The Origins of the Vocal Brain in Humans
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Pharnygeal Arch Set - Motor USMLE, Limited Edition > Neuroscience > Neuroscience
CNs 5, 7, 9, 10 - Pharnygeal Arch Set - Motor USMLE, Limited Edition > Neuroscience > Neuroscience PHARYNGEAL ARCH SET, CNS 5, 7, 9, 10 • They are derived from the pharyngeal (aka branchial) arches • They have special motor and autonomic motor functions CRANIAL NERVES EXIT FROM THE BRAINSTEM CN 5, the trigeminal nerve exits the mid/lower pons.* CN 7, the facial nerve exits the pontomedullary junction.* CN 9, the glossopharyngeal nerve exits the lateral medulla.* CN 10, the vagus nerve exits the lateral medulla.* CRANIAL NERVE NUCLEI AT BRAINSTEM LEVELS Midbrain • The motor trigeminal nucleus of CN 5. Nerve Path: • The motor division of the trigeminal nerve passes laterally to enter cerebellopontine angle cistern. Pons • The facial nucleus of CN 7. • The superior salivatory nucleus of CN 7. Nerve Path: • CN 7 sweeps over the abducens nucleus as it exits the brainstem laterally in an internal genu, which generates a small bump in the floor of the fourth ventricle: the facial colliculus • Fibers emanate from the superior salivatory nucleus, as well. Medulla • The dorsal motor nucleus of the vagus, CN 10 • The inferior salivatory nucleus, CN 9 1 / 3 • The nucleus ambiguus, CNs 9 and 10. Nerve Paths: • CNs 9 and 10 exit the medulla laterally through the post-olivary sulcus to enter the cerebellomedullary cistern. THE TRIGEMINAL NERVE, CN 5  • The motor division of the trigeminal nerve innervates the muscles of mastication • It passes ventrolaterally through the cerebellopontine angle cistern and exits through foramen ovale as part of the mandibular division (CN 5[3]). Clinical Correlation - Trigeminal Neuropathy THE FACIAL NERVE, CN 7  • The facial nucleus innervates the muscles of facial expression • It spans from the lower pons to the pontomedullary junction. -

Probing Forebrain to Hindbrain Circuit Functions in Xenopus
Received: 15 November 2016 | Accepted: 16 November 2016 DOI 10.1002/dvg.22999 REVIEW Probing forebrain to hindbrain circuit functions in Xenopus Darcy B. Kelley1 | Taffeta M. Elliott2 | Ben J. Evans3 | Ian C. Hall4 | Elizabeth C. Leininger5 | Heather J. Rhodes6 | Ayako Yamaguchi7 | Erik Zornik8 1Department of Biological Sciences, Columbia University, New York, New York Abstract 10027 The vertebrate hindbrain includes neural circuits that govern essential functions including breath- 2Department of Psychology, New Mexico ing, blood pressure and heart rate. Hindbrain circuits also participate in generating rhythmic motor Tech, Socorro, New Mexico 87801 patterns for vocalization. In most tetrapods, sound production is powered by expiration and the 3 Department of Biology, McMaster circuitry underlying vocalization and respiration must be linked. Perception and arousal are also University, Hamilton, Ontario, Ontario linked; acoustic features of social communication sounds—for example, a baby’scry—can drive L8S4K1, Canada autonomic responses. The close links between autonomic functions that are essential for life and 4Department of Biology, Benedictine University, Lisle, Illinois vocal expression have been a major in vivo experimental challenge. Xenopus provides an opportu- 5Department of Biology, St. Mary’s College, nity to address this challenge using an ex vivo preparation: an isolated brain that generates vocal St. Mary’s City, Maryland 29686 and breathing patterns. The isolated brain allows identification and manipulation of hindbrain vocal 6Department of Biology, Denison University, circuits as well as their activation by forebrain circuits that receive sensory input, initiate motor Granville, Ohio 43023 patterns and control arousal. Advances in imaging technologies, coupled to the production of Xen- 7 Department of Biology, University of Utah, opus lines expressing genetically encoded calcium sensors, provide powerful tools for imaging Salt Lake City, Utah 84112 neuronal patterns in the entire fictively behaving brain, a goal of the BRAIN Initiative. -

