Comparative Studies of Luminescence in Copepods and Other Pelagic Marine Animals*
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Bioluminescence of the Poecilostomatoid Copepod Oncaea Conifera
l MARINE ECOLOGY PROGRESS SERIES Published April 22 Mar. Ecol. Prog. Ser. Bioluminescence of the poecilostomatoid copepod Oncaea conifera Peter J. Herring1, M. I. ~atz~,N. J. ~annister~,E. A. widder4 ' Institute of Oceanographic Sciences, Deacon Laboratory, Brook Road Wormley, Surrey GU8 5UB, United Kingdom 'Marine Biology Research Division 0202, Scripps Institution of Oceanography, La Jolla, California 92093, USA School of Biological Sciences, University of Birmingham, Edgbaston. Birmingham B15 2TT, United Kingdom Harbor Branch Oceanographic Institution, 5600 Old Dixie Highway, Fort Pierce, Florida 34946, USA ABSTRACT: The small poecilostomatoid copepod Oncaea conifera Giesbrecht bears a large number of epidermal luminous glands, distributed primarily over the dorsal cephalosome and urosome. Bio- luminescence is produced in the form of short (80 to 200 ms duration) flashes from withrn each gland and there IS no visible secretory component. Nevertheless each gland opens to the exterior by a simple valved pore. Intact copepods can produce several hundred flashes before the luminescent system is exhausted. Individual flashes had a maximum measured flux of 7.5 X 10" quanta s ', and the flash rate follows the stimulus frequency up to 30 S" Video observations show that ind~vidualglands flash repeatedly and the flash propagates along their length. The gland gross morphology is highly variable although each gland appears to be unicellular. The cytoplasm contains an extensive endoplasmic reticulum. 0. conifera swims at Reynolds numbers of 10 to 50, and is normally associated with surfaces (e.g. marine snow). We suggest that the unique anatomical and physiological characteristics of the luminescent system arc related to the specialised ecological niche occupied by this species. -

Fishery Bulletin/U S Dept of Commerce National Oceanic
NEW RECORDS OF ELLOBIOPSIDAE (PROTISTA (INCERTAE SEDIS» FROM THE NORTH PACIFIC WITH A DESCRIPTION OF THALASSOMYCES ALBATROSSI N.SP., A PARASITE OF THE MYSID STILOMYSIS MAJOR BRUCE L. WINGl ABSTRACT Ten species of ellobiopsids are currently known to occur in the North Pacific Ocean-three on mysids and seven on other crustaceans. Thalassomyces boschmai parasitizes mysids of genera Acanthomysis, Neomysis, and Meterythrops from the coastal waters of Alaska, British Columbia, and Washington. Thalassomyces albatrossi n.sp. is described as a parasite of Stilomysis major from Korea. Thalassomyces fasciatus parasitizes the pelagic mysids Gnathophausia ingens and G. gracilis from Baja California and southern California. Thalassomyces marsupii parasitizes the hyperiid amphipods Parathemisto pacifica and P. libellula and the lysianassid amphipod Cypho caris challengeri in the northeastern Pacific. Thalassomyces fagei parasitizes euphausiids of the genera Euphausia and Thysanoessa in the northeastern Pacific from the southern Chukchi Sea to southern California, and occurs off the coast of Japan in the western Pacific. Thalassomyces capillosus parasitizes the decapod shrimp Pasiphaea pacifica in the northeastern Pacific from Alaska to Oregon, while Thalassomyces californiensis parasitizes Pasiphaea emarginata from central California. An eighth species of Thalassomyces parasitizing pasiphaeid shrimp from Baja California remains undescribed. Ellobiopsis chattoni parasitizes the calanoid copepods Metridia longa and Pseudocalanus minutus in the coastal waters of southeastern Alaska. Ellobiocystis caridarum is found frequently on the mouth parts ofPasiphaea pacifica from southeastern Alaska. An epibiont closely resembling Ellobiocystis caridarum has been found on the benthic gammarid amphipod Rhachotropis helleri from Auke Bay, Alaska. Where sufficient data are available, notes on variability, seasonal occurrence, and effects on the hosts are presented for each species of ellobiopsid. -

