Dephosphorylation of Threonine-14 and Tyrosine-15
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

9. Atypical Dusps: 19 Phosphatases in Search of a Role
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Digital.CSIC Transworld Research Network 37/661 (2), Fort P.O. Trivandrum-695 023 Kerala, India Emerging Signaling Pathways in Tumor Biology, 2010: 185-208 ISBN: 978-81-7895-477-6 Editor: Pedro A. Lazo 9. Atypical DUSPs: 19 phosphatases in search of a role Yolanda Bayón and Andrés Alonso Instituto de Biología y Genética Molecular, CSIC-Universidad de Valladolid c/ Sanz y Forés s/n, 47003 Valladolid, Spain Abstract. Atypical Dual Specificity Phosphatases (A-DUSPs) are a group of 19 phosphatases poorly characterized. They are included among the Class I Cys-based PTPs and contain the active site motif HCXXGXXR conserved in the Class I PTPs. These enzymes present a phosphatase domain similar to MKPs, but lack any substrate targeting domain similar to the CH2 present in this group. Although most of these phosphatases have no more than 250 amino acids, their size ranges from the 150 residues of the smallest A-DUSP, VHZ/DUSP23, to the 1158 residues of the putative PTP DUSP27. The substrates of this family include MAPK, but, in general terms, it does not look that MAPK are the general substrates for the whole group. In fact, other substrates have been described for some of these phosphatases, like the 5’CAP structure of mRNA, glycogen, or STATs and still the substrates of many A-DUSPs have not been identified. In addition to the PTP domain, most of these enzymes present no additional recognizable domains in their sequence, with the exception of CBM-20 in laforin, GTase in HCE1 and a Zn binding domain in DUSP12. -

The Involvement of Ubiquitination Machinery in Cell Cycle Regulation and Cancer Progression
International Journal of Molecular Sciences Review The Involvement of Ubiquitination Machinery in Cell Cycle Regulation and Cancer Progression Tingting Zou and Zhenghong Lin * School of Life Sciences, Chongqing University, Chongqing 401331, China; [email protected] * Correspondence: [email protected] Abstract: The cell cycle is a collection of events by which cellular components such as genetic materials and cytoplasmic components are accurately divided into two daughter cells. The cell cycle transition is primarily driven by the activation of cyclin-dependent kinases (CDKs), which activities are regulated by the ubiquitin-mediated proteolysis of key regulators such as cyclins, CDK inhibitors (CKIs), other kinases and phosphatases. Thus, the ubiquitin-proteasome system (UPS) plays a pivotal role in the regulation of the cell cycle progression via recognition, interaction, and ubiquitination or deubiquitination of key proteins. The illegitimate degradation of tumor suppressor or abnormally high accumulation of oncoproteins often results in deregulation of cell proliferation, genomic instability, and cancer occurrence. In this review, we demonstrate the diversity and complexity of the regulation of UPS machinery of the cell cycle. A profound understanding of the ubiquitination machinery will provide new insights into the regulation of the cell cycle transition, cancer treatment, and the development of anti-cancer drugs. Keywords: cell cycle regulation; CDKs; cyclins; CKIs; UPS; E3 ubiquitin ligases; Deubiquitinases (DUBs) Citation: Zou, T.; Lin, Z. The Involvement of Ubiquitination Machinery in Cell Cycle Regulation and Cancer Progression. 1. Introduction Int. J. Mol. Sci. 2021, 22, 5754. https://doi.org/10.3390/ijms22115754 The cell cycle is a ubiquitous, complex, and highly regulated process that is involved in the sequential events during which a cell duplicates its genetic materials, grows, and di- Academic Editors: Kwang-Hyun Bae vides into two daughter cells. -

