 = 0.529 A
If n = 2, r2 = 2.11 A = 4 r1
Also, the energy of the system:
1 (e)(e) PE = 4 0 r
1 1 e2 KE = mv2 m( ) 2 2 40mr
e2 Total energy = E = PE + KE = - 80r
me4 1 E = - 2 2 2 - ( 2 ) 8 0 h n n e.g.
For max. E n = ______
For min. E n = ______
7 By C K Cheung
Consider:
n5 E5
n4 E4
n3 E3
n2 E2
n1 E1
1 1 3 E12 - ( ) = 22 12 4 5 E23 36
E12 > E23 > …………….
As n E 0 continuous
Hence:
E
Formula:
1 En = - 13.6 ( ) eV n2
8 By C K Cheung
1 eV = ______J Second Theory
No electron radiates energy so long as it remains in one of the orbital energy states, and that radiation occurs only when an electron goes from a higher energy state to a lower one, the energy of the quantum of radiation = hf.
n2
E = En2 – En1 = hf
n1
me4 1 1 c ( ) hf 2 2 2 2 = h( ) 8 0 h n1 n2
1 me4 1 1 wave number ( )( ) 2 3 2 2 8 0 h c n1 n2
Rydberg constant R, R=1.0973x107m-1
9 By C K Cheung e.g. The diagram shown gives some levels of an atom. In the unexcited state the levels above - 10.4 eV are unoccupied. ( Given: h = 6.6x10 - 34 Js, C = 3x108 ms-1, e = 1.6x10 -19C)
0 eV
- 1.6eV
- 3.7eV energy in eV
- 5.5eV
- 10.4eV
a/ What is the ionization energy of the atom?
Ionization energy = 0 – (-10.4) = 10.4 eV
b/ What change is taking place if radiation of wavelength 141 nm is emitted?
Ans.: - 1.6eV to - 10.4 eV
10 By C K Cheung e.g. a/ Find the energy required to excite a hydrogen atom from the ground state to the n = 4 state. 1 1 E = 13.6 ( )eV 12 4 2 1 1 = 13.6 ( )x1.6x10 19 C 12 4 2 = 2.04x10-18 J
b/ How many different possibilities of spectral line emission are there for the atom when the electron goes from n = 4 to the ground state ?
Note:
n=4
n=3 possible number of spectral n=2 lines = 6
n=1
11 By C K Cheung Evidence of energy levels
1/ Optical line spectra (optical ~ visible region) A particular line ( or a set of lines ) in an optical emission spectrum indicates the presence of a particular frequency emitted in a transition of e ( directly or in stages ) between energy levels
2/ X-ray spectra
Photoelectric Effect Source of variable & known frequency ( f ) vacuum A Emitter collector
V
Results of Experiment ( Laws of Photoelectric Emission )
1/ When the incident light is monochromatic, the number of photoelectrons emitted per second ( current I ) is proportional to the light intensity ( I’). Such an emission occurs effectively instantaneously. I/ A V, f = constant
I’/Wm -2
Wave theory: the incident light energy is uniformly distributed amongst the free electrons in the emitter, we should predict time delays of ~ 103 seconds.
12 By C K Cheung
2/ The KE of the emitted electrons varies from 0 to a maximum value. This definite maximum depends only on the frequency of the light, and not on its intensity. ..\powerpoint\atomic structure\photoelectric effect1.ppt
I /A
f = constant
2I’
I’
V/V Vs 0
Vs: stopping potential 1 mv 2 eV 2 max s
Wave theory: we feel hotter in sunlight than that in moonlight, K.E. of the emitted e should depend on light intensity.
13 By C K Cheung 3/ Electrons are not emitted when the light has a frequency lower than a certain threshold value fo. The value of fo varies from metal to metal.
I/A I’ = constant A B
f/Hz fo fo’
Wave theory: cannot predict the existence of fo!
Explanation
Planck’s Theory (1900) When radiation was emitted or absorbed, the emitting oscillator always showed a discrete sudden change of E, where E = hf.
Einstein’s Theory (1905) Extended Planck’s original idea by suggesting that E.M. wave could exhibit particle ( called photon ) behavior
1/ Electron emission is the result of a direct collision between an electron and a photon, so there is no time delay before emission starts ..\powerpoint\atomic structure\photoelectric effect2.ppt
14 By C K Cheung 2/ Light intensity number of photon arriving per m2 per second but not energy of individual photon .
I’ , f
2I’ , f
3/ fo depends on the material because each material requires a certain minimum energy ( called work function , W ) to free an electron.
W = hfo
15 By C K Cheung Formula:
hf
1 mv 2 2
W
1 hf = W + mv 2 2 1 hf = hfo + mv 2 2 hf = hfo + eVs
This formula was verified by Millikan in 1916
Vs/V
h slope = e
f/Hz fo’
16