Ocean Deoxygenation and Copepods: Coping with Oxygen Minimum Zone Variability
Biogeosciences, 17, 2315–2339, 2020 https://doi.org/10.5194/bg-17-2315-2020 © Author(s) 2020. This work is distributed under the Creative Commons Attribution 4.0 License. Ocean deoxygenation and copepods: coping with oxygen minimum zone variability Karen F. Wishner1, Brad Seibel2, and Dawn Outram1 1Graduate School of Oceanography, University of Rhode Island, Narragansett, RI 02882, USA 2College of Marine Science, University of South Florida, St. Petersburg, FL 33701, USA Correspondence: Karen F. Wishner ([email protected]) Received: 27 September 2019 – Discussion started: 28 October 2019 Revised: 31 March 2020 – Accepted: 2 April 2020 – Published: 24 April 2020 Abstract. Increasing deoxygenation (loss of oxygen) of compression concept). These distribution depths changed by the ocean, including expansion of oxygen minimum zones tens to hundreds of meters depending on the species, oxygen (OMZs), is a potentially important consequence of global profile, and phenomenon. For example, at the lower oxycline, warming. We examined present-day variability of vertical the depth of maximum abundance for Lucicutia hulsemannae distributions of 23 calanoid copepod species in the East- shifted from ∼ 600 to ∼ 800 m, and the depth of diapause for ern Tropical North Pacific (ETNP) living in locations with Eucalanus inermis shifted from ∼ 500 to ∼ 775 m, in an ex- different water column oxygen profiles and OMZ inten- panded OMZ compared to a thinner OMZ, but remained at sity (lowest oxygen concentration and its vertical extent in similar low oxygen levels in -

Tesis Estructura Comunitaria De Copepodos .Pdf
Universidad de Concepción Dirección de Postgrado Facultad de Ciencias Naturales y Oceanográficas Programa de Magister en Ciencias mención Oceanografía Estructura comunitaria de copépodos pelágicos asociados a montes submarinos de la Dorsal Juan Fernández (32-34°S) en el Pacífico Sur Oriental Tesis para optar al grado de Magíster en Ciencias con mención en Oceanografía PAMELA ANDREA FIERRO GONZÁLEZ CONCEPCIÓN-CHILE 2019 Profesora Guía: Pamela Hidalgo Díaz Departamento de Oceanografía, Facultad de Ciencias Naturales y Oceanográficas Universidad de Concepción Profesor Co-guía: Rubén Escribano Departamento de Oceanografía, Facultad de Ciencias Naturales y Oceanográficas Universidad de Concepción La Tesis de “Magister en Ciencias con mención en Oceanografía” titulada “Estructura comunitaria de copépodos pelágicos asociados a montes submarinos de la Dorsal Juan Fernández (32-34°S) en el Pacífico sur oriental”, de la Srta. “PAMELA ANDREA FIERRO GONZÁLEZ” y realizada bajo la Facultad de Ciencias Naturales y Oceanográficas, Universidad de Concepción, ha sido aprobada por la siguiente Comisión de Evaluación: Dra. Pamela Hidalgo Díaz Profesora Guía Universidad de Concepción Dr. Rubén Escribano Profesor Co-Guía Universidad de Concepción Dr. Samuel Hormazábal Miembro de la Comisión Evaluadora Pontificia Universidad Católica de Valparaíso Dr. Fabián Tapia Director Programa de Magister en Oceanografía Universidad de Concepción ii A Juan Carlos y Sebastián iii AGRADECIMIENTOS Agradezco a quienes con su colaboración y apoyo hicieron posible el desarrollo y término de esta tesis. En primer lugar, agradezco a los miembros de mi comisión de tesis. A mi profesora guía, Dra. Pamela Hidalgo, por apoyarme y guiarme en este largo camino de formación académica, por su gran calidad humana, contención y apoyo personal. -

Bioluminescence As an Ecological Factor During High Arctic Polar Night Heather A
www.nature.com/scientificreports OPEN Bioluminescence as an ecological factor during high Arctic polar night Heather A. Cronin1, Jonathan H. Cohen1, Jørgen Berge2,3, Geir Johnsen3,4 & Mark A. Moline1 Bioluminescence commonly infuences pelagic trophic interactions at mesopelagic depths. Here receie: 01 pri 016 we characterize a vertical gradient in structure of a generally low species diversity bioluminescent ccepte: 14 Octoer 016 community at shallower epipelagic depths during the polar night period in a high Arctic ford with in Puise: 0 oemer 016 situ bathyphotometric sampling. Bioluminescence potential of the community increased with depth to a peak at 80 m. Community composition changed over this range, with an ecotone at 20–40 m where a dinofagellate-dominated community transitioned to dominance by the copepod Metridia longa. Coincident at this depth was bioluminescence exceeding atmospheric light in the ambient pelagic photon budget, which we term the bioluminescence compensation depth. Collectively, we show a winter bioluminescent community in the high Arctic with vertical structure linked to attenuation of atmospheric light, which has the potential to infuence pelagic ecology during the light-limited polar night. Light and vision play a large role in interactions among organisms in both the epipelagic (0–200 m) and mesope- lagic (200–1000 m) realms1,2. Eye structure and function in these habitats is commonly adapted for photon capture in the underwater light feld, with increasing specialization in the mesopelagic3. To avoid visual detection, species in epi- and mesopelagic habitats employ cryptic strategies such as transparency4 and counter-illumination5,6, along with diel vertical migration7,8, to remain hidden from potential predators. -