PTEN-L Is a Novel Protein Phosphatase for Ubiquitin Dephosphorylation to Inhibit PINK1–Parkin-Mediated Mitophagy
www.nature.com/cr www.cell-research.com ARTICLE OPEN PTEN-L is a novel protein phosphatase for ubiquitin dephosphorylation to inhibit PINK1–Parkin-mediated mitophagy Liming Wang1, Yik-Lam Cho1, Yancheng Tang2, Jigang Wang1, Jung-Eun Park3, Yajun Wu4, Chunxin Wang 5, Yan Tong6, Ritu Chawla1, Jianbin Zhang1,7, Yin Shi1, Shuo Deng1, Guang Lu1, Yihua Wu8, Hayden Weng-Siong Tan1,9, Pornteera Pawijit1, Grace Gui-Yin Lim10, Hui-Ying Chan1,9, Jingzi Zhang11, Lei Fang11, Hanry Yu1,12,13, Yih-Cherng Liou6, Mallilankaraman Karthik1, Boon-Huat Bay4, Kah-Leong Lim1,10, Siu-Kwan Sze 3, Celestial T. Yap1 and Han-Ming Shen1,9 Mitophagy is an important type of selective autophagy for specific elimination of damaged mitochondria. PTEN-induced putative kinase protein 1 (PINK1)-catalyzed phosphorylation of ubiquitin (Ub) plays a critical role in the onset of PINK1–Parkin-mediated mitophagy. Phosphatase and tensin homolog (PTEN)-long (PTEN-L) is a newly identified isoform of PTEN, with addition of 173 amino acids to its N-terminus. Here we report that PTEN-L is a novel negative regulator of mitophagy via its protein phosphatase activity against phosphorylated ubiquitin. We found that PTEN-L localizes at the outer mitochondrial membrane (OMM) and overexpression of PTEN-L inhibits, whereas deletion of PTEN-L promotes, mitophagy induced by various mitochondria-damaging agents. Mechanistically, PTEN-L is capable of effectively preventing Parkin mitochondrial translocation, reducing Parkin phosphorylation, maintaining its closed inactive conformation, and inhibiting its E3 ligase activity. More importantly, PTEN-L reduces the level of phosphorylated ubiquitin (pSer65-Ub) in vivo, and in vitro phosphatase assay confirms that PTEN-L dephosphorylates pSer65-Ub via its protein phosphatase activity, independently of its lipid phosphatase function. -
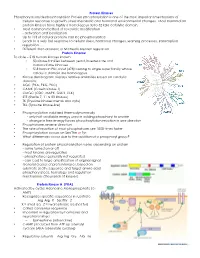
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

The Regulatory Roles of Phosphatases in Cancer
Oncogene (2014) 33, 939–953 & 2014 Macmillan Publishers Limited All rights reserved 0950-9232/14 www.nature.com/onc REVIEW The regulatory roles of phosphatases in cancer J Stebbing1, LC Lit1, H Zhang, RS Darrington, O Melaiu, B Rudraraju and G Giamas The relevance of potentially reversible post-translational modifications required for controlling cellular processes in cancer is one of the most thriving arenas of cellular and molecular biology. Any alteration in the balanced equilibrium between kinases and phosphatases may result in development and progression of various diseases, including different types of cancer, though phosphatases are relatively under-studied. Loss of phosphatases such as PTEN (phosphatase and tensin homologue deleted on chromosome 10), a known tumour suppressor, across tumour types lends credence to the development of phosphatidylinositol 3--kinase inhibitors alongside the use of phosphatase expression as a biomarker, though phase 3 trial data are lacking. In this review, we give an updated report on phosphatase dysregulation linked to organ-specific malignancies. Oncogene (2014) 33, 939–953; doi:10.1038/onc.2013.80; published online 18 March 2013 Keywords: cancer; phosphatases; solid tumours GASTROINTESTINAL MALIGNANCIES abs in sera were significantly associated with poor survival in Oesophageal cancer advanced ESCC, suggesting that they may have a clinical utility in Loss of PTEN (phosphatase and tensin homologue deleted on ESCC screening and diagnosis.5 chromosome 10) expression in oesophageal cancer is frequent, Cao et al.6 investigated the role of protein tyrosine phosphatase, among other gene alterations characterizing this disease. Zhou non-receptor type 12 (PTPN12) in ESCC and showed that PTPN12 et al.1 found that overexpression of PTEN suppresses growth and protein expression is higher in normal para-cancerous tissues than induces apoptosis in oesophageal cancer cell lines, through in 20 ESCC tissues. -