Validation of the Cortical Homunculus Using Functional Mri
VALIDATION OF THE CORTICAL HOMUNCULUS USING FUNCTIONAL MRI Rushil Anirudh Arizona State University School of Electrical, Computer & Energy Engineering ABSTRACT within the motor strip that correspond to the motor function that is being studied. The motor strip in the frontal lobe of the Human Brain is re- sponsible for all bodily movements or “motor functions”. In this paper we attempt to map the motor cortex of the Human Brain using fMRI data. The advantages of mapping it are sev- eral fold ranging from therapy to epilepsy and stroke patients, to understanding the development of these motor functions from birth. We try to recreate the homunculus, a concept which showed how the real estate of the motor cortex was distributed across these motor functions. Index Terms— fMRI, Primary Motor Cortex, Homuncu- lus. 1. INTRODUCTION The Human brain is the most important organ of the body, however we are still not entirely clear about how or why it works the way it does. While understanding the semantics behind our actions will require us to understand the entire human brain, we attempt to solve a much more low level problem of what happens when humans engage in day to day actions. To be more precise, this involves studying the Pri- mary Motor Cortex or the Motor Strip which is responsible Fig. 1. The idea of the Cortical homunculus as created by for all our motor functions, from our tongue to walking, run- Wilder Penfield. ning etc. Penfield and Boldrey [1] formulated the concept of the Cortical Homunculus which is a pictorial representation of the anatomical divisions of the motor strip. -

Adrenergic Activation of Cardiac Preganglionic Neurons in Nucleus
bioRxiv preprint doi: https://doi.org/10.1101/276998; this version posted January 8, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Adrenergic agonist induces rhythmic firing in quiescent cardiac preganglionic neurons in nucleus ambiguous via activation of intrinsic membrane excitability Isamu Aiba and Jeffrey L. Noebels Department of Neurology, Baylor College of Medicine Houston TX 77030 Correspondence: Department of Neurology, Baylor College of Medicine, One Baylor Plaza, Houston ,Texas 77030. Tel: 713 798 5862, email: [email protected]. Tel: 713 798 5860, email: [email protected] Running Head: Adrenergic activation of cardiac vagal neurons 1 bioRxiv preprint doi: https://doi.org/10.1101/276998; this version posted January 8, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract Cholinergic vagal nerves projecting from neurons in the brainstem nucleus ambiguus (NAm) play a predominant role in cardiac parasympathetic pacemaking control. Central adrenergic signaling modulates the tone of this vagal output; however the exact excitability mechanisms are not fully understood. We investigated responses of NAm neurons to adrenergic agonists using in vitro mouse brainstem slices. Preganglionic NAm neurons were identified by Chat-tdtomato fluorescence in young adult transgenic mice and their cardiac projection confirmed by retrograde dye tracing. Juxtacellular recordings detected sparse or absent spontaneous action potentials (AP) in NAm neurons. However bath application of epinephrine or norepinephrine strongly and reversibly activated most NAm neurons regardless of their basal firing rate. -

Somatotopic Mapping of the DALL
King’s Research Portal DOI: 10.1093/cercor/bhy050 Document Version Publisher's PDF, also known as Version of record Link to publication record in King's Research Portal Citation for published version (APA): Dall'Orso, S., Steinweg, J., Allievi, A. G., Edwards, A. D., Burdet, E., & Arichi, T. (2018). Somatotopic Mapping of the Developing Sensorimotor Cortex in the Preterm Human Brain. Cerebral Cortex, 28(7), 2507-2515. https://doi.org/10.1093/cercor/bhy050 Citing this paper Please note that where the full-text provided on King's Research Portal is the Author Accepted Manuscript or Post-Print version this may differ from the final Published version. If citing, it is advised that you check and use the publisher's definitive version for pagination, volume/issue, and date of publication details. And where the final published version is provided on the Research Portal, if citing you are again advised to check the publisher's website for any subsequent corrections. General rights Copyright and moral rights for the publications made accessible in the Research Portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognize and abide by the legal requirements associated with these rights. •Users may download and print one copy of any publication from the Research Portal for the purpose of private study or research. •You may not further distribute the material or use it for any profit-making activity or commercial gain •You may freely distribute the URL identifying the publication in the Research Portal Take down policy If you believe that this document breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. -