Vertical Distribution of Calanus Spp. and Metridia Longa at Four Arctic Locations
Marine Biology Research, 2008; 4: 193Á207 ORIGINAL ARTICLE Vertical distribution of Calanus spp. and Metridia longa at four Arctic locations MALIN DAASE1, KETIL EIANE1,3, DAG L. AKSNES2 & DANIEL VOGEDES1 1The University Centre in Svalbard, Longyearbyen, Norway, 2Department of Biology, University of Bergen, Bergen, Norway, and 3The University College in Bodø, Norway Abstract We investigated the vertical distribution of Calanus finmarchicus, C. glacialis, C. hyperboreus and Metridia longa at four locations around the archipelago of Svalbard in autumn. The older and larger copepodites of Calanus spp. were generally located deeper in the water column. Differences in vertical distribution between stations partly reflected a southÁnorth gradient in developmental progress with higher abundance of older stages in the southern locations. The C. finmarchicus and M. longa observations were consistent with the hypothesis that the developmental stages distributed according to certain preferences for light intensity, and different optical properties at the four locations are likely to have affected the vertical distributions. Diel vertical migration was only observed for older developmental stages of M. longa while young stages of M. longa remained in deep waters both day and night. A mortality index indicated that non-migrating Calanus spp. suffered higher mortality than migrating M. longa. Key words: Calanus, Metridia longa, mortality, optics, vertical distribution Introduction along the vertical axis in the sea is to some extent due to the rapid attenuation of light in water (Jerlov The depth distribution of many copepods in cold- 1968). This sets up thermal stratification and limits temperate regions is characterized by a strong seasonality that is closely related to the annual cycle the depth range available for positive primary produc- in primary production (Vinogradov 1997). -

Molecular Species Delimitation and Biogeography of Canadian Marine Planktonic Crustaceans
Molecular Species Delimitation and Biogeography of Canadian Marine Planktonic Crustaceans by Robert George Young A Thesis presented to The University of Guelph In partial fulfilment of requirements for the degree of Doctor of Philosophy in Integrative Biology Guelph, Ontario, Canada © Robert George Young, March, 2016 ABSTRACT MOLECULAR SPECIES DELIMITATION AND BIOGEOGRAPHY OF CANADIAN MARINE PLANKTONIC CRUSTACEANS Robert George Young Advisors: University of Guelph, 2016 Dr. Sarah Adamowicz Dr. Cathryn Abbott Zooplankton are a major component of the marine environment in both diversity and biomass and are a crucial source of nutrients for organisms at higher trophic levels. Unfortunately, marine zooplankton biodiversity is not well known because of difficult morphological identifications and lack of taxonomic experts for many groups. In addition, the large taxonomic diversity present in plankton and low sampling coverage pose challenges in obtaining a better understanding of true zooplankton diversity. Molecular identification tools, like DNA barcoding, have been successfully used to identify marine planktonic specimens to a species. However, the behaviour of methods for specimen identification and species delimitation remain untested for taxonomically diverse and widely-distributed marine zooplanktonic groups. Using Canadian marine planktonic crustacean collections, I generated a multi-gene data set including COI-5P and 18S-V4 molecular markers of morphologically-identified Copepoda and Thecostraca (Multicrustacea: Hexanauplia) species. I used this data set to assess generalities in the genetic divergence patterns and to determine if a barcode gap exists separating interspecific and intraspecific molecular divergences, which can reliably delimit specimens into species. I then used this information to evaluate the North Pacific, Arctic, and North Atlantic biogeography of marine Calanoida (Hexanauplia: Copepoda) plankton. -