Purification and Characterization of Two Members of the Protein
PURIFICATION AND CHARACTERIZATION OF TWO MEMBERS OF THE PROTEIN TYROSINE PHOSPHATASE FAMILY: DUAL SPECIFICITY PHOSPHATASE PVP AND LOW MOLECULAR WEIGHT PHOSPHATASE WZB By Paula A. Livingston A Thesis Submitted to the Faculty of The Charles E. Schmidt College of Science in Partial Fulfillment for the Degree of Master of Science Florida Atlantic University Boca Raton, Florida December 2009 i ACKNOWLEDGMENTS The author wishes to thank her thesis advisor, Dr. Stefan Vetter, and the members of her committee, Dr. Estelle Leclerc and Dr. Predrag Cudic, for their advice and support throughout her years at Florida Atlantic University. The author also wishes to thank her family for their unending support and encouragement without which this would not have been possible. The author would also like to thank her nephew and niece, Griffin and Skye, for inspiring her throughout her entire graduate experience. iii ABSTRACT Author: Paula A. Livingston Title: Purification and Characterization of Two Members of the Protein Tyrosine Phosphatase Family: Dual Specificity Phosphatase PVP and Low Molecular Weight Phosphatase WZB Institution: Florida Atlantic University Thesis Advisor: Dr. Stefan W. Vetter Degree: Master of Science Year: 2009 Two protein tyrosine phosphatases, dual specificity phosphatase PVP and low molecular weight phosphatase WZB were purified and characterized. PVP was expressed as inclusion bodies and a suitable purification and refolding method was devised. Enzyme kinetics revealed that p-nitrophenylphosphate and β-naphthyl phosphate were substrates with KM of 4.0mM and 8.1mM respectively. PVP showed no reactivity towards phosphoserine. Kinetic characterization of WZB showed that only p- nitrophenylphosphate was a substrate with no affinity for β-naphthyl phosphate and phosphoserine. -

Protein Phosphatase 1-Α Regulates AS160 Ser588 and Thr642
2606 Diabetes Volume 65, September 2016 Pragya Sharma,1 Edward B. Arias,1 and Gregory D. Cartee1,2,3 Protein Phosphatase 1-a Regulates AS160 Ser588 and Thr642 Dephosphorylation in Skeletal Muscle Diabetes 2016;65:2606–2617 | DOI: 10.2337/db15-0867 Akt substrate of 160 kDa (AS160) phosphorylation on insulin resistance is crucial for whole-body insulin re- Thr642 and Ser588 by Akt is essential for insulin’s full ef- sistance and type 2 diabetes (1). Muscle insulin resistance fect on glucose transport. However, protein phosphory- is secondary, in large part, to defective GLUT4 transloca- lation is determined by the balance of actions by kinases tion and glucose transport (2). Insulin’s stimulation of glu- fi and phosphatases, and the speci c phosphatase(s) cose transport is triggered by a complex insulin-signaling controlling AS160 dephosphorylation is (are) unknown. pathway that begins with insulin’s binding to its receptor, Accordingly, we assessed roles of highly expressed leading to receptor autophosphorylation and activation of skeletal muscle serine/threonine phosphatases (PP1, receptor tyrosine kinase (2). The insulin receptor kinase PP2A, PP2B, and PP2C) on AS160 dephosphorylation. Preliminary screening of candidate phosphatases used phosphorylates insulin receptor substrate (IRS) proteins an AS160 dephosphorylation assay. Lysates from insu- on multiple tyrosine residues, resulting in IRS protein en- lin-stimulated skeletal muscle were treated with phar- gagement with phosphatidylinositol (PI) 3-kinase (PI3K), macological phosphatase inhibitors and assessed for that in turn, phosphorylates PI 4,5-bisphosphate to create AS160 Ser588 and Thr642 dephosphorylation. AS160 de- 3,4,5-trisphosphate (PIP3). The serine/threonine kinase phosphorylation on both phosphorylation sites was un- Akt is recruited to bind PIP3 and become activated second- altered by PP2B or PP2C inhibitors. -