Gene Therapy for Recurrent Laryngeal Nerve Injury
G C A T T A C G G C A T genes Review Gene Therapy for Recurrent Laryngeal Nerve Injury Koji Araki * ID , Hiroshi Suzuki, Kosuke Uno ID , Masayuki Tomifuji and Akihiro Shiotani Department of Otolaryngology-Head & Neck Surgery, National Defense Medical College, Saitama 3598513, Japan; [email protected] (H.S.); [email protected] (K.U.); [email protected] (M.T.); [email protected] (A.S.) * Correspondence: [email protected]; Tel.: +81-4-2995-1686 Received: 2 June 2018; Accepted: 20 June 2018; Published: 25 June 2018 Abstract: Recurrent laryngeal nerve (RLN) injury has considerable clinical implications, including voice and swallowing dysfunction, which may considerably impair the patient’s quality of life. Recovery of vocal fold movement is an essential novel treatment option for RLN injury. The potential of gene therapy for addressing this issue is highly promising. The target sites for RLN gene therapy are the central nervous system, nerve fibers, laryngeal muscles, and vocal cord mucosa. Gene transduction has been reported in each site using viral or non-viral methods. The major issues ensuing after RLN injury are loss of motoneurons in the nucleus ambiguus, degeneration and poor regeneration of nerve fibers and motor end plates, and laryngeal muscle atrophy. Gene therapy using neurotrophic factors has been assessed for most of these issues, and its efficacy has been reported. Another important matter for functional vocal fold movement recovery is misdirected regeneration, in which the wrong neurons may innervate other laryngeal muscles, where even if innervation is reestablished, proper motor function is not restored. -
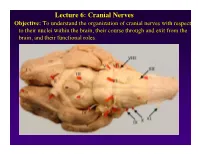
Lecture 6: Cranial Nerves
Lecture 6: Cranial Nerves Objective: To understand the organization of cranial nerves with respect to their nuclei within the brain, their course through and exit from the brain, and their functional roles. Olfactory Eye Muscles 3, 4 &6 Cranial Nerves 1-7 I overview Table, Page 49 II Lecture notes Cranial Nerves and their Functions V Trigeminal VII Facial VIII IX X XII XI Cranial Nerves 8-12 Overview sternocephalic I. Factors Responsible for the Complex Internal Organization of the Brain Stem-> leads to altered location of cranial nerve nuclei in adult brain stem 1. Development of the Fourth Ventricle a. Medulla and Pons develop ventral to the 4th ventricle cerebellum b. Alar plate is displaced lateral to basal plate 4 Medulla Developing Neural Tube 2. Cranial nerve nuclei form discontinuous columns Rostral 12 SE Page 48 Notes 3. Some cranial nerve nuclei migrate from their primitive embryonic positions (e.g., nuclei of V and VII) Facial N. Factors responsible for the complex internal organization of the brainstem: 4) Special senses develop in association with the brain stem. Nuclei of special senses 5) Development of the cerebellum and its connections Cerebellum II. Cranial Nerve Nuclei: Nucleus = column of neuron cell bodies. Efferent nuclei are composed of cell bodies of alpha or gamma motor neurons (SE) or preganglionic parasympathetic neurons (VE). III. Motor Efferent Nuclei (Basal Plate Derivatives): 1. SE (Somatic Efferent) Nuclei: SE neurons form two longitudinally oriented but discontinuous columns of cell bodies in the brain stem. Neurons that comprise these columns are responsible for innervating all of the skeletal musculature of the head. -

The Singing Athlete Chapter 2
2. BRAIN BASICS Think about the best singing experience of your life: it may have taken place in a performance, a voice lesson, or just driving around town. Can you remember the euphoria you felt when, suddenly, everything worked the way it should? This state of effortless flow is what you are seeking every time you open your mouth to sing. And yet, no matter how vocally skilled you get, there are inevitably days when singing feels challenging. The issues you experience on a “bad voice day” may not be noticeable to those who hear you, but you’ll be aware that you’re not at the top of your game. If you know how well you can sing, why isn’t it perfect every time? The reason is simple: humans are survival-based organisms. Staying alive is more important than performing well. The majority of your brain doesn’t care if you can float a high C; it cares that you survive the attempt. On a subconscious level, your brain is constantly assessing your environment for threatening signals. Is that person walking toward you friendly or crazy? Do you hear the sound of fireworks or gunshots? Are you smelling a cozy bonfire or a forest ablaze? These assessments occur in an ancient, reflexive part of your nervous system that we will call the first brain (Fig. 2.1). This first brain, which includes the brainstem and the cerebellum, is always asking the question, “Is it safe?” Let’s say you’ve been struggling to belt a high note without strain. -

Krubitzer * and Tony J
Special Issue: Time in the Brain Review The Combinatorial Creature: Cortical Phenotypes within and across Lifetimes 1, 2 Leah A. Krubitzer * and Tony J. Prescott The neocortex is one of the most distinctive structures of the mammalian brain, Highlights yet also one of the most varied in terms of both size and organization. Multiple There are multiple time-scales that are relevant for understanding how a given processes have contributed to this variability, including evolutionary mecha- phenotype emerges. Brains change nisms (i.e., alterations in gene sequence) that alter the size, organization, and across large, evolutionary time-scales, shorter time-scales such as genera- connections of neocortex, and activity dependent mechanisms that can also tions, and within the life of an individual. modify these same features. Thus, changes to the neocortex can occur over different time-scales, including within a single generation. This combination of Any given phenotype is a combination of genes involved in brain and body devel- genetic and activity dependent mechanisms that create a given cortical opment, behavior,and the environmentin phenotype allows the mammalian neocortex to rapidly and flexibly adjust to which an individual develops. A similar different body and environmental contexts, and in humans permits culture to phenotype in different species may be due to homology, but can also be the impact brain construction. result of a different combination of factors. Brain, Body, and Environment Interactions There are several constraints that restrict In this review we discuss the evolution and development of mammalian neocortex. We examine the avenues along which evolution of the brain and body can proceed. -