Bioscien Ceslibr Aryunive Rsityof Alaskaf Airbank S
Early life history of Metridia pacifica brodsky (Copepoda: Calanoida) from the southeastern Bering Sea and Gulf of Alaska Item Type Thesis Authors Pinchuk, Alexei I. Download date 07/10/2021 06:44:04 Link to Item http://hdl.handle.net/11122/5039 EARLY LIFE HISTORY OF METRIDIA PACIFICA BRODSKY, (COPEPODA: CALONIDA) FORM THE SOUTHEASTERN BERING SEA AND GULF OF ALASKA By: Alexei I. Pincuck BIOSCIENCES LIBRARYUNIVERSITY OF ALASKA FAIRBANKS EARLY LIFE HISTORY OF METRIDIA PACIFICA BRODSKY, (COPEPODA: CALANOIDA) FROM THE SOUTHEASTERN BERING SEA AND GULF OF ALASKA By Alexei I. Pinchuk RECOMMENDED: SjLiSitr Advisory Committee Chair /JjrUlsiA*- C i Program Head APPPROVED: Dean, School of Fisheries and Ocean Sciences Dean of the Graduate School Date EARLY LIFE HISTORY OF METRIDIA PACIFICA BRODSKY, (COPEPODA CALANOIDA) FROM THE SOUTHEASTERN BERING SEA AND GULF OF ALASKA A THESIS Presented to the Faculty of the University of Alaska Fairbanks in Partial Fulfillment of the Requirements for the'Degree of MASTER OF SCIENCE By Alexei I, Pinchuk Fairbanks, Alaska BIOSCI August 1997 QL 444 €72 P56 1997 BIOSCIENCES LIBRARY 3 ABSTRACT The ontogenetic morphological changes of naupliar stages of Metrldia pacifica, an important prey taxon for larval walleye pollock, were described to facilitate their identification from field samples and to clarify some uncertainties in existing descriptions of co-occurring genera. Clutch sizes and sperm storage potential were determined for females captured from the southeastern Bering Sea and Gulf of Alaska. None of the females produced more than one egg clutch in captivity. The mean clutch size was 12-13 eggs.d-1 for females from both sites and there was no relationship between body size and number of eggs per clutch or egg diameter, Intermolt periods for the egg through N IV stages were 4 9-120 hours for animals reared at 3°C, 33-69 hours at 6°C and 27-74 hours at 9°C. -

Observing Copepods Through a Genomic Lens James E Bron1*, Dagmar Frisch2, Erica Goetze3, Stewart C Johnson4, Carol Eunmi Lee5 and Grace a Wyngaard6
Bron et al. Frontiers in Zoology 2011, 8:22 http://www.frontiersinzoology.com/content/8/1/22 DEBATE Open Access Observing copepods through a genomic lens James E Bron1*, Dagmar Frisch2, Erica Goetze3, Stewart C Johnson4, Carol Eunmi Lee5 and Grace A Wyngaard6 Abstract Background: Copepods outnumber every other multicellular animal group. They are critical components of the world’s freshwater and marine ecosystems, sensitive indicators of local and global climate change, key ecosystem service providers, parasites and predators of economically important aquatic animals and potential vectors of waterborne disease. Copepods sustain the world fisheries that nourish and support human populations. Although genomic tools have transformed many areas of biological and biomedical research, their power to elucidate aspects of the biology, behavior and ecology of copepods has only recently begun to be exploited. Discussion: The extraordinary biological and ecological diversity of the subclass Copepoda provides both unique advantages for addressing key problems in aquatic systems and formidable challenges for developing a focused genomics strategy. This article provides an overview of genomic studies of copepods and discusses strategies for using genomics tools to address key questions at levels extending from individuals to ecosystems. Genomics can, for instance, help to decipher patterns of genome evolution such as those that occur during transitions from free living to symbiotic and parasitic lifestyles and can assist in the identification of genetic mechanisms and accompanying physiological changes associated with adaptation to new or physiologically challenging environments. The adaptive significance of the diversity in genome size and unique mechanisms of genome reorganization during development could similarly be explored. -