A Protein Phosphatase 2Cα–Ca
1914 • The Journal of Neuroscience, February 23, 2005 • 25(8):1914–1923 Cellular/Molecular A Protein Phosphatase 2c␣–Ca2ϩ Channel Complex for Dephosphorylation of Neuronal Ca2ϩ Channels Phosphorylated by Protein Kinase C Dongjun Li, Fushun Wang, Meizan Lai, Yuan Chen, and Ji-fang Zhang Department of Physiology, Jefferson Medical College, Philadelphia, Pennsylvania 19107 Phosphorylation and dephosphorylation are primary means for rapid regulation of a variety of neuronal functions, such as membrane excitability, neurotransmitter release, and gene expression. Voltage-gated Ca 2ϩ channels are targets for phosphorylation by a variety of second messengers through activation of different types of protein kinases (PKs). Protein phosphatases (PPs), like PKs, are equally important in regulating Ca 2ϩ channels in neurons. However, much less is understood about whether and how a particular type of PP contributes to regulating neuronal Ca 2ϩ channel activities. This is primarily because of the lack of specific inhibitors/activators for different types of PPs, particularly the PP2c family. The functional roles of PP2c and its substrates in the brain remain virtually unknown. During our yeast two-hybrid screening, PP2c␣ was pulled out by both N- and P/Q-type Ca 2ϩ channel C termini. This raised the possibility that PP2c␣ might be associated with voltage-gated Ca 2ϩ channels for regulation of the Ca 2ϩ channel activity. Biochemical studies show ϩ that PP2c␣ binds directly to neuronal Ca 2 channels forming a functional protein complex in vivo. PP2c␣, unlike PP1, PP2a and PP2b, is more effective in dephosphorylation of neuronal Ca 2ϩ channels after their phosphorylation by PKC. In hippocampal neurons, disruption of the PP2c␣–Ca 2ϩ channel interaction significantly enhances the response of Ca 2ϩ channels to modulation by PKC. -

Faqs for DNA End Modification: Dephosphorylation
FAQs for DNA End Modification: Dephosphorylation 1. Does the DNA need to be purified after a restriction digest and prior to the dephosphorylation step? If the enzymes used in the restriction digest are heat inactivatable, then a purification step is not needed. If the enzymes used are not heat-inactivatable, then a clean-up step between the restriction digest and the dephosphorylation step is recommended. 2. Does the DNA need to be purified after the dephosphorylation step and prior to the ligation step? If you use rSAP or Antarctic Phosphatase, which are heat-inactivatable, then a clean- up step is not necessary. If the alkaline phosphatase used is CIP, which is not heat- inactivatable, then a clean-up step is necessary. 3. Which alkaline phosphatase, CIP, Antarctic or rSAP, works best? rSAP (NEB #M0371) is a superior choice because it combines the advantages of both CIP (NEB #M0290) and Antarctic Phosphatase (NEB #M0289). rSAP has a very high specific activity comparable to CIP, but CIP cannot be completely heat-inactivated. rSAP is heat-inactivated in 5 minutes, as is Antarctic Phosphatase, but rSAP is preferred over Antarctic Phosphatase because it does not require added zinc or any other co-factors. As a result, rSAP can be added directly to restriction enzyme digests. Additionally, after heat-inactivation of rSAP, it is not necessary to purify vector DNA prior to the ligation reaction. 4. Are the alkaline phosphatases active in NEBuffers? rSAP: rSAP is active in all restriction enzyme NEBuffers 1.1, 2.1, 3.1 and CutSmart™ Buffer, and can be added directly to digested DNA. -

AMP-Activated Protein Kinase: the Current Landscape for Drug Development
REVIEWS AMP-activated protein kinase: the current landscape for drug development Gregory R. Steinberg 1* and David Carling2 Abstract | Since the discovery of AMP-activated protein kinase (AMPK) as a central regulator of energy homeostasis, many exciting insights into its structure, regulation and physiological roles have been revealed. While exercise, caloric restriction, metformin and many natural products increase AMPK activity and exert a multitude of health benefits, developing direct activators of AMPK to elicit beneficial effects has been challenging. However, in recent years, direct AMPK activators have been identified and tested in preclinical models, and a small number have entered clinical trials. Despite these advances, which disease(s) represent the best indications for therapeutic AMPK activation and the long-term safety of such approaches remain to be established. Cardiovascular disease Dramatic improvements in health care coupled with identifying a unifying mechanism that could link these (CVD). A term encompassing an increased standard of living, including better nutri- changes to multiple branches of metabolism followed diseases affecting the heart tion and education, have led to a remarkable increase in the discovery that the AMP-activated protein kinase or circulatory system. human lifespan1. Importantly, the number of years spent (AMPK) provided a common regulatory mechanism in good health is also increasing2. Despite these positive for inhibiting both cholesterol (through phosphoryla- Non-alcoholic fatty liver disease developments, there are substantial risks that challenge tion of HMG-CoA reductase (HMGR)) and fatty acid (NAFLD). A very common continued improvements in human health. Perhaps the (through phosphorylation of acetyl-CoA carboxylase disease in humans in which greatest threat to future health is a chronic energy imbal- (ACC)) synthesis8 (BOx 1). -