Differentiation of the Bulbar Motor Nuclei and the Coincident Develop- Ment of Associated Root Fibers in the Rsbbit
DIFFERENTIATION OF THE BULBAR MOTOR NUCLEI AND THE COINCIDENT DEVELOP- MENT OF ASSOCIATED ROOT FIBERS IN THE RSBBIT DONALD L. KIMMEL Department of Anatomy, University of Michigan,’ Aim Arbor THIRTY-ONE FIGURES CONTENTS Introduction ........................................................ 83 Material and methods ................................................ 84 General survey of the literature ....................................... 85 Description of material studied ........................................ 86 Somatic efferent component. Its central and peripheral development .... 86 The hypoglossal ............................................. 86 The abdueens ............................................... 96 Visceral efferent component. Its central and peripheral development .... 103 The vago-accessory ........................................... 103 The glossopharyngeal ........................................ 124 The facial .................................................. 129 The trigemiiial .............................................. 137 Geiicrxldisrussion .................................................... 143 INTRODUCTION This present study concerns itself primarily with the onto- genetic development of the nuclear centers of bulbar cranial nerves in the rabbit and with the embryonic and adult distri- butions of their branches. Its purpose is to show that the central development proceeds stage for stage with the pro- A dissertation submitted in partial fulfillment of the requirements for the degree of doctor of philosophy -

Of the Human Cortical Somatosensory System
bioRxiv preprint doi: https://doi.org/10.1101/675611; this version posted June 21, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. The "creatures" of the human cortical somatosensory system N. Saadon-Grosman,1,2* Y. Loewenstein,3,4 † S. Arzy 1,2† 1Neuropsychiatry Lab, Department of Neurology, Hadassah Hebrew University Medical Center, Jerusalem, 9112001, Israel. 2 Department of Medical Neurobiology, Faculty of Medicine, Hadassah Hebrew University Medical School, 9112001, Jerusalem, Israel. 3 The Edmond and Lily Safra Center for Brain Sciences, The Alexander Silberman Institute of Life Sciences, 4 Dept. of Cognitive Sciences and The Federmann Center for the Study of Rationality, The Hebrew University, 9190401 , Jerusalem, Israel. † These authors contributed equally to this work. * Correspondence: [email protected] bioRxiv preprint doi: https://doi.org/10.1101/675611; this version posted June 21, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract Penfield’s description of the “homunculus”, a “grotesque creature” with large lips and hands and small trunk and legs depicting the representation of body-parts within the primary somatosensory cortex (S1), is one of the most prominent contributions to the neurosciences. Since then, numerous studies have identified additional body- parts representations outside of S1. -

Nuclear Architecture in the Medulla Oblongata of the Adult African Giant Pouched Rat (Cricetomys Gambianus, Waterhouse - 1840)
Int. J. Morphol., 29(2):382-388, 2011. Nuclear Architecture in the Medulla Oblongata of the Adult African Giant Pouched Rat (Cricetomys gambianus, Waterhouse - 1840) Arquitectura Nuclear en la Médula Oblonga de la Rata Gigante de Carillos Africana Adulta (Cricetomys gambianus, Waterhouse - 1840) *Ibe, C. S; *Onyeanusi, B. I.; *Hambolu, J. O. & **Ayo, J. O. IBE, C. S.; ONYEANUSI, B. I.; HAMBOLU, J. O. & AYO, J. O. Nuclear architecture in the medulla oblongata of the adult African giant pouched rat (Cricetomys gambianus, Waterhouse - 1840). Int. J. Morphol., 29(2):382-388, 2011. SUMMARY: The architecture of cranial and non-cranial nerve nuclei in the medulla oblongata of the African giant pouched rat was studied by means of light microscopy. Serial sections of the medulla oblongata, in coronal and saggital planes, were stained with the cresyl fast violet and silver stains, respectively. Sections in the saggital plane were used as a guide, while coronal sections were used to identify the nuclei in the rostrocaudal extent of the medulla oblongata. With the obex serving as the landmark, nuclei rostral and caudal to the obex were delineated. Cranial nerve nuclei whose architecture were defined were the motor nucleus of hypoglossal nerve, motor nucleus of vagus nerve, cochlear nucleus, vestibular nucleus and nucleus ambiguus, while non-cranial nerve nuclei identified were the olivary nucleus, solitary tract nucleus, gracile nucleus, cuneate nucleus, spinal nucleus of trigeminal nerve, motor nucleus of corpus trapezoideum, lateral nucleus of reticular formation and gigantocellular nucleus. The olivary nucleus was the most prominent nucleus, while the solitary tract nucleus was faint, and thus, less developed.