Molecular Evidence on Evolutionary Switching from Particle-Feeding To
JOURNAL OF NATURAL HISTORY, 2016 http://dx.doi.org/10.1080/00222933.2016.1155779 Molecular evidence on evolutionary switching from particle-feeding to sophisticated carnivory in the calanoid copepod family Heterorhabdidae: drastic and rapid changes in functions of homologues Takeshi Hirabayashia, Susumu Ohtsukaa, Makoto Urataa, Ko Tomikawab and Hayato Tanakaa aTakehara Marine Science Station, Graduate School of Biosphere Science, Hiroshima University, Hiroshima, Japan; bGraduate School of Education, Hiroshima University, Higashi-Hiroshima, Japan ABSTRACT ARTICLE HISTORY The oceanic calanoid copepod family Heterorhabdidae is unique Received 10 January 2015 in that it comprises both particle-feeding and carnivorous genera Accepted 21 January 2016 with some intermediate taxa. Both morphological and molecular KEYWORDS (nuclear 18S and 28S rRNA) phylogenetic analyses of the family Heterorhabdidae; mandible; suggest that an evolutionary switch in feeding strategy, from poison; rRNA; evolution transitions of typical particle-feeding through some intermediate modes to sophisticated carnivory, might have occurred with the development of a specially designed ‘poison’ injection system. In view of the small amount of genetic differentiation among genera in this family, the switching of feeding modes and re-colonization from deep to shallow waters might have occurred over a short geological period since the Middle to Late Miocene. Introduction The feeding habits and modes of planktonic calanoid copepods have been investigated since the 1910s (Esterly 1916; Lebour 1922;Cannon1928) because of their ecological importance for energy flow in aquatic ecosystems (Huys and Boxshall 1991). Since the 1980s, many studies adopting high-speed cinematography have revealed that particle- Downloaded by [University of Cambridge] at 04:16 26 May 2016 feeders employ suspension feeding rather than filter feeding (Alcaraz et al. -
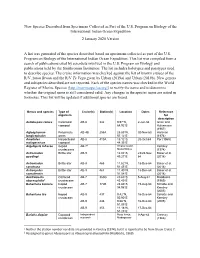
New Species Described from Specimens Collected As Part of the U.S
New Species Described from Specimens Collected as Part of the U.S. Program on Biology of the International Indian Ocean Expedition 2 January 2020 Version A list was generated of the species described based on specimens collected as part of the U.S. Program on Biology of the International Indian Ocean Expedition. This list was compiled from a search of publications cited by scientists involved in the U.S. Program on Biology and publications held by the Smithsonian Institution. The list includes holotypes and paratypes used to describe species. The cruise information was checked against the list of known cruises of the R/V Anton Bruun and the R/V Te Vega given by Urban (2019a) and Urban (2019b). New genera and subspecies described are not reported. Each of the species names was checked in the World Register of Marine Species (http://marinespecies.org/) to verify the name and to determine whether the original name is still considered valid. Any changes in the species name are noted in footnotes. This list will be updated if additional species are found. Genus and species Type of Cruise(s) Station(s) Location Dates Reference organism for description Aetideopsis retusa Calanaoid AB-6 342 9.97°S, 2-Jun-64 Grice and copepod 64.92°E Hulsemann (1967) Aglaophamus Polychaete AB-4B 255A 25.83°N, 30-Nov-63 Hartman longicephalus worm 57.12°E (1974) Ameliotes Harpacticoid AB-8 410A 15.12°S, 20-Oct-64 Por (1969) malagassicus copepod 44.35°E Argeiopsis inhacae isopod AB-71 Inhaca Island, Kensley crustaceans Mozambique (1974) Astrocladus Brittle star AB-9 12.83°S, 23/26-Nov- Baker et al. -

Four New Species of Heteromysis (Crustacea: Mysida) from Public Aquaria in Hawaii, Florida, and Western to Central Europe
European Journal of Taxonomy 735: 133–175 ISSN 2118-9773 https://doi.org/10.5852/ejt.2021.735.1247 www.europeanjournaloftaxonomy.eu 2021 · Wittmann K.J. & Abed-Navandi D. This work is licensed under a Creative Commons Attribution License (CC BY 4.0). Research article urn:lsid:zoobank.org:pub:F1CE3697-319D-4D02-A99F-11A0E16A8743 Four new species of Heteromysis (Crustacea: Mysida) from public aquaria in Hawaii, Florida, and Western to Central Europe Karl J. WITTMANN 1,* & Daniel ABED-NAVANDI 2 1 Medical University of Vienna, Department of Environmental Health, Kinderspitalgasse 15, 1090 Vienna, Austria. 2 Haus des Meeres – Aqua Terra Zoo, Fritz Grünbaum Platz 1, 1060 Vienna, Austria. * Corresponding author: [email protected] 2 Email: [email protected] 1 urn:lsid:zoobank.org:author:C90E7BC4-A27A-4B41-93F3-6224D17795FF 2 urn:lsid:zoobank.org:author:179B83B0-8C8B-4DF5-8986-4227B8E1BB9B Abstract. Four new species of the subgenus Heteromysis (Olivemysis) were detected in material from (sub)-tropical aquaria in six public aquarium institutions around the globe. Modifications of pleopods by spines represent the strongest structural complex used for differentiation within this subgenus: male pleopods 1–4 modified in H. smithsoniana sp. nov., male pleopods 2–4 plus female pleopod 2 in H. hornimani sp. nov. and H. waikikensis sp. nov. Additional important diagnostic characters are provided by the antennulae, uropods, and telson. The male of H. sixi sp. nov. represents a very rare case within the genus Heteromysis by having only pleopod 2 modified by flagellate spines. The definition of the subgenus Olivemysis is modified in order to include H.