Multiple Protein Phosphatases Are Required for Mitosis in Drosophila
Supplemental References S10. Margolis, S.S., Walsh, S., Weiser, D.C., Yoshida, M., Shenolikar, S., and Kornbluth, S. (2003). PP1 control of M phase entry exerted through 14-3-3-regulated Cdc25 dephosphorylation. Embo J 22, 5734-5745. S11. Margolis, S.S., Perry, J.A., Weitzel, D.H., Freel, C.D., Yoshida, M., Haystead, T.A., and Kornbluth, S. (2006). A role for PP1 in the Cdc2/Cyclin B-mediated positive feedback activation of Cdc25. Mol Biol Cell 17, 1779-1789. S12. Axton, J.M., Dombradi, V., Cohen, P.T., and Glover, D.M. (1990). One of the protein phosphatase 1 isoenzymes in Drosophila is essential for mitosis. Cell 63, 33-46. S13. Rogers, E., Bishop, J.D., Waddle, J.A., Schumacher, J.M., and Lin, R. (2002). The aurora kinase AIR-2 functions in the release of chromosome cohesion in Caenorhabditis elegans meiosis. J Cell Biol 157, 219-229. S14. Sassoon, I., Severin, F.F., Andrews, P.D., Taba, M.R., Kaplan, K.B., Ashford, A.J., Stark, M.J., Sorger, P.K., and Hyman, A.A. (1999). Regulation of Saccharomyces cerevisiae kinetochores by the type 1 phosphatase Glc7p. Genes Dev 13, 545-555. S15. Katayama, H., Zhou, H., Li, Q., Tatsuka, M., and Sen, S. (2001). Interaction and feedback regulation between STK15/BTAK/Aurora-A kinase and protein phosphatase 1 through mitotic cell division cycle. J Biol Chem 276, 46219-46224. S16. Ohashi, S., Sakashita, G., Ban, R., Nagasawa, M., Matsuzaki, H., Murata, Y., Taniguchi, H., Shima, H., Furukawa, K., and Urano, T. (2006). Phospho-regulation of human protein kinase Aurora-A: analysis using anti-phospho-Thr288 monoclonal antibodies. -

Dual-Specificity Phosphatases in Immunity and Infection
International Journal of Molecular Sciences Review Dual-Specificity Phosphatases in Immunity and Infection: An Update Roland Lang * and Faizal A.M. Raffi Institute of Clinical Microbiology, Immunology and Hygiene, Universitätsklinikum Erlangen, Friedrich-Alexander-Universität Erlangen-Nürnberg, 91054 Erlangen, Germany * Correspondence: [email protected]; Tel.: +49-9131-85-22979 Received: 15 May 2019; Accepted: 30 May 2019; Published: 2 June 2019 Abstract: Kinase activation and phosphorylation cascades are key to initiate immune cell activation in response to recognition of antigen and sensing of microbial danger. However, for balanced and controlled immune responses, the intensity and duration of phospho-signaling has to be regulated. The dual-specificity phosphatase (DUSP) gene family has many members that are differentially expressed in resting and activated immune cells. Here, we review the progress made in the field of DUSP gene function in regulation of the immune system during the last decade. Studies in knockout mice have confirmed the essential functions of several DUSP-MAPK phosphatases (DUSP-MKP) in controlling inflammatory and anti-microbial immune responses and support the concept that individual DUSP-MKP shape and determine the outcome of innate immune responses due to context-dependent expression and selective inhibition of different mitogen-activated protein kinases (MAPK). In addition to the canonical DUSP-MKP, several small-size atypical DUSP proteins regulate immune cells and are therefore also reviewed here. Unexpected and complex findings in DUSP knockout mice pose new questions regarding cell type-specific and redundant functions. Another emerging question concerns the interaction of DUSP-MKP with non-MAPK binding partners and substrate proteins.